9
Cosmetic Surgery
INTRODUCTION
Facial cosmetic surgery is a unique area of practice within the scope of oral and maxillofacial surgery in that interventions offered to the patient are typically not required from a medical standpoint but are considered elective procedures. Although some cosmetic procedures offer an adjunctive functional benefit such as the visual field improvement seen with upper blepharoplasty, by and large the goal of the therapy is to achieve a visual appearance that is more aesthetically pleasing to the patient. For this reason, patient tolerance of adverse outcomes is often quite low. The facial cosmetic surgeon must be adept at proper patient selection for cosmetic surgery, as well as prevention of complications and recognition of impending problems with prompt intervention when indicated. Once a complication has been recognized, appropriate intervention is performed in order to correct, or minimize, any adverse sequelae. The surgeon should allocate appropriate additional time to counsel the patient honestly on the nature of the complication, solutions that are available, likely outcomes, and a reasonable timetable until a final result is achievable. This strategy can lead to an acceptable result for the patient, and, often, to one of the more loyal patients to the cosmetic practice.
This chapter aims to outline the more common complications seen with cosmetic procedures of the head and neck. Management strategies will be discussed to minimize or eliminate long-term sequelae of these complications. Common facial cosmetic treatments will be grouped together as either conservative (minimally invasive), or surgical in nature.
CONSERVATIVE COSMETIC PROCEDURE COMPLICATIONS
1. Botulinum toxin
2. Injectable soft tissue volumizers and fillers
3. Nonablative and ablative cosmetic skin therapy
4. Ablative skin resurfacing
Botulinum Toxin
Cosmetic botulinum toxin therapy for the face is typically aimed at reducing mimetic rhytids. Additionally, by adjusting the vector forces of the facial muscles by selective paralysis, nonsurgical brow elevation or lifting of the commissures of the mouth can be achieved. The safety and efficacy of botulinum toxin is well established by the literature and by its extensive use in the population with low reported rate of complications.1,2 In 2007, there were 4.6 million patients treated with botulinum toxin therapy (BTX) and very few reports of complications as a result of the medication.3 Even when evaluating the literature on the use of BTX use for movement disorders such as cervical dystonia where doses reach 300 units, at much higher doses than in the case for cosmetic use (typically much less than 100 units), severe complications are rare.4 More commonly, there are unsatisfactory results as a result of administration errors or unanticipated effect. Since BTX does not cause permanent paralysis of the musculature, long-term complications are rare. This has contributed greatly to making this treatment the most popular of cosmetic procedures.
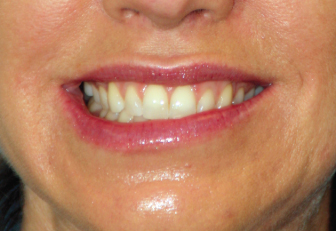
Due to its primary use to achieve muscle paralysis, nonselective muscle weakness comprises the most frequent distressing complication encountered following BTX administration. The three most common inadvertent muscle weaknesses seen are lid ptosis, brow droop, and lip droop (from weakness in the marginal mandibular branch of the facial nerve distribution after treatment of the depressor anguli oris muscle) (Fig. 9.1). Rarely, difficulty closing the eye may be seen from excessive BTX administration into the orbicularis oculi muscle.
Fig. 9.1. Asymmetric lip position following treatment of the depressor angulae oris muscle with BTX.
Upper Lid Ptosis
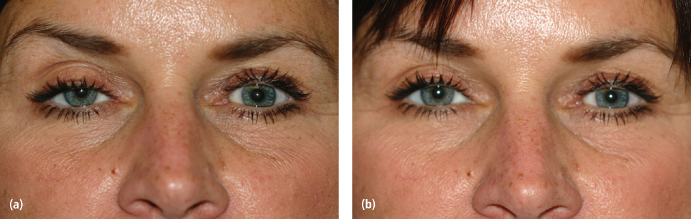
Ptosis of the upper lid is usually mild and is the result of the inadvertent blocking of Mueller’s muscle in the superior orbit. This complication can be avoided by an injection technique that places BTX in an upward direction at least 1 cm above the superior orbital rim. If ptosis is noted following BTX treatment, intervention may be considered if the patient has significant concerns. One therapy commonly recommended for symptomatic ptosis is the ophthalmic administration of apraclonidine (Iodipine), which is an alpha-adrenergic agonist agent (Fig. 9.2). This agent increases sympathetic tone to Mueller’s muscle. One to three drops daily of 0.5% ophthalmic solution in the affected eye should limit ptosis for approximately 4 hours.5
Fig. 9.2. (a) Iatrogenic lid ptosis after BTX treatment of corrugators. (b) Same patient 5 minutes after one drop of ophthalmic iopodine.
Brow and Lid Droop
Brow and lip droop result from the overaggressive treatment of the lateral frontalis muscle and depressor anguli oris muscle, respectively. Prevention of this problem is best achieved by undertreating these areas until the practitioner develops a sense of the BTX dose that is best tolerated by an individual patient. Unfortunately, when excessive weakness is noted, there is little option for treatment except to wait for the effects of BTX to diminish. One option is to treat the less weak side of the face with an additional small dose of BTX (1–2 units) in an attempt to achieve symmetry. The patient should be reassured that the undesired weakness typically only lasts 2–3 weeks, far less than the expected duration of usual BTX treatment (3–4 months).
Inability to Close the Eye
A far less frequent complication is the inadvertent block of the orbicularis oculi muscle leading to the inability to close the eye. Typically, a large field dose of BTX encompassing the lateral orbicularis oculi is the culprit initiating this complication. Avoidance of BTX doses in this area of greater than 5–15 units/side, in addition to proper placement technique, should help prevent this problem. If, after treatment with BTX, the patient is unable to close the eye, concern for preventing corneal damage must be considered. The eyelid should be taped shut to protect the globe and prescription of lubricating agents is initiated. It is important to remind the patient that the paralysis will be temporary. The ability to close the eye should return prior to the expected lifespan of the BTX cosmetic effect (3–4 months).
Injection Site Bleeding
Injection site bleeding is usually quite minor and is managed by pressure on the site. The area should not be rubbed or massaged, since this may disseminate the BTX to unintended areas or inactivate the BTX. Bruising with hematoma formation and skin discoloration may be quite distressful for the patient, but this complication has a self-limited course of a few weeks.
Injectable Soft Tissue Volumizers and Fillers
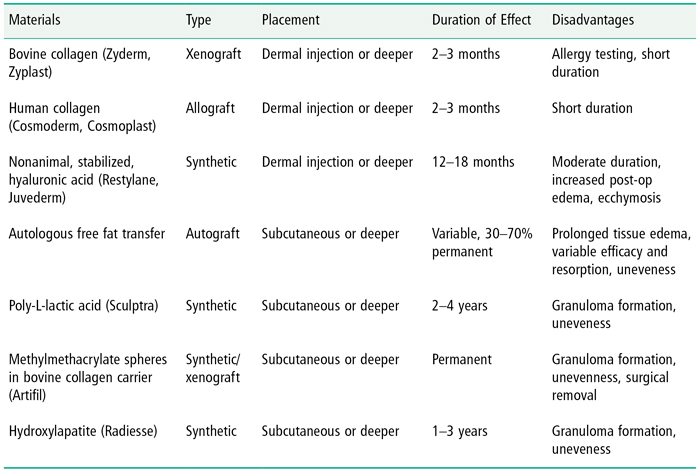
Soft tissue filler materials have been used for years for cosmetic purposes and represent one of the fastest growing areas within facial cosmetic practices. Fillers may be used to smooth folds and wrinkles, replace facial volume, or change actual facial morphology depending on the filler characteristics and placement location (Table 9.1). In the United States there are many choices available based upon patient need, anatomic area, and specific characteristics of the filler material. Although there is great variety in available fillers, many complications are common to all and will be discussed together. Other complications are more specific to individual fillers and will be discussed separately.
Table 9.1. Injectable Fillers Commonly used in the United States
Soft tissue fillers are generally considered to have an excellent safety profile, and most complications are not considered life threatening. However, some infrequent complications can lead to exceptional morbidity, and, of course, the patient with a high cosmetic demand can be devastated by an even minor complication. An interesting side effect of the relatively high safety quotient of fillers, and their perceived high profitability, is that an increasing number of medical and dental practitioners with very limited training have introduced filler placement into their practice. In certain communities it has become common for hairdressers and independent aestheticians to integrate the medical placement of fillers into their business practices. Without commenting on the legal or ethical ramifications of this trend, it should be expected that the cosmetic oral and maxillofacial surgeon may see more patients with filler complications from other providers in their region.
The undesirable result in the cosmetic patient can be due to expectations prior to treatment that were not met by the treatment, or by a truly objective unsatisfactory result. Either way, the cosmetic surgeon should have knowledge on treatment of various problems resulting from the injection of fillers and possible techniques for eliminating or decreasing the defect.
Inappropriate Location of Filler Placement
Early post-treatment “lumps,” “bumps,” and asymmetry are easily recognized problems by patients and surgeons, and are perhaps the most common complications seen with soft tissue fillers. In general, this complication results from injection of the filler material in an inappropriate location, or extension of the material beyond the intended boundaries of the specific site or rhytid, either spontaneously or induced by the patient by manipulating or rubbing the area. The timing of the appearance of the irregularity and the nature of the filler used can provide an important clue as to the source of the problem and the appropriate treatment to resolve it. This may require additional filler material to gently blend the area, or, in some cases, removal of the material surgically. It is best to plan appropriately and isolate the filler material to the specific area of concern to avoid this complication.
Injection Hematoma
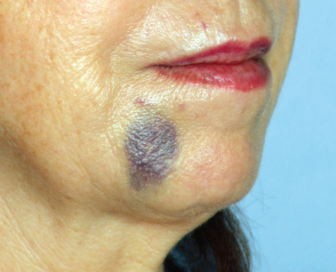
Since most fillers are placed using a needle, an injection hematoma at either the site of skin penetration or filler deposition must be considered as a possible cause of early skin irregularities in the area. Typically, injection hematomas will be noticed sometime after filler placement, either at the treatment visit, or within the next 24 hours (Fig. 9.3). The clinician can be fairly certain that if the treated area appears smooth and symmetric initially, but becomes asymmetric or uneven within 24 hours, a small hematoma or uneven edema is the likely cause. In the case of fillers that provide immediate substance (e.g., the hyaluronic acids), one must also consider that the patient may have manipulated the material into a new location if it is first recognized after the treatment visit, as discussed. If a hematoma is suspected, the patient should be reassured that improvement will occur within a few days. Manipulation and massage of the area should be avoided so as not to disturb the location of filler placement. Topically placed cold packs may limit the extent of the hematoma if recognized early. After 24 hours, warm compresses and topical arnica gel placement may help to decrease ecchymosis. Injection hematomas can be best avoided by keeping the number of penetrations of the needle through the dermal plexus to a minimum. If many areas are treated, a fresh needle should be used to prevent tearing of tissue from a dull needle tip. In planning the injection pathway, the surgeon should also consider the anatomical position of major vascular bundles and avoid them when possible.
Fig. 9.3. Injection hematoma 24 hours after hyaluronic acid filler placement.
Inappropriate placement of filler will usually be noticed almost immediately by the astute clinician when using substantive fillers such as the hyaluronic acids, collagens, hydroxyapatite or autologous fat. Poor placement of fillers that rely on the patient’s own collagen production to produce volume, such as poly-L-lactic acid, will usually develop late irregularities, after one or more months following placement. Avoidance of this problem is best achieved by considering a number of factors: (1) the recommended tissue depth placement of the filler and its relative viscoelastic properties compared to the regional soft tissues; (2) the relative thickness of the tissue overlying the area to be augmented and the ability to conceal minor irregularities of deep fillers; and (3) avoidance of excessive filler placement to an area in a single visit.
When a clinician recognizes that an irregularity, “lump,” “ bump,” or skin blanching has appeared immediately after placement of a substantive filler, vigorous massage of the area is recommended. This often resolves the undesired result and distributes the filler more evenly. If a large amount of filler has been carelessly injected to form the “lump,” consideration must given to either removing the filler via puncture and manual expression, or destruction of the filler agent. Asymmetry that is noticed post-treatment between the right and left side of the face may be caused either by a preexisting, but unrecognized, facial asymmetry, an asymmetric placement of filler, or possible injection hematoma. In the case of asymmetric filler placement, this outcome can be improved by additional injection in the “undertreated” side. A larger problem exists when the patient perceives an asymmetry and believes one side is overcorrected. The only resolution for this complication may be patient management and a tincture of time for the effect of the filler to diminish or consideration of techniques to remove the filler. For this reason it is recommended that in technically demanding areas such as the tear trough deficiency, the use of fillers with a shorter half-life is considered, and underfilling rather than overtreating the location is more prudent (Fig. 9.4). The patient should be made aware that it is always easier to add more filler to achieve a more desirable result than to try to remove filler material. Destruction or removal of fillers depends upon the chemical properties of the agent used. Hyaluronic acid fillers can to some degree be reversed, or their half-life dramatically shortened, by injection of hyaluronidase.6 Typically, 10–15 units of hyaluronidase is injected directly into the area of excess hyaluronic acid filler. A change is usually noticed by the patient on the following day. Additional hyaluronidase may be injected if needed. Excess collagen injection is often treated by vigorous massage and warm compresses. Older forms of collagen typically have a short half-life and resolve quickly. Newer, cross-linked collagens may be softened by injection of hyaluronidase, or steroid injection (0.1 cc of kenalog-10). Injectable hydroxyapatite (HA) that forms a visible lump can be a very difficult problem to resolve. Massage, warm compresses, and limited steroid injection may be helpful if initiated early. The surgeon may attempt to disrupt a persistent “lump” of hydroxyapatite by passing an 18 g needle transcutaneously and sweeping through the collection of HA particles with multiple passes. If this fails, the final options are either surgical removal or observation with the expectation that the irregularity may remain for several years. Surgical removal often reveals clumped calcium-like deposits spread throughout the underlying soft tissues. Clumping of hydroxyapatite may be minimized by using the recommended technique of the manufacturer to decrease the concentration and viscosity of the material. Occasionally it may be appropriate to add additional filler adjacent to a visible deposit to camouflage the area. It is best to consider using a reversible filler such as hyaluronic acid so that if an increasingly worse result is achieved, it can be returned to the level of the initial complication. Irregular deposits sometimes seen after autologous fat transfer are best avoided by only volumizing areas where thick soft tissue coverage over the augmented area exists. It is not recommended to transfer fat to areas of the face where there is thin soft tissue present over bone, such as over the lateral zygoma or the tear trough region. Irregular fat placement or resorption can create a difficult irregularity to manage. Vigorous massage, steroid injection, and other mesotherapy agent injections may have some success. Liposuction of the localized fat deposit can have an unpredictable result with irregularities. An adjacent camouflage injection with another filler agent is also an option.
Fig. 9.4. (a) Visible nodularity after attempted correction of tear trough deficiency with hydroxyapatite filler. (b) Lip irregularity after hyaluronic acid filler placement. Both complications were corrected after local injection with hyaluronidase, massage, and placement of camouflaging hyaluronic acid filler.
Nonsubstantive fillers rely on the patient’s natural collagen forming response in reaction to the filler material to create volume. Examples include poly-L-lactic acid and microdroplet silicone injections, and these typically do not form early irregularities; instead, this complication will often appear a month or more after treatment. The degree and dimension of collagen formation can be difficult to predict, making it important to place the material deeply where nodules may not be visible or to use small amounts superficially. Similar to fat transfer, extreme caution should be taken in areas with thin soft tissue covering bone. When visible nodules do appear that are not granulomatous in nature, management is directed at interrupting the collagen formation process. Injection directly into the nodule with low dose steroid is a first line option. Persistent nodules may respond to an intralesional low dose chemotherapy agent (e.g., 5-fluorouracil) or needle disruption. Surgical removal is usually the final option for management of this complication.
Filler Agent Allergic Reaction and Granuloma Formation
Allergic reactions range from minor to life threatening and may be possible with any of the soft tissue filler materials. Minor, local allergic reaction to filler treatment usually presents as an early or late finding of fiery erythema and edema immediately surrounding the area of injection. Infection due to inoculation with skin biofilm must be ruled out. Local allergic reaction often responds well to a tapered dose oral steroid regimen. Local injection with low dose steroid may be useful. If a hyaluronic acid has been used, destruction of the material with hyaluronidase should be considered. Occasionally the affected area may form a sterile abscess that should be drained and irrigated. Systemic anaphylactic reaction is possible, but fortunately is very rare. A systemic reaction should be treated aggressively to stabilize the patient with attention then turned to removing the causative material, if possible. Allergic reaction is most common with the fillers based on bovine collagen, which were more commonly used before the introduction of hyaluronic acid fillers and others. The use of bovine collagen (Zyderm®, Zyplast®) requires allergy testing, which is typically performed 30 days prior to filler placement. A small amount (0.1ml) of the bovine collagen is placed in the forearm; if no reaction is found after 30 days it is assumed filler placement can proceed without risk. This 30-day delay is recommended due to early experience with the material. Many patients were treated after a negative skin reaction evaluated after 6 hours. It was noted that despite negative skin reaction, 1.2–6.3% of patients experienced redness and swelling that lasted for months.7 This prolonged planning and delay in treatment is in direct contrast to the typical spontaneity of most cosmetic filler procedures used today.8 The Allergan website (the company that produces Zyderm and Zyplast) states the rate of allergic reaction to be approximately 1–2%.9
Granulomatous reactions are more commonly associated with the semi-permanent or permanent fillers such as poly-L-lactic acid, polymethylmethacrylate spheres, and microdroplet silicone injection. Foreign body reaction rarely appears earlier than 1 month after injection and has been reported to occur sometimes years later. Polymethylmethacrylate microspheres (ArteFill®/ArteSense®) are permanent fillers in a bovine collagen transport medium with a low complication profile. Previously, methylmethacrylate microspheres were produced and granulomatous reaction was a more commonly reported incidence. After changes in the production regimen and change of the transport medium from gelatin to bovine collagen, the foreign body granuloma formation rate decreased dramatically.10 Since ArteFill®/ArteSense® contain bovine collagen, allergy testing similar to Zyderm® and Zyplast® are necessary. A case was reported by Fischer of a patient who developed a sarcoid-like reaction after initiation of antiviral therapy for hepatitis C. The lesions were restricted to the glabellar region, nasolabial area, and lower lips. The patient received an injection of Artecoll® in those areas 10 years prior to initiation of antiviral therapy. The author concluded the low-grade foreign body reaction to the polymethylmethacrylate became exaggerated after treatment for hepatitis C had begun. The foreign body reaction resulted in ulceration of the glabellar region and a quite large nodule in the nasolabial area requiring excision.11 If foreign body granuloma is noted, an early intralesional steroid is initial treatment as described earlier. Intralesional injection with a chemotherapeutic agent such as 5-FU should also be considered. If the lesion is recalcitrant to intralesional injections then excision may be necessary. If the area allows, a fold or rhytid is used to hide the incision. If the granulomatous reaction extends to the skin, an ellipse should be planned to remove the affected area. If the granuloma is located deeply, a simple linear incision for access is recommended. The granulomatous chain is identified and removed and primary closure is obtained; ablative laser skin resurfacing of the incision may be considered to help blend the resulting scar.
Of all the fillers currently available, the use of silicone injection has been the most fiercely debated and has been the subject of controversy in both the scientific and public literature. Silicone has been used for multiple areas of the body and in different formulations. It continues to be used as facial cosmetic filler because it has excellent flow properties, is thought to have a “natural” feel, and is long lasting. The novice clinician is cautioned about it use because of its permanence. Most cosmetic surgeons who use silicone plan for multiple sessions with injection of small amounts at each visit until the desired effect is achieved. Once the material is placed, massage is prohibited since the material may flow through tissues and diminish its effect, cause asymmetry, or irregular clumping.
Silicone granulomas range from asymptomatic masses to painful erythematous and disfiguring lesions. Occasionally, the silicone masses can ulcerate, cause cellulitis or abscess, or a combination. Like polymethylmethacrylate, the granulomas may be surgically removed. Excision of silicone granulomas is often challenging and many times unsuccessful. Intralesional steroids are often used as the first line of treatment for silicone granulomas, although systemic steroids are another option. Minocycline or imiquinod are treatments used with reportedly good clinical success. Minocycline is prescribed at 100 mg twice daily until resolution of symptoms is observed. A trial decrease in dosage can be attempted after resolution of granulomas but may need to be reinitiated if exacerbation develops.12 Imiquinod 5% (Aldara®) is available in a cream and is applied twice daily with reportedly good success in the treatment of silicone granulomas.13 Pasternak has reported on the use of Etanercept, a tumor necrosis factor (TNF) inhibitor, in the treatment of recalcitrant silicone granulomas.14 Granulomatous reactions are thought to be a T-cell activation by TNF-α. This treatment requires subcutaneous injections biweekly for an extended period of time, and the patient must be tested for tuberculosis prior to initiation of treatment.15
Intra-arterial Injection of Filler Materials
Intra-arterial injection of filler material is an extremely rare occurrence but can have severe complications. Intra-arterial injection and its sequelae are theoretically possible with any filler and is the result of inadvertently entering an artery and forcing filler material either anterograde or retrograde along its vascular distribution. For this reason, it is recommended to only inject filler along a defined pathway during retraction of the needle. It is not recommended to inject ahead of the needle as it is advanced into tissue. If blanching of tissue is noticed, then the injection should be immediately discontinued. Case reports of facial skin necrosis and blindness have been reported in the literature;15–17 De Castro et al.18,19 described a case of extensive necrosis of the facial artery distribution after injection of polymethylmethacrylate. If occlusion of a vessel is encountered after filler is injected, close observation should be undertaken. Hospital admission with use of heparin and other anticoagulant methods has been employed with unknown benefit. If necrosis occurs then limitation of extension of the area affected is the goal. Judicious debridement and prevention of infection are begun until the time for attempts at secondary reconstruction. Other authors have described blindness after injection of the glabellar region thought to be due to retrograde injection into the ophthalmic artery distribution.15 If blindness or other ophthalmologic complications are suspected the patient should be referred to an ophthalmologist emergently for assessment in order to salvage sight or eye function.
Nonablative and Ablative Cosmetic Skin Therapy Complications
There are various modalities of therapy available to improve the appearance of the facial and neck skin. Goals of treatment involve the improvement or removal of pigmentations and dyspigmentations, telangectasias and vascular anomalies, tattoo removal, and improvement of age and actinic generated fine lines and rhytids. Therapies can be broadly divided into nonablative and ablative treatments depending upon their effect on the epidermis (Table 9.2).
Table 9.2. Managing Facelift Complications by Region of Incision
Nonablative, subsurface therapies include intense pulsed light, Nd-YAG, pulsed dye and the diode (585,1450 nm) lasers, and radiofrequency delivery systems that are used appropriately to treat dyspigmentations, tattoos, vascular anomalies, very fine rhytids, and loss of skin elasticity. These devices use epidermal surface cooling in combination with deeply penetrating wavelengths and energies that selectively target water-containing tissue, resulting in selective heating and subsequent thermal injury to the dermis. Complications typically relate to exuberant application of energy to the subsurface tissues and may be transitory or long lasting.
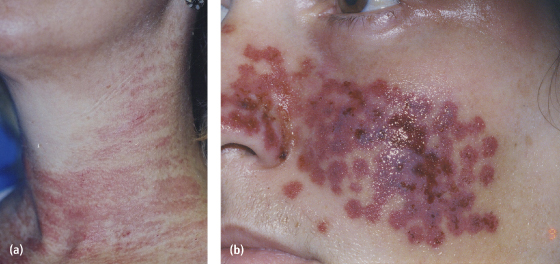
Edema is an expected post-treatment finding with most subsurface treatments. It is transitory in nature and usually lasts for 1 to 3 days. Purpura and blistering of the affected area are less common and probably represent an injudicious delivery of energy to the tissue (Fig. 9.5). Patients who normally use topical retin-A homecare should discontinue treatment for several days prior to treatment. If multiple treatments are scheduled for the patient, the clinician should keep an accurate record and change the energy parameters appropriately. Purpura is generally a self-limiting problem. Topical application of an arnica-containing gel may be recommended. Blisters should not be unroofed, but should be maintained as a “biologic dressing” and protected by a moist occlusive dressing and a nonadhering dressing (Telfa) until resolved.
Fig. 9.5. (a) Mild erythema post IPL treatment. (b) Erythema, purpura, and blistering post KTP laser treatment.
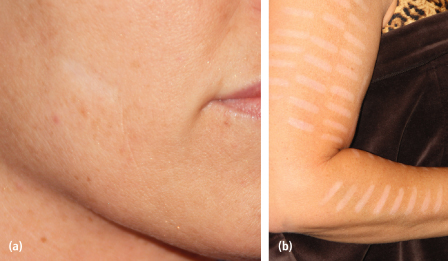
In the treatment of dyspigmentations there is a potential to worsen the initial lesion, or to change the surrounding tissue pigmentation thus creating a new problem. Most modalities for treating pigmentations require that the patient abstain from tanning or sun exposure for any noticeable extent prior to beginning therapy. Darker skin, and tanned skin, carries a higher risk of generalized damage to melanocytes leading to either temporary or permanent hypopigmentation (Fig. 9.6). Once hypopigmentation occurs, it is notoriously difficult to treat, so avoidance of excessive energy delivery or deferment of treatment in these patients is paramount. Treatment of hypopigmentation is directed at decreasing the general darkness of the skin surrounding the hypopigmented area. High-level sun block, 4% hydroquinone topically twice daily, or topical kojic acid may be helpful. A more limited, low-energy setting using the initial device can be helpful to decrease pigment in the unaffected area surrounding the hypopigmented site, although this has the potential to worsen the problem. Fortunately, most hypopigmentation from subsurface treatments improve with time. Occasionally, in instances of limited permanent hypopigmentation, a medical tattoo artist may be employed to permanently blend the area with surrounding skin. The tattooed skin lacks the ability to tan and thus may be more or less visible as the patient’s skin color continues to change with sun exposure. Reactive hyperpigmentation should be treated with topical hydroquinone or kojic acid.
Fig. 9.6. Hypopigmentation following IPL treatment of (a) face; (b) arm.
Subsurface Fat Atrophy and Irregularity
Development of subsurface fat atrophy and irregularity has been reported primarily in conjunction with older modalities of radiofrequency dermal tightening procedures.20 Utilizing current technologies and recommended energy settings this phenomena is easily avoided but remains as a cautionary reminder of untoward consequences from exuberant treatment. Attempts to correct subsurface irregularity are aimed at adding volume to the affected area. Injectable nonanimal stabilized hyaluronic acid (NASHA), autologous fat transfer, and subcision techniques have been tried with varying levels of success. Areas that exhibit dermal scar tethering to deeper tissues may require scar lysis to allow a potential space to form to accept a filler substance. An easy method of detecting scar adhesions below the dermis is to inject a volume of normal saline subcutaneously into the depressed area. If a “doughnut” of saline forms around the depression, it is prudent to plan on scar lysis prior to filler placement.
Allergic Reaction to Tattoo Removal
Allergic reaction to tattoo ink during the initial placement is well documented, but it also may occur during attempted tattoo removal. Tattoo inks, both temporary and permanent, contain a plethora of substances that can lead to an immediate or delayed reaction. Mercury- and chromium-containing materials are common offenders in permanent tattoo allergic reactions. During the process of tattoo removal by laser, the ink is liberated and is freely exposed to the immune system where it can be labeled as foreign. Though not a common occurrence, with few reports in the literature, the clinician should be aware of this possibility when confronted with a patient who exhibits skin reactions after tattoo removal. In the few case reports, patients have been referred to dermatologists to confirm the diagnosis. During subsequent treatments the patients were treated with prednisone in order to mitigate the immune reaction.21,22
Ablative Skin Resurfacing
Ablative skin laser resurfacing and chemexfoliation offer a relatively noninvasive option for patients desiring improvement to actinic changes of the skin, especially fine lines and rhytids. However, unlike the previously discussed subsurface procedures, this spectrum of treatment inherently involves the controlled destruction of the epidermis, in addition to possible injury of the dermis. Complications are more common and can be quite disfiguring if not avoided or managed appropriately in the preoperative and postoperative phases of care. Preoperative evaluation of skin color and type are essential to anticipate potential complications and modify treatment based upon these criteria.
Herpetic Infection
Herpetic infection after laser treatment is a well-known complication. It carries the potential for permanent scarring. Preoperative evaluation of the patient who requests laser surgery should include questions regarding known history of herpes infection or previous symptoms consistent with herpes infection. It is prudent when performing laser therapy to begin antiviral suppressive therapy for herpes prior to surgery. Therapy is continued after treatment until the treated area has re-epithelialized, typically for 10–14 days. Assessment in the early postoperative period may be confounded by the absence of blistering, with pain being the presenting symptom. If herpetic infection is suspected, or confirmed by Tzank smear, than institution of higher dose levels of antiviral medications typical for zoster is provided.
Persistent Erythema
Persistent erythema lasting over 6 months from the time of treatment should trigger investigation since most mild erythema should resolve within 2–3 months (Fig./>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


