4
Maxillofacial Trauma
INTRODUCTION
Complications in the treatment of cranio-maxillofacial injuries occur regularly and, even in the most experienced of hands, should be expected. Many, but not all, complications can be prevented by adherence to proper surgical technique and established treatment protocols, and by recognizing the potential for functional or aesthetic complications. While expected surgical complications such as infection and nonunion of mandibular fractures are still associated with excellent outcomes if managed properly, those related to inaccurate reduction of midfacial and orbital fractures that result in facial widening and/or enophthalmos, respectfully, are exceedingly difficult to correct secondarily. The ultimate outcome for patients with cranio-maxillofacial trauma is thus less dependent upon the occurrence of complications and more upon the recognition of pitfalls during treatment and management of complications when they inevitably occur.
SOFT TISSUE INJURIES
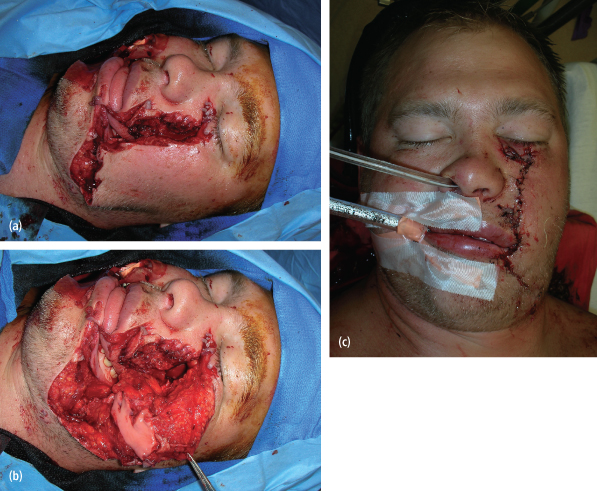
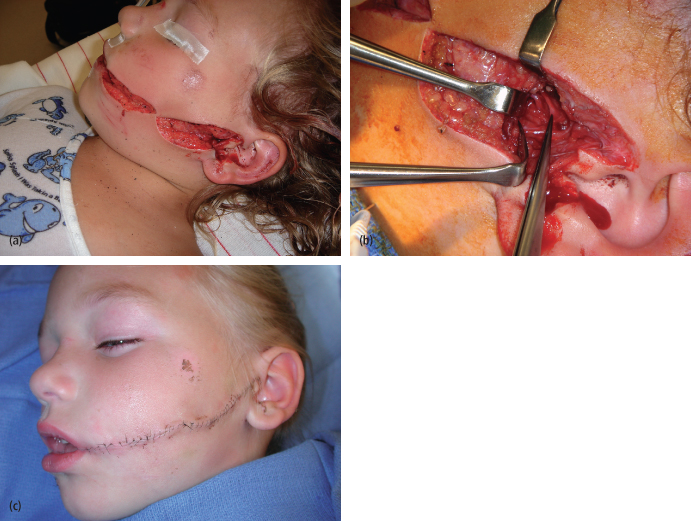
Soft tissue injuries to the maxillofacial region may be complicated by infection or functional impairment of regional anatomy. Identification of risk factors for the development of complications is of paramount importance. Primary repair is both feasible and preferred for most facial wounds, and neurovascular and ductal injuries, even in the severely contaminated wound [Figs. 4.1(a)–(c)]. The rationale for this approach is that a significant number of patients will not develop infections and thus can benefit from the aesthetic and functional advantages of primary wound healing. Secondary reconstruction may be considered for highly contaminated wounds, or those with avulsive components. Administration of tetanus prophylaxis and appropriate antibiotics for contaminated wounds is the standard of care and should follow current guidelines; however, this topic is beyond the scope of this chapter.
Fig. 4.1. Complex soft tissue injury. Primary repair is both feasible and preferred for most facial wounds, neurovascular, and ductal injuries, even in the severely contaminated wound. (a) Preoperative appearance. (b) Intraoperative view prior to anatomic layered closure. (c) Postoperative appearance.
Animal Bites
Dog and cat bites comprise an estimated 1% of emergency room visits per year in the United States.1–2 An estimated 60% of animal bites are from dogs, and 10–20% are from cats, although the incidence of dog bite injuries is declining.3 Death associated with dog bites is very rare, with only 300 dog-bite related deaths occurring from 1979–1996.4 The incidence of fracture or laceration associated with dog bite injuries is between 4% and 7%.5
Infection is the most common complication associated with dog and cat bites, occurring in approximately 20% of cases.6 The most common bacteria implicated in animal bite-related infections are Capnocytophaga, Canimorsus, and Pasteurella species. Infections from C canimorsus are aggressive in nature, and laboratory diagnosis is often difficult; therefore, antibiotic therapy should be started as early as possible when C canimorsus infection is suspected.
Canine Bites
Larger dog breeds such as Pit bull terriers, Rottweilers, and German shepherds, which lead the list of aggressive dog injuries, mostly cause high pressure crush injuries, while smaller breeds most often cause soft-tissue skin injuries.6 While adults most often sustain bites to the hand, children are most commonly bitten in the face.7,8,9 Bacteria specific to dog bite wounds include aerobes such as Pasteurella, Streptococcus, Staphylococcus, and Neisseria, and anaerobes such as Fusobacterium, Bacteroides, Porphyromonas, Prevotella, and Capnocytophaga canimorsus.8
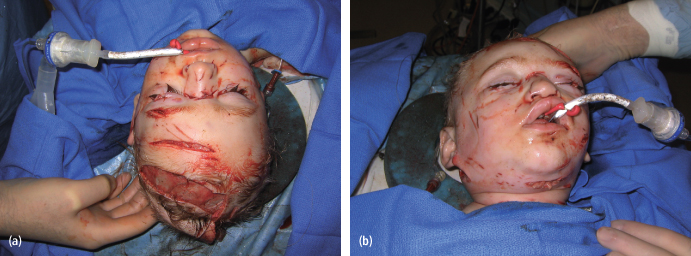
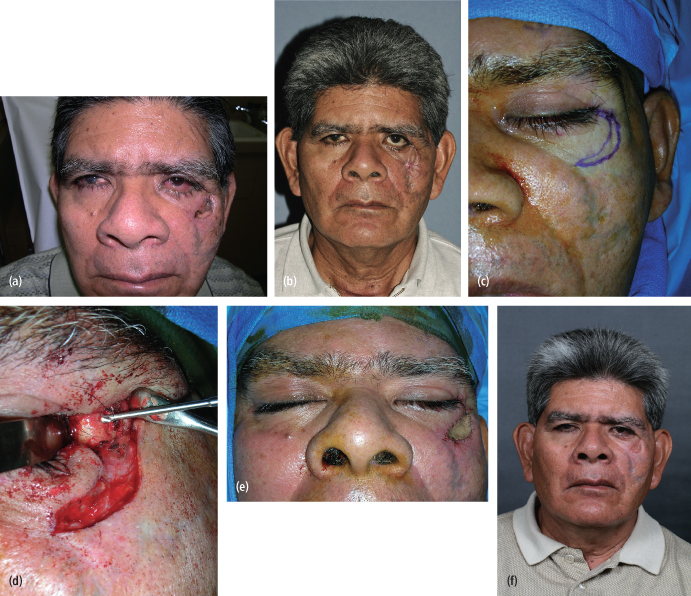
Severe “mauling” type of dog bites can cause devestating neurovascular complicaitons, resulting in cranial neuropathy or exanguinating hemmorhage [Figs. 4.2(a) and (b)]. Injuries involving the neck or parotid region in particular should undergo thorough clinical and radiographic interrogation prior to definitive repair. Primary neurorraphy, duct repair, and/or vascular repair should be considered for major neurovascular injury or disruption of the nasolacrimal or parotid duct.
Fig. 4.2. Three-year-old child involved in “mauling” injury from dog. (a) Bird’s-eye view. (b) Worm’s-eye view.
Feline Bites
Cat bite injuries are more likely than dog bites to cause injury to the face.10 In addition, due to cats’ narrow and sharp teeth, they are more likely to “inoculate” the victim by inflicting puncture wounds to the deeper soft tissue layers, which results in a higher complication rate than dog bites. The most common complications related to cat bites are localized wound infection, septic arthritis, and osteomyelitis.
Human Bites
Unlike animal wounds that become infected from the oral flora of the offending animal, infections from human bite wounds are usually secondary in origin and can occur in an estimated 10–20% of human bite wounds.11,12 Primary closure is safe and advised after proper wound cleansing.13,14
Management of Bite Injuries
Animal bites presenting for treatment more than 8 hours after injury are at high risk for the development of suppurative complications, with cat bite wounds progressing at a faster rate than dog bite wounds.8 Prophylactic antibiotics should be given in selected cases with coverage based on specific animal type. Capnocytophaga canimorsus and Pasteurella species are not susceptible to clindamycin, erythromycin, dicloxacillin, or cephalexin; therefore, these antibiotics should not be used. Amoxicillin–clavulanic acid provides excellent coverage against Pasteurella multocida, Capnocytophaga canimorsus, anaerobes, and susceptible S aureus, and should be considered first-line antibiotic therapy. Doxycycline combined with metronidazole should be considered in penicillin-allergic patients. If Methicillin-resistant Staphylococcus aureus (MRSA) is highly prevalent in the community, doxycycline can be considered as oral prophylaxis. For inpatient management, ampicillin–sulbactam, piperacillin–tazobactam, or ticarcillin–clavulanic acid should be considered first-line agents. Ceftriaxone, aztreonam, or a fluoroquinolone have good gram-negative coverage; combined with metronidazole, they are adequate alternatives. Monotherapy with a carbapenem such as ertapenem, meropenem, doripenem, or imipenem–cilastatin may also be considered. Indications for hospitalization include fever, sepsis, uncontrolled cellulitis, edema or fracture injury, loss of function, patients with immunocompromised status, or noncompliant patients.
Parotid Duct Injury
Injury to the parotid gland and duct should be suspected with any deep laceration to the cheek or neighboring structures. Van Sickels15 divided the parotid gland and duct injuries into three different anatomic regions: region A, the area of the gland; region B, the site of the duct as it runs superficial to the masseter muscle; and region C, the region of the duct from the masseter muscle to where the duct meets the oral cavity exiting the buccal mucosa adjacent to the maxillary second molar. Initial assessment should include a full cranial nerve examination. Since the buccal branch of the facial nerve travels with the parotid duct after it crosses the superficial layer of the masseter muscle after exiting the parotid gland, facial motor function should also be assessed with any parotid duct injury—specifically motor function of the upper lip. Prior to primary closure, the wound should be explored to ascertain the integrity of the parotid duct and facial nerve.
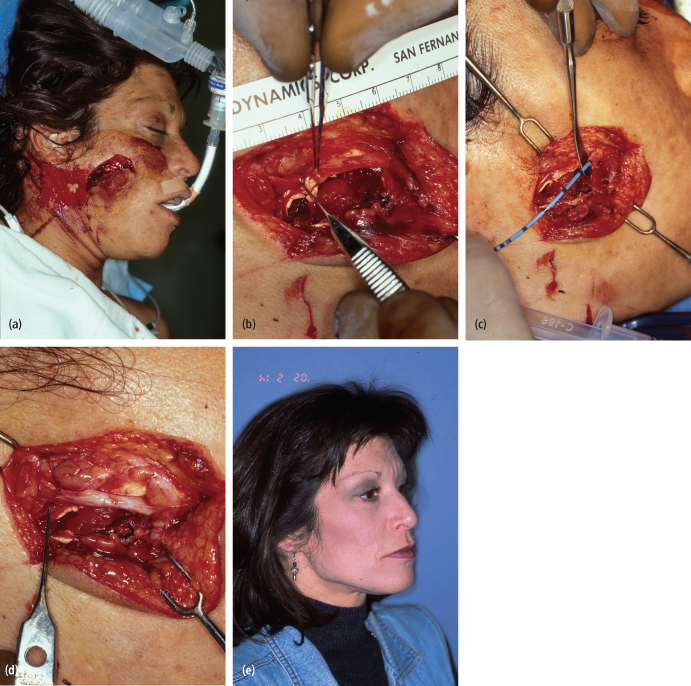
Surgical options for parotid duct repair include: (1) primary repair; (2) ligation; and (3) fistulization of the duct into the oral cavity.16 Stenting and primary repair of the duct becomes increasingly difficult with surrounding soft tissue edema and maceration. Some authors suggest leaving stents in for several weeks following duct repair,17,18 while others recommend stent removal following primary closure of the duct.16,19 Detractors of long-term stent placement cite a higher occurrence of sialocele formation. Delayed parotid duct reconstruction has been successfully reported and may be more feasible from a technical standpoint. In a study by Lewis and Knottenbelt,20 nineteen patients with parotid duct injuries underwent primary closure of the wound. Though ten patients had complications (seven with salivary fistula and four with sialoceles), all healed without surgical intervention. Primary repair whenever feasible is preferred [Figs. 4.3(a)–(e)]. If the proximal and distal ends of the parotid duct cannot be reapproximated, a pseudo-duct can be created by placing a drain with its origin at the parotid gland and terminal end in an orifice created into the buccal mucosa (fistulization).
Fig. 4.3. Forty-three-year-old woman involved in a knife assault. (a) Preoperative appearance; (b) Identification of the proximal and distal stumps of Stenson’s duct. (c) Stenting of Stenson’s duct. (d) Primary anastomosis with 7-0 nylon suture. (e) Postoperative appearance.
Sialocele Formation
Sialoceles are common sequelae following parotid duct injury and repair. Sialoceles more proximal to the gland have been shown to respond to medical treatment, including the administration of antisialogogues and antibiotics. Surgical management include aspiration and pressure dressing placement, as well as fistulization as described above. The use of botulinum toxin injection into parotid glands with fistulas has also been shown to produce resolution of sialocele.21,22 Long-lasting complications to the duct and gland that are unresponsive to previously described management may be definitely treated by parotidectomy, although the need for this is exceedingly rare.
Peripheral Facial Nerve Injury
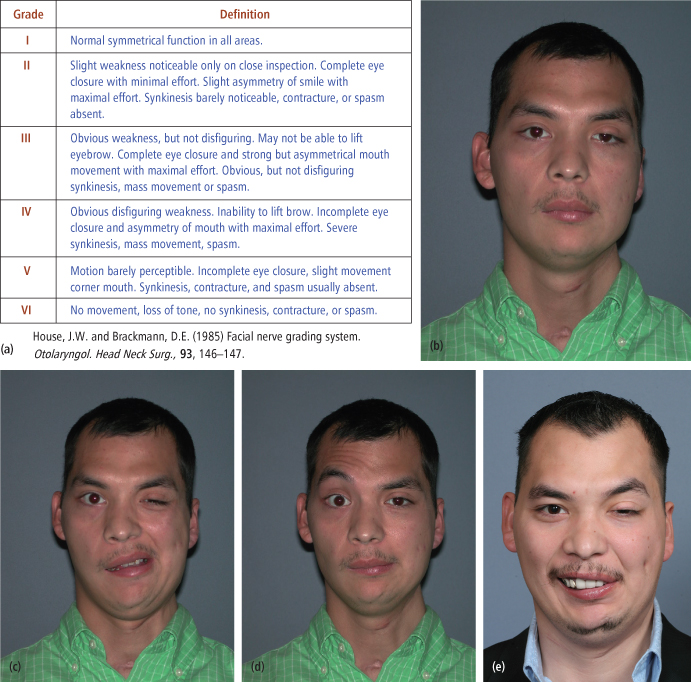
Extracranial facial nerve injury may occur with any trauma to the region between the tragus of the ear and the commissure of the lip. The House-Brackmann classification of facial function is helpful in quantifying and documenting facial nerve function after an injury [Figs. 4.4(a)–(e)]. An accurate assessment of the involved anatomy and identification of the location of an injury following cranio-maxillofacial trauma is most important for determining the most ideal therapeutic intervention.
Fig. 4.4. House-Brackmann scale of facial function. (a) Outline. (b) Facial nerve injury, repose. (c) Facial nerve injury, animation. (d) Facial nerve injury; animation. (e) Facial nerve injury, 5 years after nerve grafting, note synkinesis.
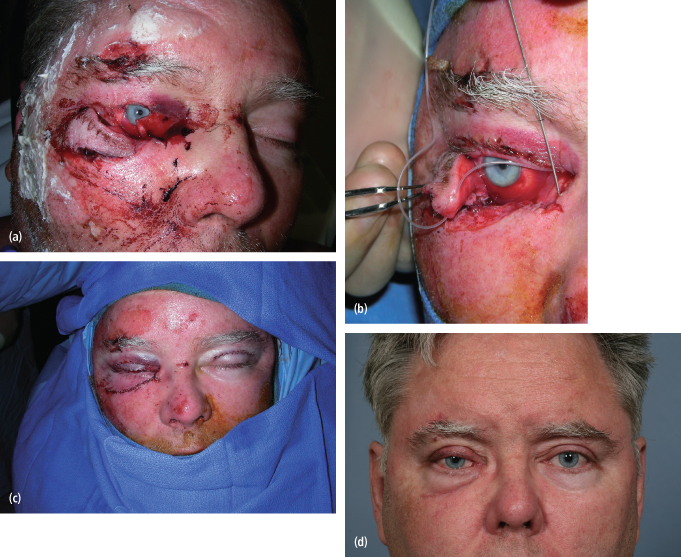
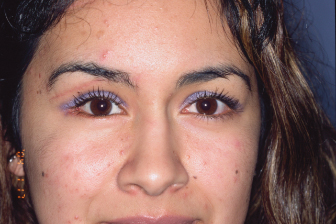
Following a complete neurological examination (if possible), the wound should be thoroughly evaluated prior to repair. Injury to the peripheral segment of the facial nerve proximal to a line drawn vertically from the lateral cantus of the eye is preferentially repaired by direct coaptation using microsurgical technique, without grafting if possible [Figs. 4.5(a)–(c)]. The goal in facial nerve repair should be restoration of facial tone, facial symmetry, and voluntary facial movement.23 If tension-free coaptation is not possible, consideration can be given to the use of an interpositional nerve graft. The great auricular nerve is usually available and can be harvested without a second incision. Alternatively, a sural nerve graft may be harvested for this purpose. Another option is the hypoglossal-facial nerve (XII to VII) neurorrhaphy with or without grafting, although this technique may be preferred in the secondary setting due to the donor site morbidity.24,25 Lagophthalmos should be managed with a gold weight relatively soon after injury and neurorrhaphy.
Fig. 4.5. Five-year-old girl with a deep laceration of the cheek, proximal to the lateral canthus of the eye. (a) Preoperative appearance. (b) Intraoperative exploration of facial nerve. (c) Wound closure.
Nasolacrimal Injury
Injury to the nasolacrimal apparatus commonly occurs following injury to the eyelids and is associated with nasoethmoidal fractures approximately 20% of the time. The presenting sign is typically epiphora. Primary disruption of the nasolacrimal apparatus must be distinguished from eyelid malposition such as ectropion, in which the punctum is not opposed to the globe, both of which result in epiphora. Additionally, facial nerve paresis may be associated with epiphora secondary to paralytic ectropion or weakness of the orbicularis muscle that is required for normal lacrimal pump function. Chronic inflammation and obstruction of the puncta may be complicated by dacryocystitis, which produces a red, swollen, and painful mass in the medial canthus, requiring prompt attendtion.26
The Schirmer secretion test can be used to assess and quantify tear production. After topical anesthetic is administered, white filter paper strips are placed at the junction of the middle and lateral thirds of the lower eyelids and held there with the patient’s eyes closed for 5 minutes. The test is normal with greater than 10 mm but less than 30 mm of tear production. A fluorescein dye disappearance test may also be used and is performed by administering topical anesthetic and placing fluorescein dye at the inferior fornix of both eyes. Tear films are compared over a period of 5 to 10 minutes. The dye should drain rapidly through a patent outflow system. Dye in the tear film after 10 minutes indicates an abnormality of lacrimal outflow. Punctal dilation and canalicular probing, as well as lacrimal irrigation, Jones tests, dacryocystography, and dacryoscintigraphy are also used to evaluate lacrimal function.
In the primary setting, disruption of the nasolacrimal duct may be managed by cannulation anastomosis and intubation of the lacrimal duct [Figs. 4.6 (a)–(d)].27 Chronic lacrimal duct obstruction is most successfully managed with dacryocystorhinostomy,28 which today is performed endonasally, with an operating microscope to provide increased visualization.
Fig. 4.6. Fifty-eight-year-old with complex through-and-through eyelid injury. (a) Preoperative appearance (b) Cannulation of the lacrimal duct. (c) Eyelid repair with canthopexy and upper eyelid skin graft. (d) Postoperative appearance.
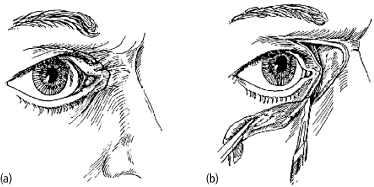
Ectropion
Several types of eyelid ectropion exist, including congenital, senile, cicatricial, and neurogenic. Cicatricial ectropion is most common in the post-traumatic setting. Scarring involving the midface may cause contracture of the lower eyelid and the resultant scleral show. Alternatively, the complication may be iatrogenic, resulting from improper lid position following lower lid approaches to the orbit. Ectropion may involve the upper lid, lower lid, or both, and result in epiphora and/or other ocular complications. The scar contracture may be medial or lateral, and it may include vertical shortening, horizontal shortening, or both.
Medial Ectropion
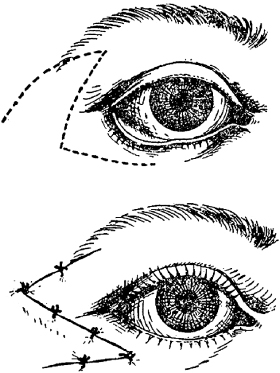
Medial ectropion may result from burns or tissue loss on the nasal dorsal skin, which causes scar contracture of the medial canthus in a medial and forward vector. This is often manifested as epicanthal folding, epiphora, and corneal exposure. The general principle of treatment is to restore the original size and location of distorted tissues, and to replace missing tissues with those of the similar structure. Scar release for medial ectropion involves a C-shaped incision over the upper eyelid, lateral nose, and lower eyelid (Fig. 4.7).29 The resulting defect requires coverage with a full thickness skin graft to minimize secondary contraction. To optimize results and limit recurrence, the skin over the nose beyond the release incision is undermined to allow maximal release, and the periosteum over the frontal process of the maxilla is excised to allow adherence of the graft to the underlining periosteum. Adjunctive medial canthoplasty may be required and can be performed.30
Fig. 4.7. Technique for medial ectropion repair. (a) “C-shaped” incision. (b) Tissue advancement with skin grafting the defect.
Lateral Ectropion
Lateral ectropion is a relatively common iatrogenic complication related to lower eyelid approaches to the orbit (Fig. 4.8). While ectropion is inevitable in maxillofacial trauma surgery, avoiding the traditional “subciliary” approach, in favor of the transconjunctival and midlid incisions, can minimize its incidence.
Fig. 4.8. Lower eyelid ectropion with increased scleral show.

The initial description of the Z-plasty by Denonvilliers was employed for treatment of lateral ectropion of the lower eyelid (Fig. 4.9).31 The Z-plasty has the advantage of transposing the retracted lower eyelid tissue superiorly and posteriorly, achieving better adaptation of the lid margin to the globe. However, the additional scar that results from a Z-plasty in this area may be unsightly.32
Fig. 4.9. Denonvilliers Z-plasty for lateral lower eyelid ectropion.
Cicatricial ectropion of either eyelid with vertical shortening is approached with a subciliary release, or alternatively via a subconjuntival approach. The incision is made short of the medial and lateral canthi. Skin flaps are raised to allow approximation of the upper and lower eyelid margins. A bone-anchored lateral canthopexy is performed followed by skin graft if necessary. The eyelids can be separated either immediately, or in delayed fashion, if recurrence is of concern. If necessary, the C-shaped release can be combined with this approach, but should be completed first [Figs. 4.10(a)–(f)].30 In addition to release and grafting, various local flaps have been described and can be employed for repair of lower eyelid cicatricial ectropion.32,33
Fig. 4.10. Lateral ectropion. (a) Exposure keratopathy with secondary conjunctivitis. (b) Preoperative appearance demonstrating lateral ectropion secondary to scar contracture. (c) “C-shaped” incision outline. (d) Bone anchored lateral canthopexy. (e) Full thickness skin graft of the defect with lower lid repositioning and lateral canthopexy. (f) Postoperative appearance.
Entropion
Post-traumatic entropion is usually seen as complication of lower eyelid approaches which turn the eyelid and lashes inward toward the globe and is associated with increased scleral show and eye injury (Fig. 4.11).34 Complications of entropion include ocular discomfort, trichiasis, corneal abrasion, microbial keratitis, corneal vascularization, and visual loss.35 While senile entropion is caused by different pathological mechanisms such as loss of lid laxity, loss of tension of lower lid retractors, and alterations to the musculus orbicularis, post-traumatic entropion is virtually always cicatricial in nature, and typically associated with transconjunctival approaches to the orbit. Cicatricial entropion can be treated by a keratinized tissue graft from the hard palate.36 Management may also include anchoring the lateral tarsal tip to the orbital rim and using a single stitch lateral wedge technique. Description of these techniques is outside the scope of this chapter, but abundance of texts and articles are available for further reading.37
Fig. 4.11. Right lower lid entropion following transconjunctival approach to the orbital floor with lateral canthotomy.
HARD TISSUE INJURIES
Mandibular Fractures (Symphysis, Body, Angle)
The primary goals of treating mandibular fractures are: (1) to restore form and function by returning the patient to the pre-injury occlusion and achieving anatomic reduction when possible and (2) to achieve osseous union predictably. The appropriate technique to achieve these goals varies based upon location of the fracture, the energy of the injury causing the fracture, and whether or not there is load sharing potential. The most common complications of mandibular fractures include infection, malunion, nonunion, tooth injury, and the need for hardware removal. Adherence to proper surgical techniques and the principles of reduction, stabilization and fixation with appropriate means will assist in minimizing these complications.
Principles of Fixation
A fixation system will provide either absolute (rigid) stability or functional stability. Rigid stability occurs when no movement whatsoever occurs across the fracture gap; functional stability occurs when movement is possible across the fracture gap but is balanced by external forces and remains within the limits that allow for the fracture to progress to union. While excessive mobility at a fracture site will lead to bone resorption and fibrous tissue ingrowth, absolute rigidity also will not achieve bony union. When excessive mobility is present, any fixation device will promote bone resorption and infection.
Functionally stable fixation is all that is necessary for successful healing of most fractures in the maxillofacial skeleton. Micromotion occurs in this paradigm that permits for secondary bone healing to occur. An example of nonrigid fixation is the use of a single miniplate at the angle of the mandible as described by Champy.38 Thus, functionally stable fixation may also result in osseous healing and achieve predictable results.
Fixation requirements are considered by the ability of the host bone to share some of the functional loads. Load-bearing fixation is of sufficient strength to resist the functional masticatory forces during the healing phase, and the host bone fracture sites share none or little of the functional load. In contrast, load-sharing fixation refers to a scheme whereby the functional load is shared between the hardware and the bone along the fracture site. The indications for providing load-bearing fixation are those fractures with comminuted segments, atrophic mandibular fractures, and fractures with avulsed or missing segments. Load-sharing fixation is indicated in cases where no comminution or bone defects are present, and when intact bone cortices are opposed to one another after fracture reduction. The majority of mandibular fractures can be adequately treated with load-sharing fixation.
Teeth in the Line of Fracture
The management of teeth in the line of fracture has been a source of controversy in the literature for decades.39 While it is clear that retaining grossly mobile or infected teeth in the line of fracture may invite wound healing complications, it is not clear how teeth in the line of fracture in all patients should be managed.
Various authors have attempted to use specific criteria such as tooth mobility, interference with fracture reduction, pulpal pathology, and location of the fracture to determine whether or not the tooth should be removed.39–46 The preponderance of evidence suggests that teeth in the line of fracture (including third molars) may be retained providing that they do not interfere with favorable reduction, stabilization, and fixation of the fracture, and are not grossly mobile or infected. This approach has yielded a complication rate similar to other reported complication rates, regardless of the presence or absence of teeth in the line of fracture.47
Infections
Infection is the most common complication in patients undergoing treatment of mandibular fractures, occurring between 1% and 32% of the time.48–58 Numerous risk factors have been associated with postoperative infection, including substance abuse or patient noncompliance with postoperative care regimens,48 as well as significant delay in treatment.59 Antibiotics have been shown to be effective in preventing infection when instituted before the repair of mandibular fractures.60,61 However, the same effect for postoperative antibiotic administration has not been demonstrated.62,63 While some investigators have concluded that the timing of treatment of mandible fractures following injury does not seem to have any prognostic value in success with infection rates relatively equal in early and late repairs,64–67 others have shown that treatment within 3 to 5 days after trauma is optimal in terms of minimizing the rate of infection.68,69 Other risk factors include high-velocity injuries and severe comminution or gross contamination.
Most infections related to mandibular fractures are polymicrobial, with both aerobes and anaerobes routinely cultured. The most common organisms are Staphylococcus, alpha-hemolytic Streptococcus, and Bacteriodes, as well as gram-negative organisms. Penicillin G, (with or without Flagyl, depending on the gram stain) or clindamycin are the drugs of choice.
Successful management of the infection requires adequate drainage, removal of the source, and appropriate antibiotic coverage. If the cause of the infection is related to mobile hardware and there is favorable bony union, removal of the loose hardware may be all that is necessary. Alternatively, if inadequate stability of the fractured segments is apparent, the previously placed fixation should be removed and replaced, usually with more rigid fixation. With careful patient selection, immediate bone grafting of infected mandibular fractures can be employed. This approach, in conjunction with appropriate fixation and intraoperative debridement, can result in bony union.
Malunion/Malocclusion
Malunion occurs when the fractured segments achieve osseous union in a position that results in either dysfunction or deformation. Complex or high velocity injuries increase the risk for malunion, as does patient noncompliance with postoperative instructions. Unfavorable functional or esthetic outcomes can also occur when the surgeon violates the basic principles of reduction, stabilization, and fixation.
Mandibular Symphysis, Parasymphysis, and Body Fractures
Malocclusion following treatment of mandibular fractures that involve the dentate portions of the mandible typically occurs when the fractured segments are not adequately reduced and stabilized, and appropriate fixation is not applied [Figs. 4.12(a)–(c)]. Evaluation of the occlusion for the presence of crossbite, accurate positioning of occlusal wear facets, as well as evaluation for mandibular angle flaring are important prior to fixation. Preexisting anterior open bite malocclusion can be challenging to assess, but careful examination for the lack of wear facets and the presence of mamelons on the incisor teeth should be of assistance.
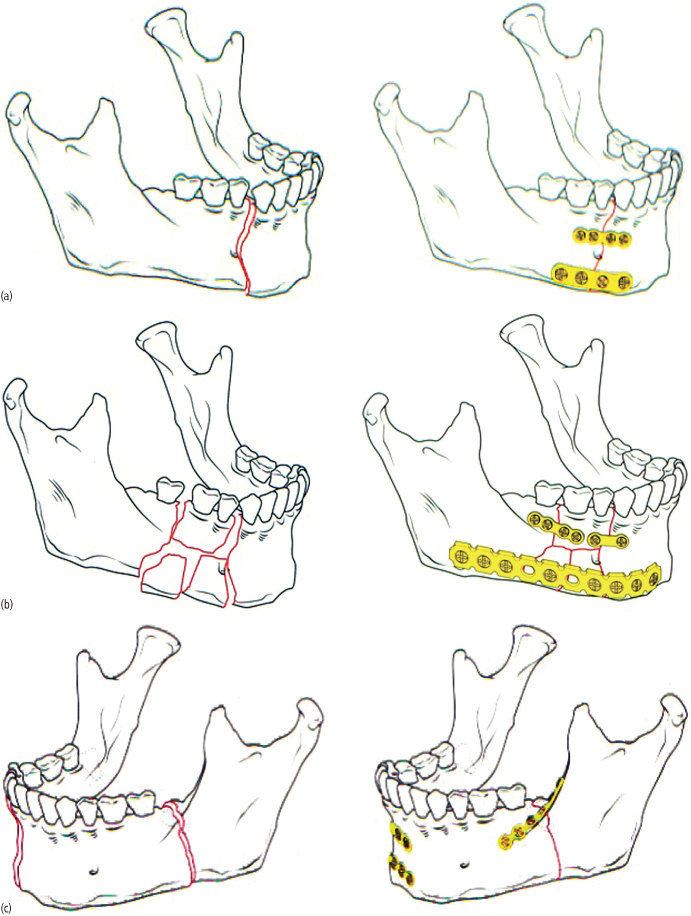
Fig. 4.12. Principles of rigid internal fixation of the mandible. (a) Load-sharing fixation, with linear fracture of the right parasymphysis (left); stability is provided by a fixation system in conjunction with stabilizing forces provided by anatomic abutment of noncomminuted fracture segments (right). Fixation is applied to the inferior border (zone of compression) and superior border (zone of tension). (b) Load-bearing fixation, with comminuted fracture of the right mandibular body (left); functional stability is provided solely by the fixation system (right). Contemporary fixation systems involve 2.3- or 2.4-mm locking reconstruction plate at inferior border combined with secondary fixation of comminuted segments at the superior border. (c) Functional stabilization along Champy’s ideal lines of osteosynthesis. Fractures located proximal to the first premolar may be safely stabilized with a single miniplate placed in the midbody position (2.0 mm). Fractures anterior to the first premolar should be stabilized with two plates (a tension band and compression band) separated by 4–5 mm and generally placed on either side of the mental nerve.
(All images: Prein J, ed. 1997. Manual of Internal Fixation in the Craniofacial Skeleton: Techniques Recommended by the AO/ASIF Maxillofacial Group. Springer.)
Following reduction, stabilization, and fixation of the fracture, the patient is assessed for the presence of a malocclusion. If one exists, then the fixation is removed and the procedures repeated. If malocclusion is recognized in the first or second postoperative week, it is advisable to take the patient back to the operating room for revision. If the malocclusion is recognized later than 2 to 3 weeks postoperatively, it is advisable to allow for bone union to occur and then consider secondary treatment if necessary.
Secondary correction of a mild occlusal discrepancies occurring from treatment of fractures of the dentate portions of the mandible should include consideration for comprehensive orthodontics to level and align the dentition. Orthodontic treatment alone in certain cases may be all that is necessary to correct spacing or step problems. If the occlusion cannot be corrected with orthodontic means alone, or if there is a significant aesthetic concern, then mandibular osteotomies are generally necessary.
Mandibular Angle Fractures
The ideal treatment of mandibular angle fractures has been controversial. Fractures of the mandibular angle, often complicated by the presence of a third molar, have been shown to have the highest rate of postoperative complications of all mandibular fractures.45,70,71,72 Numerous techniques have been utilized to treat mandibular angle fractures.
Complication rates for the treatment of mandibular angle fractures of 0% to 32% have been described, depending upon the technique utilized.49–58 Complication rates are often difficult to interpret, as the definition of a complication is variable. Bell and Wilson47 described a complication rate of 32% in a series of 162 angle fracture patients. However, this number is misleading because virtually all patients had a favorable outcome with successful bony union and a return to premorbid occlusion. All but two patients had their complications managed on an outpatient basis, under local anesthesia or IV sedation, and almost always after bony union had been achieved. The majority of complications consisted of hardware removal, which is a limitation inherent to some fixation techniques employed for treatment.
Malocclusion resulting from angle fractures typically results in an ipsilateral posterior open bite in the case of unilateral fracture, or in an anterior open bite in the case of a bilateral angle fractures. In contrast to the malocclusions associated with the tooth-bearing segments of the mandible, which can often be treated with orthodontics alone, the open bite associated with angle fractures typically requires osteotomies for correction [Figs. 4.13(a)–(b)]. Typically, a unilateral or bilateral sagittal split osteotomy or combination with vertical ramus osteotomy are required for correction. Concurrent orthodontic treatment, although recommended, may not be essential for excellent treatment outcomes.
Fig. 4.13. Malunion of untreated bilateral mandibular angle fractures. (a) Lateral view (note facial elongation). (b) Occlusion (note anterior open bite).
Nonunion
Nonunion of mandibular fractures is an uncommon sequela of treatment, but may occur even in the most experienced hands. High velocity injuries resulting in severe comminution, inadequate or improperly placed fixation, and poor patient compliance are common etiologies [Figs. 4.14(a)–(d)].
Fig. 4.14. Inadequate fixation resulting in infection and nonunion. (a) Nonbuttressed fractures, such as this comminuted fracture of the mandibular body are at high risk for healing complications. (b) Similarly unbuttressed fracture treated with inadequate (2.0 mm plates) and improper (DCP) fixation. (c) Postoperative panoramic radiograph of patient in (b) following repair with load-bearing 2.4-mm locking reconstruction plate. (d) Radiographic evidence of long-term union.
Comminuted fractures require load-bearing fixation because the surrounding bony fragments are incapable of sharing any of the functional loads transmitted during healing. Once the major proximal and distal segments are adequately stabilized, the remaining segments may be further secured. In cases of severe comminution, it is often helpful to stabilize the smaller fractures with miniplates first and then apply rigid fixation to the remaining construct in order to provide load-bearing support. Reoperation for debridement of necrotic soft tissue and nonviable bone and further stabilization potentially with grafting may be required in these cases.
Atrophic Edentulous Mandibular Fractures
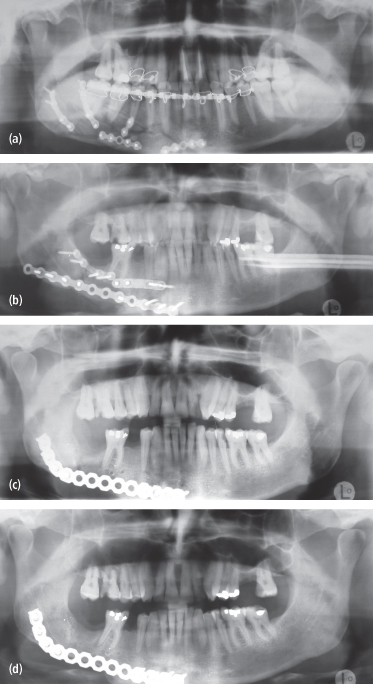
Fractures of the atrophic edentulous mandible have traditionally been a major risk factor for nonunion. The attached muscular forces often cause significant displacement, patients are unable to wear dentures, and associated pain and masticatory dysfunction quickly renders these patients oral cripples. Furthermore, fracture-healing potential is impaired because there is little endosteal or periosteal blood supply and there is a severely diminished surface area that allows no load sharing capacity. Because of this, load-bearing fixation is necessary to achieve bony union. The use of miniplates is contraindicated due to high rates of nonunion [Figs. 4.15(a)–(c)].
Fig. 4.15. (a) Panoramic radiograph demonstrating a failed attempt at closed reduction of an atrophic edentulous mandible fracture with Gunnings splints and skeletal fixation with circummandibular and circumzygomatic wiring techniques. (b) Diagram demonstrating the appropriate use of load-bearing fixation with a reconstruction plate to provide adequate stability in the case of an atrophic edentulous mandible fracture. (c) Panoramic radiograph showing adequate reduction and stabilization of an atrophic edentulous mandible fracture with a load-bearing reconstruction plate.
Although controversy still exists with regard to the efficacy or necessity of simultaneous bone grafting in the primary setting,73,74 there is little disagreement over the need for bone grafting in secondary reconstruction of nonunited mandibular fractures in general or in management of nonunion of a severely atrophic mandible fractures in particular. The theoretical advantage of primary bone grafting is that a corticocancellous bone graft would augment the osteogenic potential of the bone at the fracture gap and enhance healing.75
Mandibular Widening
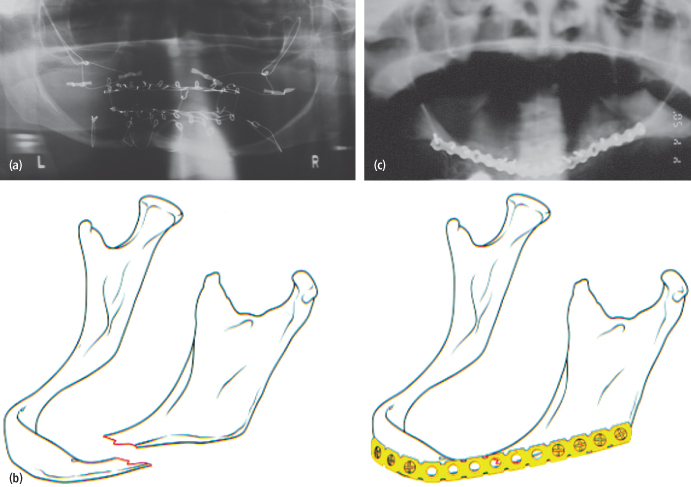
Careful attention should be paid to cases of bilateral mandibular fractures involving the symphysis or parasymphysis. Lateral muscular forces will cause widening of the mandible by splaying the angles outward and creating a defect on the lingual aspect of the anterior-most fracture. Failure to adequately account for the lateral muscular forces will result in a gap along the lingual cortex and facial/mandibular widening [Figs. 4.16(a), (b)]. Treatment, if necessary, involves “re-osteotomizing” the symphysis fracture, over-reducing the fractures by pressing in at the mandibular angles, and applying load-bearing fixation to stabilize the symphysis.
Fig. 4.16. Bilateral mandibular fractures with facial widening. (a) Preoperative axial CT image demonstrating inadequate reduction and fixation of the symphysis fracture (note diastasis at the lingual border of mandible). (b) Illustration of planned surgical treatment to include load-bearing fixation placed at the symphysis and open reduction with internal fixation (ORIF) of the mandibular condyles bilaterally (overbent plate).
(All images: Prein J, ed. 1997. Manual of Internal Fixation in the Craniofacial Skeleton: Techniques Recommended by the AO/ASIF Maxillofacial Group. Springer).
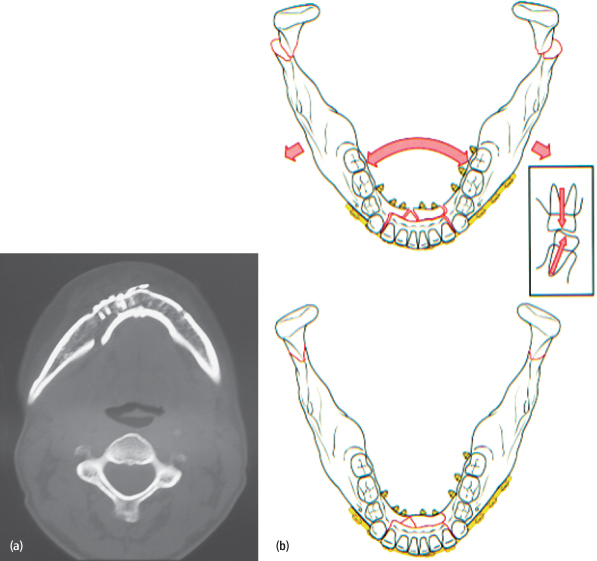
Facial Asymmetry
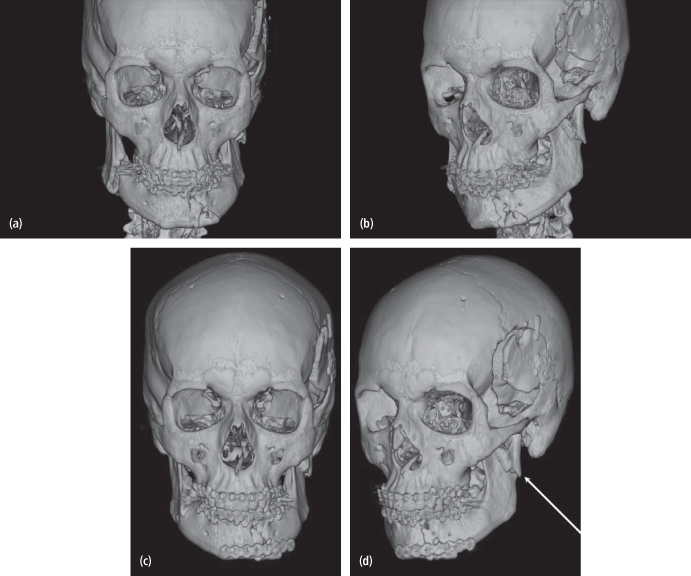
Cillo and Ellis76 recognized the importance of managing so-called “double unilateral” fractures of the mandible to prevent facial asymmetry [Figs. 4.17(a)–(d)]. In a review of 1,287 patients with mandibular fractures, they found that approximately 2.5% of the patients had more than one fracture on the same side, and in 25% of this subgroup, complications involving gonial flaring and cross bite occurred.
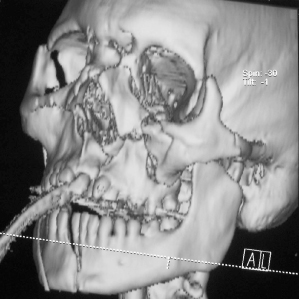
Fig. 4.17. Facial asymmetry related to undertreated “double unilateral” mandibular fractures. (a) Preoperative 3D CT image of patient with fractures of the right angle, left parasymphysis (comminuted), and left condyle. (b) Oblique view. (c) Postoperative CT image of patient following ORIF of the right angle fracture and left parasymphysis with closed treatment of the left subcondylar fracture (note: medial rotation and shortening of the left ramus with flaring of the left angle). (d) Oblique view (note: shortening of the left condyle ramus unit and medial displacement of the ramus/body segment. Ideally, double unilateral fractures should have both ipsilateral fractures reduced and stabilized.
In order to prevent facial asymmetry, absolute anatomic reduction of all fractures must be achieved and adequate fixation should be applied.
Mandibular Condyle Fractures
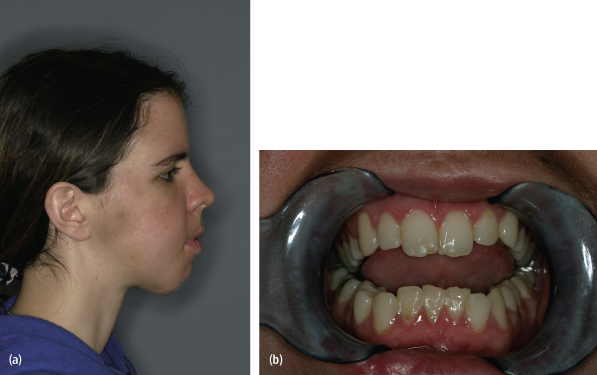
Complications of trauma to the mandibular condyle can occur and include disturbance of occlusal function, facial asymmetry, ankylosis of the temporomandibular joint, limited mouth opening, and degenerative joint disease. The goals of treatment include prevention of functional limitations and establishment of a pain-free maximal incisal opening to preoperative levels; symmetrical pain-free movement of the jaw in all excursions; preinjury occlusion; and good facial symmetry. The methods used to achieve these goals are several and controversial. Currently these methods include the use of maxillomandibular fixation for a finite period of time followed by functional therapy; immediate functional therapy without the use of maxillomandibular fixation; and open reduction and internal fixation. The optimal treatment approach has not been universally agreed upon and indeed is probably one of the most controversial topics in the of maxillofacial surgery field.
Favorable results have been reported by a number of authors in large series of patients utilizing only closed treatment.77–83 From these data it is clear that the goals of treatment for the great majority of patients with mandibular condyle fractures can be met utilizing closed methods. The question is “Which fractures, if treated closed, will most likely result in complications?”
The existing literature suggests that there are a number of potential predictors of complications following closed treatment. These include significant loss of ramus height, comminuted intracapsular fractures,84 increased age,85 bilateral subcondylar fractures, double unilateral mandibular fractures,76 and condyle fractures associated with midface or panfacial fractures. In each of these instances, consideration is given to the risks, benefits and options of open treatment.
Malunion/Malocclusion
Malunion or malocclusion following closed or open treatment of mandibular condyle fractures is typically the result of loss of vertical dimension of the ramus/condyle unit [Figs. 4.18(a)–(g)]. Most of the time, the consequence of this loss in height is deviation of the mandible to the affected side, although occlusal prematurities and centric relation/centric occlusion discrepancies are also relatively common. In some cases of unilateral fracture, a malocclusion will manifest itself as an ipsilateral open bite. An anterior open bite may result in cases of bilateral condylar or subcondylar fractures.
Fig. 4.18. Fifteen-year-old male with bilateral mandibular condyle fractures, treated closed, and complicated by anterior open bite. (a) Pretreatment appearance. (b) Pretreatment occlusion. (c) Post closed-treatment panoramic radiograph. (d) Post closed-treatment occlusion. (e) Panoramic radiograph 6 weeks following bilateral sagittal split osteotomies (BSSO) for correction of occlusion. (f) Facial appearance 6 weeks following BSSO. (g) Occlusion 6 weeks following BSSOs.
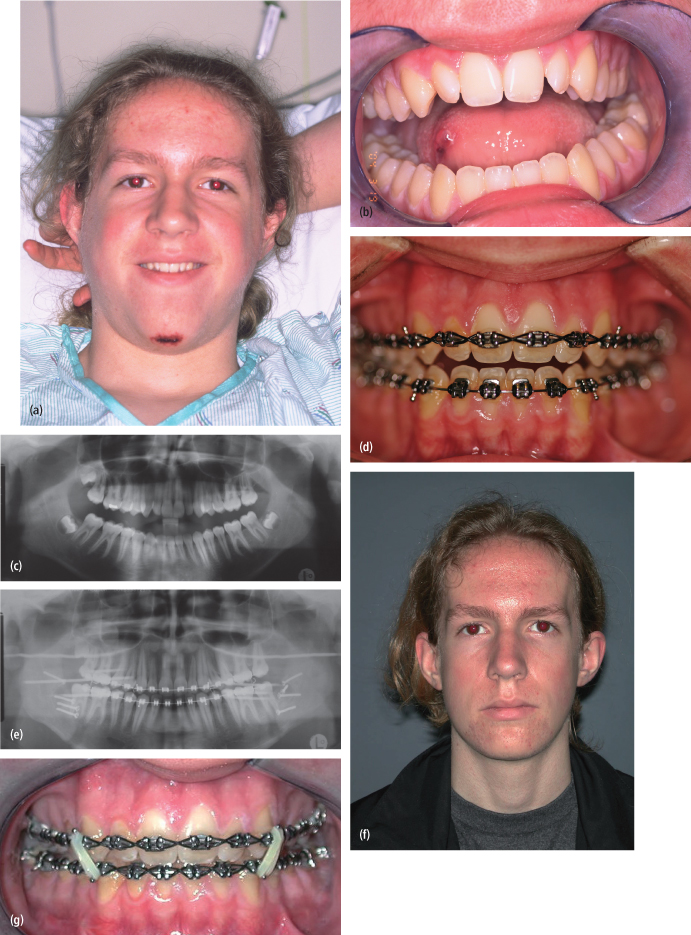
Treatment of significant malocclusions ideally involves a combination of orthodontics and surgery to reposition the tooth-bearing segment by surgically rotating the mandible in a counterclockwise fashion. The choice of technique is dependent upon the level of the original fracture. For fractures located within the mandibular condyle, intraoral sagittal split osteotomy is the preferred method of mandibular repositioning. If the original fractures were in the subcondylar or ramus regions, then the “inverted L” osteotomy may be required, as it allows for greater lengthening of the pterygomasseteric sling.
Facial Asymmetry
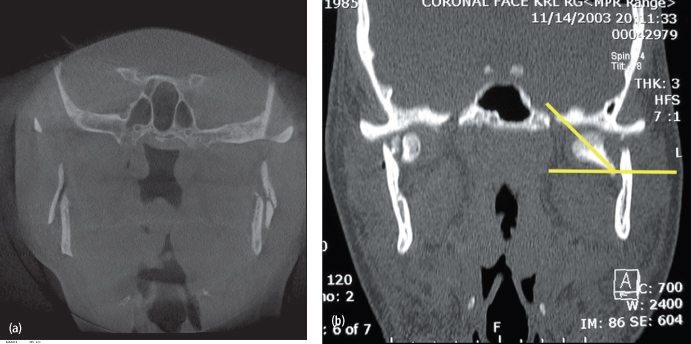
Ellis and Throckmorton86 in 2000 emphasized the differences in facial symmetry related to open and closed treatment of fractures of the mandibular condylar process. Patients with overriding fractures or significant angulation who were managed with closed treatment developed facial asymmetry by virtue of the shortening of the condyle ramus unit on the affected side.87 While they did not provide objective guidelines for open reduction, a study by Kleinheinz and colleagues87 found that displacements less than 37 degrees from the sagittal axis of the ascending ramus had negligible loss of vertical height when treated by conservative techniques [Figs. 4.19(a), (b)]. Therefore, to prevent complications associated with closed treatment, their recommendations for open repair of condyle fractures included displacement in excess of 37 degrees and significant decrease in vertical ramus height.87
Fig. 4.19. Risk factors for unfavorable results for the management of mandibular condyle fractures with closed treatment. (a) Overlapping left mandibular condyle fracture with loss of vertical dimension. (b) Angulations of less than 37–45 degrees will result in negligible loss of vertical height when treated by closed techniques.
Mandibular Hypomobility/Temporomandibular Joint Ankylosis
The etiology of post-traumatic mandibular hypomobility is unclear. One proposed mechanism is related to temporomandibular joint trauma, which causes hemarthrosis and initiation of a cascade of events that includes altered disc mechanics, cartilage degeneration, and the release of inflammatory mediators. These mediators of inflammation in turn cause effusion, internal derangement, fibrous adhesions, and hypomobility. Prevention of hypomobility following trauma to the mandibular condyle is of paramount importance.
The management of nonankylotic temporomandibular joint (TMJ) hypomobility, once it occurs, is beyond the scope of this chapter. In general, nonsurgical management, with nonsteroidal anti-inflammatory medications, soft diet, heat, and physical therapy with aggressive range of motion exercises is the best approach.
TMJ ankylosis is a severe complication that may arise following mandibular condyle fracture. The etiology of post-traumatic TMJ ankylosis is unclear but may be related to the fracture pattern. He et al. examined a series of patients with TMJ ankylosis secondary to condylar fractures and observed that a significant percentage of their patient population had sustained intracapsular fractures in addition to a second concomitant fracture. The authors postulated that the combination of sagittal intracapsular fractures, an associated fracture of the body or symphysis, and inadequate reduction of the fractures, led to a widening of the mandible and displacement of the lateral pole of the condyle or condylar stump laterally or superolaterally in relation to the zygomatic arch, where it fuses (Fig. 4.20). The authors concluded that anatomic reduction of the fractures in this population of patients may minimize the incidence of post-traumatic TMJ ankylosis.
Fig. 4.20. The postulated theory behind TMJ ankylosis and facial fractures involves those with a combination of sagittal intracapsular fractures, an associated fracture of the body or symphysis, and inadequate reduction of the fractures, which leads to widening of the mandible and displacement of the lateral pole of the condyle or condylar stump laterally or superolaterally in relation to the zygomatic arch, where it fuses.
Bony ankylosis should be managed with gap arthroplasty and TMJ reconstruction. Numerous autogenous options are available for the purpose of reconstructing the mandibular condyle, and they have a long history of success. Custom alloplastic total joint prostheses for reconstruction of the condyle and temporal fossa following gap arthroplasty for bony ankylosis is an excellent, and perhaps preferred, alternative.
Facial Nerve Injury
The facial nerve may be encountered in open approaches to the condylar neck and head. The marginal mandibular branch that innervates the depressor anguli oris, depressor labii inferioris, and lower fibers of the orbicularis oris and mentalis may be encountered during standard submandibular or Risdon, low cervical, retromandibular, and preauricular approaches. In standard approaches, when posterior to the facial artery, the marginal mandibular branch of the facial nerve will be roughly 1 cm below the inferior border of the mandible, whereas when crossing the facial artery, the marginal mandibular branch is usually above the inferior border of the mandible.88 However, some patients may show two, three, or even four branches of the marginal mandibular branch of the facial nerve between the angle of the mandible and the facial artery and vein. The facial vessels lie deep to the facial nerve; therefore, risk to the facial nerve is low once the plane of facial vessels is reached. It has been advocated to dissect to the level of the capsule of the submandibular gland to protect the marginal mandibular branch.89 Using this approach, the capsule of the submandibular gland is included in the flap that is elevated toward the mandibular border and contains the marginal mandibular branch.
Alternatively, a transmasseteric approach can be performed where an incision is made though the masseter, 10–20 mm above the mandibular basilar edge.90 In addition, endoscopically assisted fixation of condylar neck fractures, though time consuming, is associated with less morbidity to the facial nerve.91
Trigeminal Nerve Injuries
Injuries to the all three branches of the trigeminal nerve can occur following maxillofacial trauma.92 The prevalence of inferior nerve injury following mandibular trauma approximates 58.5%. Causes of nerve injuries include soft tissue edema, secondary ischemia, transection and crush injuries of the nerve, and when the line of fracture occurs at a foramen and bony fragments impinge on the nerve.92 The latter cause can lead to permanent anesthesia, parathesia, and dysesthesia if not addressed in a timely manner. Additionally, disruption of the inferior alveolar canal may cause bony proliferation and stenosis of the canal.93
Bagheri and colleagues described an algorithm for the approach to the patient with trauma-related trigeminal nerve injury.92 In the preoperative period, in patients with neurosensory dysfunction, exploration and repair of the nerve should be carried out. However, if microsurgical repair is not possible, open reduction of the fracture should be performed and neurosensory testing should be conducted for 3 months. Patients with persistent neurosensory dysfunction after 3 months should be referred to a microneurosurgeon. Nerve exploration and repair should tak/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


