Grafts Frequently Used During Orthognathic Surgery and for Adjunctive Procedures
Bone Grafting: Definitions and Options
During maxillofacial surgery, grafting is a procedure that replaces missing structures with tissues from the patient’s own body or with artificial, synthetic, or natural substitutes.24,33,40,50,56,60,62,69–71,78,106,111,112 It is estimated that more than 500,000 bone-grafting procedures are performed annually in the United States, and these numbers easily double globally. Approximately half of the procedures that involve grafts relate to spinal fusion. The exact number of grafts used in head and neck reconstruction is not known, but it is significant.
Bone grafting is possible because bone—unlike most other biologic tissues—has the ability to regenerate completely if it is provided with space into which it can grow.3–5,7,13,29,34,49,64,65,83,125–128 As native bone grows, it will generally replace any resorbable graft material in its path, which results in a fully integrated region of new bone. The biologic mechanisms that provide a rationale for bone grafting are osteoconduction, osteoinduction, and osteogenesis.
Osteoconduction occurs when the graft material serves as a scaffold that facilitates new growth from the native bone (Fig. 18-1, A). Osteoblasts from the margin of the defect use the graft material as a framework upon which to spread and generate new bone. The selected graft material for the management of a bone defect should at the very least be osteoconductive.
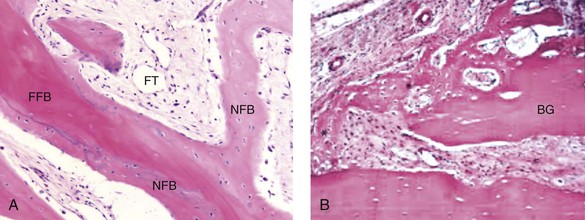
Figure 18-1 A, Osteoconduction. A photomicrograph of a bone biopsy, where NFB surrounds the pre-existing bone and the residual of FFB. NFB presents features of mature bone, with well-organized lamellae and numerous small osteocytic lacunae. B, Osteoinduction. Peripheral region of the graft at 10 days. New bone formation, with primary bone tissue from the bone graft (BG) fragment and the parietal bone. A from Acocella A, Bertolai R, Nissan J, Sacco R: Clinical, histological and histomorphometrical study of maxillary sinus augmentation using cortico-cancellous fresh frozen chips. J Craniomaxillofac Surg 39:192–199, 2011. B from Esteves JC, Borrasca AG, Aranega AM, et al: Histomorphometric analysis of the repair process of autogenous bone grafts fixed at rat calvaria with cyanoacrylate. J Appl Oral Sci 19:529–534, 2011.
Osteoinduction involves the stimulation of osteoprogenitor cells to differentiate into osteoblasts that then begin new bone formation (Fig. 18-1, B). The most widely studied types of osteoinductive cell mediators are bone morphogenic proteins (BMPs). A bone graft material that is both osteoconductive and osteoinductive will serve as a scaffold for existing osteoblasts, and it will also stimulate the formation of new osteoblasts, thereby theoretically promoting the faster integration of the graft.
Types of Bone Grafts
Bone Autografts
Fresh autogenous cancellous and corticocancellous bone are benchmark graft materials that allograft and bone substitutes attempt to match in their in vivo performance.2,25,117–124 They incorporate all of the aforementioned properties, and they have no associated risk of viral transmission. They are incorporated into surrounding bone through creeping substitution. Unfortunately, the availability of an autograft for a specific need may be limited, and the harvest may be associated with donor site morbidity.10
Bone Graft Substitutes
The use of premarket-approved rh-BMP-2 (Infuse Bone Graft; Medtronic, Minneapolis, Minn) as an autograft replacement for spinal fusion and the treatment of open tibia fractures. This substance has also been approved for maxillary sinus augmentation and localized alveolar ridge augmentation.
The use of rh-BMP-7 (OP-1 Implant; Olympus Biotech, Hopkington, Mass) as a humanitarian device. This substance has been approved as an autograft substitute for the non-union of long bones.
The use of rh-BMP-7 (Op-1 Putty; Olympus Biotech, Hopkington, Mass) as a humanitarian device alternative to autograft in compromised patients. This substance is approved for patients who require revision posterolateral lumbar spinal fusion or for those in whom autologous bone and bone marrow recovery (i.e., harvesting) are not feasible or are not expected to promote fusion.
Fresh autogenous cancellous bone and, to a lesser extent, cortical bone are the benchmark graft materials. Their shortcomings include limited availability, donor site morbidity, and the potential for resorption.
The advantages of allogenic bone include its availability in various sizes and shapes as well as the avoidance of host donor harvesting. The transmission of infection (particularly of human immunodeficiency virus) has been virtually eliminated as a concern when the grafts are properly treated.
The ideal bone graft substitute is biocompatible, bioresorbable, osteoconductive, osteoinductive, structurally similar to bone, easy to use, and cost-effective. Bone substitute products are currently available on the market. They vary with regard to their composition and their claimed mechanism of action.
U.S. Food and Drug Administration approvals of specific uses of recombinant human growth factors (i.e., rh-BMP-2 and rh-BMP-7) for the spine and the long bones have been made on the basis of demonstrated bone repair in human trials. Approved head and neck applications are limited but are likely to increase.
The orthognathic surgeon has choices in the realm of bone grafting. Selection should be based on reasonable burdens of proof. Examining the products’ claims and whether such claims are supported by preclinical and clinical studies for the specific sites to be used during clinical practice should be considered.
Grafting Interpositional Defects and Gaps of the Mandible and the Maxilla
Autogenous Iliac (Particulate Cancellous) Graft Donor Site ( Video 8)
Video 8)
Iliac (hip) particulate cancellous bone remains the preferred graft material for the management of congenital cleft defects of the alveolus, the palate, and the floor of the nose.1,9,12,15,26,28,46–48,52,63,66,77,82,90,115 This is especially true when the canine tooth is expected to erupt through the graft (i.e., cleft defect); when a tooth will be orthodontically moved into the grafted site; or when a dental implant will later be placed (Fig. 18-2). The cleft defect that requires a graft may be on one side (i.e., with unilateral cleft lip and palate) or on both sides (i.e., with bilateral cleft lip and palate). Previous attempts at grafting a bone deficiency may have failed as a result of an unsuitable graft material being selected; a soft-tissue deficiency or poor flap management; or inadequate postoperative management or patient cooperation (see Chapters 32 and 33).
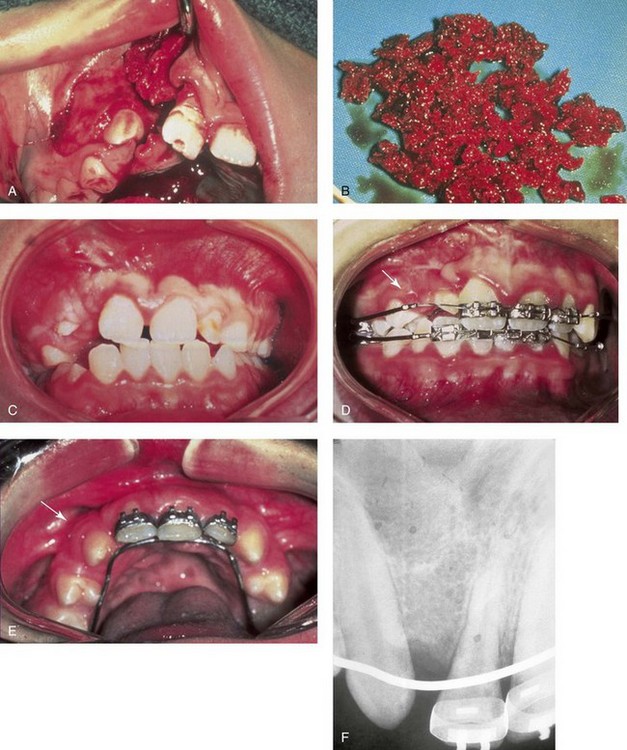
Figure 18-2 A child was born with unilateral cleft lip and palate and underwent arch expansion orthodontic treatment. This was followed by autogenous iliac bone grafting and fistula closure during the mixed dentition. A, Intraoperative view after the elevation of the mucogingival flap indicating the extent of the cleft defect. B, Harvested autogenous cancellous iliac graft used to fill the cleft defect. C, Occlusal view early after surgery. D, Later occlusal view with the early eruption of the canine. E, Occlusal and palate view after the eruption of the permanent canine through the bone graft. The canine and the other posterior teeth are orthodontically guided anteriorly to close the cleft and dental gap in the region of the congenitally missing lateral incisor. F, Periapical radiograph indicating normal alveolar ridge formation after the eruption of the canine through the bone-grafted cleft site.
Donor site discomfort in the anterior iliac region is generally caused by the injury or contusion of the surrounding muscles on the medial (i.e., external oblique, internal oblique, and rectus abdominis) and lateral (i.e., medial gluteus) sides of the bone.* In a thin individual with a minimal fat layer and without baseline ankle, knee, or lumbar region arthritis or myalgia who is relatively young (i.e., <50 years), only minimum discomfort should occur. However, there are reports of complications associated with anterior iliac crest graft harvesting, including gait disturbance, paresthesia, superficial infections, hematoma, poor cosmesis, and chronic donor site pain.6,20,41,42,53,57,81,92,103–105,110,113,114,131 The experienced surgeon can harvest the graft rapidly and efficiently. There is no need for the placement of a drain, minimal blood loss (i.e., <50 cc) is expected, and the head and neck region will have been simultaneously prepped and draped.
Step-by-Step Approach (Fig. 18-3)
Preparation and Draping
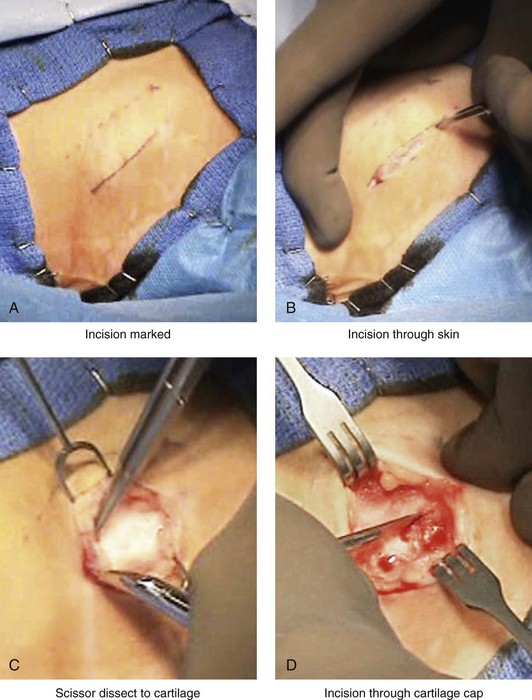
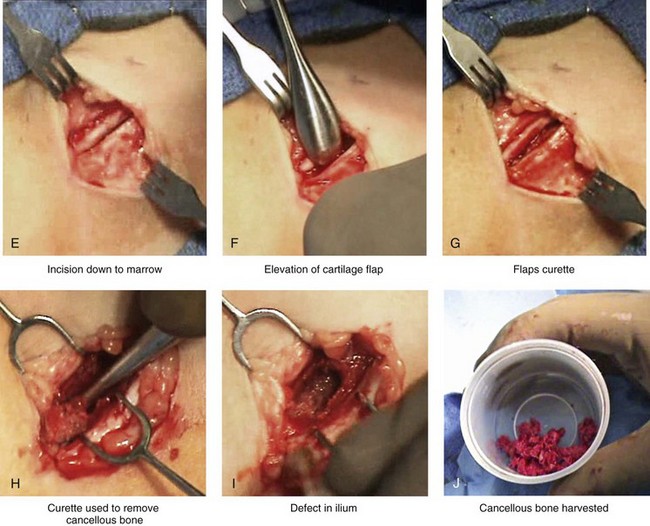
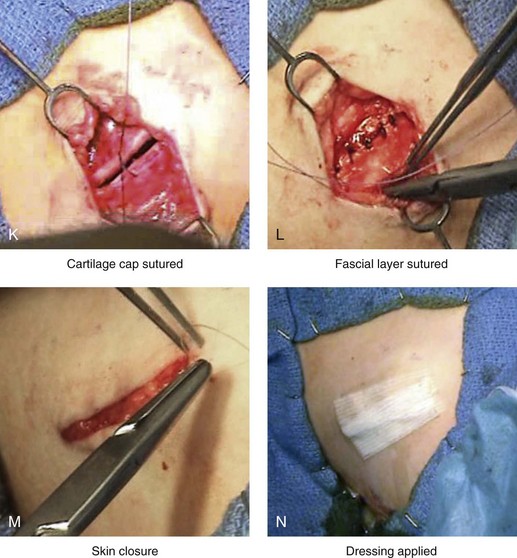
Figure 18-3 Autogenous iliac (particularly cancellous) graft donor site harvesting. A, The planned incision is parallel and 1 cm lateral to the iliac crest. The incision starts 1 cm posterior to the anterior superior iliac spine. B, After the injection of local anesthesia, the incision is carried out through the skin. C, Scissors dissection continues through the subcutaneous tissue and the superficial and deep fascia while avoiding the cutting and contusion of the gluteus muscle laterally or the abdominal oblique and transverse abdominis muscles medially. D, With a knife, the cartilaginous cap is split down the center directly over the crest and down to the marrow cavity. E, With a knife, perpendicular relaxing incisions are completed through the cartilage at either end of the main incision. F, With a periosteal elevator, each half of the cartilaginous cap (i.e., medial and lateral) is elevated off of the underlying marrow space. G, The cancellous marrow bone is now exposed. H, Exposed cancellous bone is harvested with curettes. I, With the necessary cancellous bone harvested, a defect in the marrow space can be seen. J, Harvested cancellous marrow bone to be used for reconstruction. K, The cartilaginous cap is repositioned back in place with interrupted ties (3-0 Vicryl). L, The deep and superficial fascia layers are closed with interrupted suture ties (3-0 Vicryl). M, Subdermal closure is performed with interrupted suture ties (4-0 Vicryl). The skin is closed with subcuticular running suture (5-0 Monocryl). N, Steri-Strips and then an occlusive dressing (Tegaderm) are placed over the skin.
• An endotracheal tube is placed and secured in accordance with the patient’s head and neck reconstruction needs.
• Ophthalmic ointment and corneal shields are placed for eye protection.
• The patient is supine on the operating room table.
• The patient’s arm is tightly tucked to his or her side.
• Betadine solution is used for both hip and head and neck preparation.
• At the hip, the patient is prepped halfway down the thigh, to the groin, across the abdominal midline, up to the rib cage, and posterior down the side of the buttock.
• Draping is completed, with limited exposed skin left around the planned incision site.
Skin Incision and Flap Elevation
• The incision is carried through the skin, the subcutaneous tissue, and the superficial and deep fascia directly over the iliac crest where the graft is to be harvested.
• The cutting or contusion of the gluteus muscle laterally or of the abdominal oblique and transverse abdominis muscles medially is to be avoided. Injury to these muscles when approaching the iliac crest should be avoided, because this is the primary cause of buttock and abdominal wall discomfort after surgery.
Graft Harvesting
Cartilage Cap Over Crest (Patient Typically <12 Years Old)
• If a cartilaginous cap remains over the crest, it is split down the center directly over the crest and down to the marrow cavity. Perpendicular relaxing incisions are completed through the cartilage at either end of the main incision. This allows for the lifting of each half of the cartilage cap (i.e., medial and lateral) off of the underlying marrow space. The lifting of the vascularized cartilage flaps is accomplished with a periosteal elevator.
• The exposed cancellous bone is then harvested with curettes.
No Cartilage Cap Over Crest (Patient Typically >12 Years Old)
• When the iliac crest is mature, no cartilaginous cap remains. In this case, a window of cortical bone is removed from the medial aspect of the crest and down the medial plate for approximately 2 cm. This is an efficient method for the exposure of the medullary cavity.
• The subperiosteal dissection of the medial crest and down the medial plate is followed by the placement of a toed-out retractor down the medial plate.
• The removal of the cortex is facilitated with the following: 1) an oscillating saw with a short fan blade on a long shaft to cut through the cortex of the medial plate at the inferior aspect; 2) an oscillating saw with a wide fan blade on a short shaft to cut through the cortex of the medial crest; and 3) a reciprocating saw with a short, straight blade to cut through the cortices of the medial plate to connect the other two cuts.
• The removal of the window of cortical bone is then easily accomplished with the use of a chisel and a mallet.
• With the window of cortical bone removed, cancellous marrow is harvested with curettes.
• The removed cortical bone may also be chopped or used as an intact cortical graft. There is no advantage to replacing the small cortical bone segment, because the strength of the crest has not been compromised. The bone defect will not be visible or palpable through the closed skin wound. The removed marrow and cortical bone regenerate themselves.
Wound Closure
• After adequate graft is harvested, bone wax is used for hemostasis. The bone wax is not used to fill the cavity; rather, a thin layer is placed directly over the bleeding medullary surfaces. Interestingly, the wax does not prevent the regeneration of bone that will fill the cavity.
• If present, the cartilaginous cap is repositioned and sutured back into place with interrupted ties (3-0 Vicryl). If a cap is not present, then the overlying periosteum is closed with interrupted ties (3-0 Vicryl).
• The deep and superficial fascia layers are closed with interrupted suture ties (3-0 Vicryl).
• Subdermal closure is performed with interrupted suture ties (4-0 Vicryl).
• The skin is closed with subcuticular running suture (5-0 Monocryl).
Autogenous Iliac (Corticocancellous Bloc) Graft Donor Site ( Video 9)
Video 9)
When a Le Fort I osteotomy with significant horizontal advancement and vertical lengthening is carried out, an interpositional defect (i.e., dead space) is created. Despite rigid plate and screw fixation of the repositioned maxilla, inadequate bone contact may jeopardize successful healing (e.g., fibrous union; see Chapter 16) or leave the upper jaw prone to skeletal relapse (see Chapter 17). The advantage of placing an interpositional graft is sometimes obvious and at other times borderline. When an interpositional graft is deemed necessary, I prefer to use either autogenous or allogenic iliac bone.107 A crafted corticocancellous anterior iliac (hip) graft is interposed between the pyriform and the zygomatic titanium plates that are used to secure the upper jaw in its new location (Fig. 18-4). The graft is tightly wedged between the advanced and lengthened anterior maxillary wall and the more posterior baseline maxillary wall. Any sharp edges of the inset graft are smoothed using a rotary drill with a watermelon bur. The graft is then fixed in place with an additional titanium plate and screws.
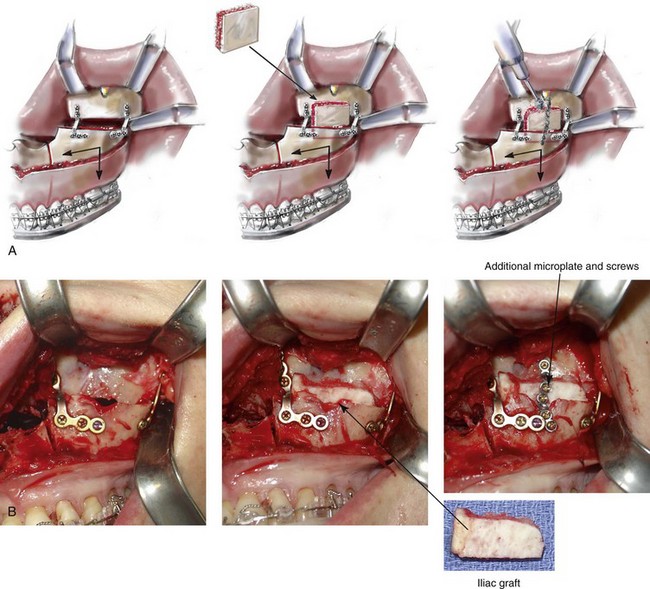
Figure 18-4 When the extent of horizontal advancement and vertical lengthening of the maxilla after Le Fort I osteotomy are judged to be significant, then an interpositional bone graft may be required and beneficial for the achievement of precise healing. In these circumstances, I prefer to use either autogenous or allogenic iliac bone graft. A bloc of corticocancellous bone is crafted and tightly wedged into the gap on each side. Each graft is inset in between the pyriform rim and zygomatic buttress titanium plates. An additional plate is then contoured to extend from the anterior maxilla above, across the graft, and then onto the alveolar process of the anterior maxilla. Titanium screws (1.2 mm in diameter) are then used to secure the plate to the bone. A, Illustrations and B, intraoperative views of interpositional corticancellous iliac graft in a Le Fort I osteotomy site.
Another indication for a corticocancellous bloc graft is when a ramus osteotomy (e.g., an inverted L or straight horizontal osteotomy) is completed. After the proximal segment is seated with the condyle in the glenoid fossa (i.e., the terminal hinge position) and the distal mandible is secured to the maxillary teeth via intermaxillary fixation, a significant interpositional gap may remain. In these circumstances, a crafted corticocancellous iliac graft can be tightly interposed between the proximal and distal segments with additional cancellous bone packed into the remaining dead space. Rigid plate and screw fixation of the osteotomy segments and the graft is always required (see Chapter 28).
When an anterior maxillary or mandibular segmental alveolar defect requires reconstruction (e.g., after trauma or tumor resection), a crafted corticocancellous anterior iliac graft is generally preferable to other options. After successful graft healing (i.e., 4 to 6 months), dental rehabilitation including implant and crown placement may follow (see Chapter 35).2
Donor site discomfort in the anterior iliac region is generally caused by the injury or contusion of the surrounding muscles on the medial (i.e., external oblique, internal oblique, and rectus abdominis) and lateral (i.e., medial gluteus) sides of the bone.* In a thin individual with a minimal fat layer and without baseline ankle, knee, or lumbar region arthritis or myalgia who is relatively young (i.e., <50 years old), only minimum muscle contusion and discomfort should occur. However, there are reports of complications associated with anterior iliac crest graft harvesting, including gait disturbance, paresthesia, superficial infections, hematoma, poor cosmesis, and chronic donor site pain.6,20,41,42,53,57,81,92,103,104,105,110,113,114,131 The experienced surgeon can harvest the graft rapidly and effectively. In general, there is no need for the placement of a drain, minimal blood loss (i.e., <50 cc) is expected, and the head and neck region will have been simultaneously prepped and draped.
Step-by-Step Approach (Fig. 18-5)
Preparation and Draping
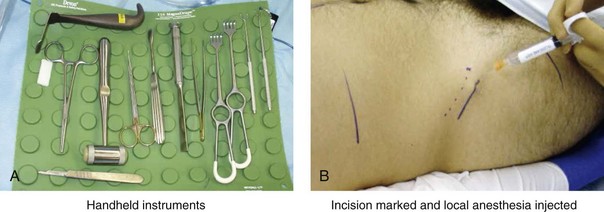
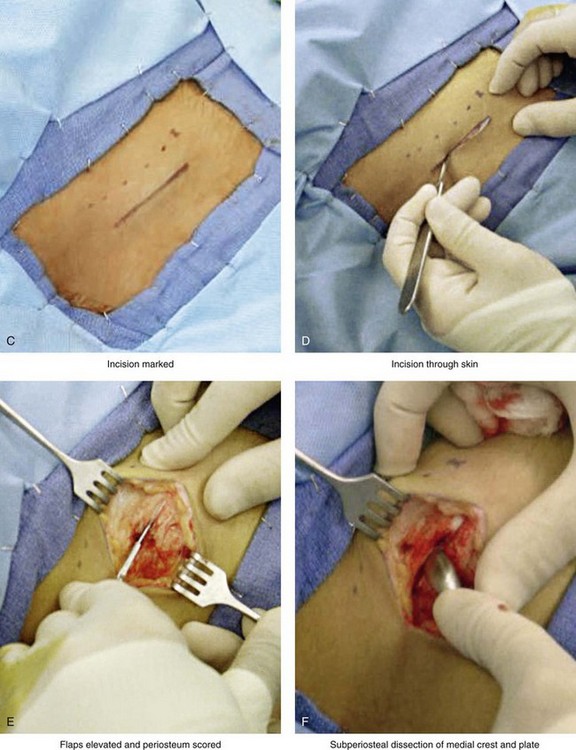
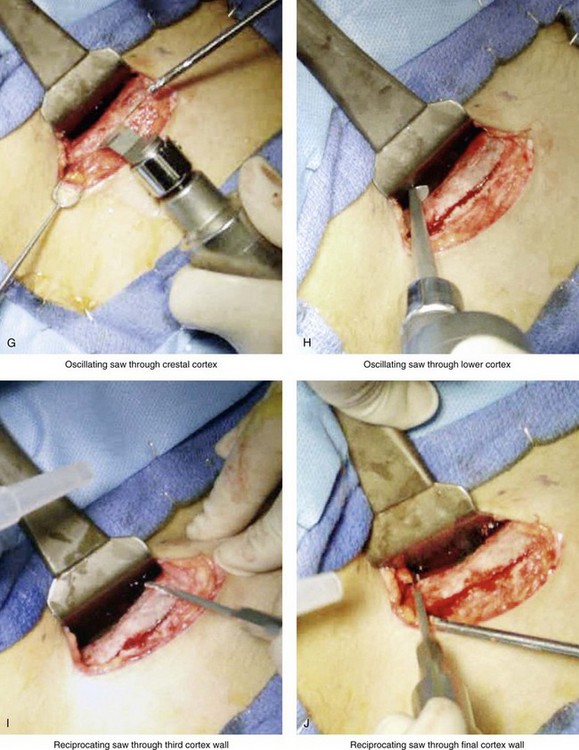
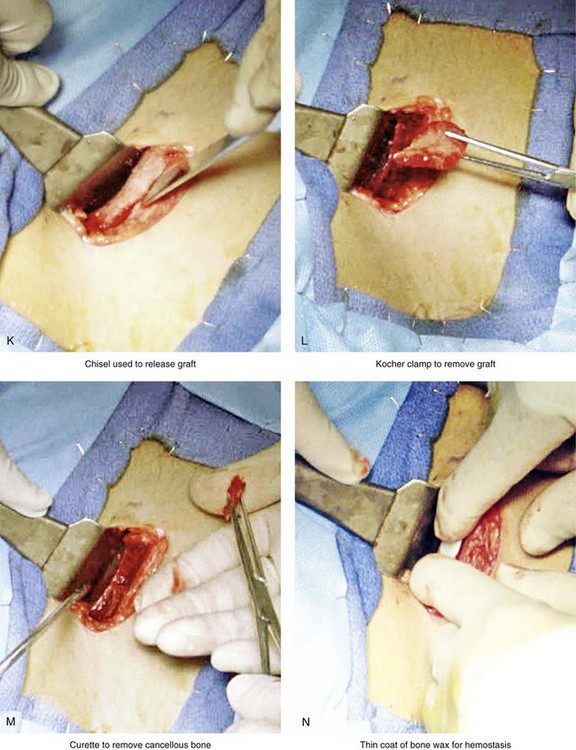
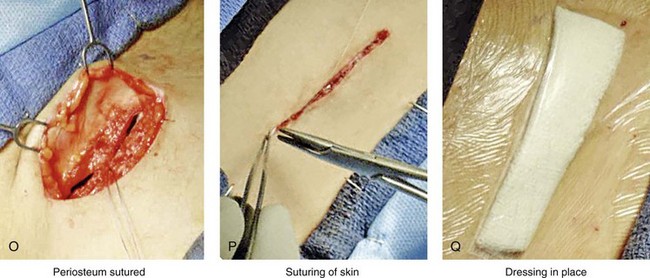
Figure 18-5 Intraoperative views of the harvesting of autogenous iliac corticocancellous bone. A, The basic handheld instruments that are used to harvest the graft. B, The planned incision is parallel and lateral to the iliac crest. It starts just posterior to the anterior superior iliac spine. Local anesthesia is injected below the skin directly over the surgical marking and down to the periosteum over the iliac crest. C, The planned incision is shown. D, With a knife, the incision is carried through the skin and the subcutaneous tissue. The scissors dissection continues through superficial and deep fascia directly over the crest, where the graft is to be harvested. The contusion of the gluteus muscle laterally or the abdominal oblique and transverse abdominis muscles medially is avoided. E, With the use of a knife, the periosteum is scored directly over the midline of the crest from just posterior to the anterior superior iliac spine. The incision continues posteriorly for a distance that depends on the extent of exposure required for the planned graft. F, Subperiosteal dissection of the medial crest and down the medial plate continues. G, A towed-out wide retractor is placed down the medial plate. The efficient removal of a bloc of corticocancellous bone graft is performed by first cutting through all four cortical wall borders. An oscillating saw with a wide blade on a short shaft is used to cut through the wall directly over the medial aspect of the crest. H, An oscillating saw with a short fan blade on a long shaft is next used to cut through the inferior cortical wall. I, A reciprocating saw with a short straight blade is used to cut through the lateral wall. J, A reciprocating saw with a short straight blade is used to cut through the last cortical wall. K, The separation and removal of the corticocancellous block is accomplished with the use of a chisel that is 10 mm in width. After the chisel is in place, a twisting motion is carried out to fully separate the graft. L, The bloc graft is removed with a Kocher clamp. M, Additional cancellous marrow is harvested with curettes as needed. N, A thin coat of bone wax is placed onto the marrow walls for hemostasis. O, The periosteum is closed with interrupted suture ties (3-0 Vicryl). P, After closing the deep and superficial fascia layers with interrupted sutures (3-0 Vicryl), subdermal and then subcuticular closure are accomplished (5-0 Monocryl). Q, Steri-Strips and then an occlusive dressing (Tegaderm) are placed over the skin.
• An endotracheal tube is placed and secured in accordance with the patient’s head and neck reconstruction needs.
• Ophthalmic ointment and corneal shields are placed for eye protection.
• The patient is supine on the operating room table.
• The patient’s arm is tightly tucked to his or her side.
• Betadine solution is used for both hip and head and neck preparation.
• At the hip, the patient is prepped halfway down the thigh, to the groin, across the abdominal midline, up to the rib cage, and posterior down the side of the buttock.
• Draping is completed, with limited exposed skin left around the planned incision.
Skin Incision and Flap Elevation
• The incision is carried through the skin, the subcutaneous tissue, and the superficial and deep fascia directly over the iliac crest where the graft is to be harvested.
• The cutting or contusion of the gluteus muscle laterally or of the abdominal oblique and transverse abdominis muscles medially is to be avoided. Injury to these muscles when approaching the iliac crest is the primary cause of buttock and abdominal wall discomfort after surgery.
Graft Harvesting
• With the use of a knife (no. 15 blade), the periosteum is scored directly over the midline of the crest from just posterior of the anterior superior iliac spine. The incision continues posteriorly for a distance that is dependent on the extent of exposure required for the planned graft.
• Subperiosteal dissection of the medial crest and down the medial plate continues. A toed-out retractor is placed down the medial plate.
• The efficient removal of a bloc of corticocancellous bone graft is accomplished using an oscillating saw (with a short fan blade on a long shaft and a wide fan blade on a short shaft) and a reciprocating saw (with a short, straight blade) to cut through all four bordering cortical walls.
• The corticocancellous bloc graft is then removed from the medial aspect of the crest and down the medial plate. The length of the graft along the crest and its depth into the medullary cavity are variable, depending on the patient’s reconstructive needs.
• The actual removal of the corticocancellous bloc is accomplished with the use of a 10-mm chisel and a twisting motion to fully separate the graft.
• With the bloc graft removed, additional cancellous marrow is harvested with curettes, as needed.
• The strength of the crest and of the ilium as a whole should not be compromised by the bloc graft removal. In general, the bone defect will not be detectable through the closed skin wound, because the lateral aspect of crest is preserved. The removed marrow and the cortical bone will regenerate, often with greater than normal thickness.
Wound Closure
• After adequate graft is harvested, bone wax is used for hemostasis. The bone wax is not used to fill the cavity; rather, a thin layer is placed directly over the bleeding medullary surfaces. The wax should not prevent bone regeneration.
• The periosteum is closed with interrupted suture ties (3-0 Vicryl).
• The deep and superficial fascia layers are closed with interrupted suture ties (3-0 Vicryl).
• Subdermal closure is performed with interrupted suture ties (4-0 Vicryl).
• The skin is closed with subcuticular running suture (5-0 Monocryl).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


