16
General oncology
Introduction
Oncology refers to the branch of medicine that deals with tumours, literally meaning the science of new growths, whilst the term cancer is applied somewhat more loosely to malignant tumours in general. Cancer is essentially a genetic disease caused by somatic mutations. The multistage theory of carcinogenesis believes that individual cancers arise from several sequential mutations in cellular DNA. There is a close correlation between cancer incidence and increased age, reflecting the time required to accumulate the critical number of genetic abnormalities needed for malignant change.
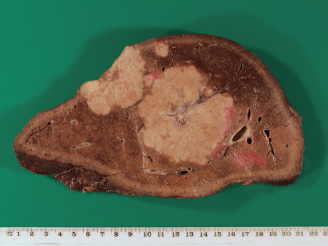
Malignant tumours defy the normal growth control mechanisms of the body, leading to excessive cell proliferation, loss of differentiation and ultimately local tissue invasion, destruction and widespread metastasis (spread of primary tumours to distant body sites as secondaries). Death may result from tumour deposition within vital organs such as the liver (Fig. 16.1), lungs or brain, from generalised effects of cancer, e.g. metabolic, or from uncontrolled disease at primary tumour sites.
Cancer may affect any organ or tissue in the body, but arises commonly from epithelial tissues of the skin, lungs and gastrointestinal tract which are directly affected by carcinogens (cancer-inducing agents) such as radiation, tobacco smoke, chemical agents in foodstuff, and alcohol.
In order to appreciate the nature and significance of malignant disease in the body, it is necessary to understand how the normal physiological control of cell proliferation, tissue growth and differentiation becomes disrupted during tumourigenesis.
Disorders of growth and differentiation
Introduction
Coordinated cell proliferation, differentiation and tissue morphogenesis are essential for the successful development and maintenance of a large complex multicellular organism. The process of embryogenesis starts with a single cell, the fertilised ovum. The genetic information encoded within this cell is the blueprint for life. During cell division, the genetic information is precisely copied and disseminated to the daughter cells. It is the coordinated expression of this genetic information (genotype) that determines the fate of the cell and its role in the organism (phenotype).
The progeny of stem cells are destined to become specialised cells with specific functions in the organism (differentiation). There is accumulating evidence that sets of genes, e.g. homeobox genes, are important in determining the movement and physical positioning of cells (morphogenesis). The process of apoptosis (programmed cell death) is also essential in development and tissue maintenance, to counterbalance excessive proliferation and delete unwanted cells.
In addition to increase in cell numbers, termed multiplicative growth, individual cells can also increase in size, which is called auxetic growth. Skeletal muscle increases in size by auxetic growth. In some tissues, e.g. bone, growth is mainly achieved by deposition and remodelling of extracellular matrix, which is called accretionary growth.
When considering multiplicative growth, cells in the adult can be classified by their potential to proliferate. This determines the ability of different tissues to respond to environmental changes and tissue damage. Labile cells are continuously lost and replaced. They include the haemopoietic cells of the bone marrow, and epithelial cells that constitute the epidermis and line the mucous membranes. Stable cells are not continuously replaced, but can be induced to proliferate in certain conditions, e.g. hepatocytes of the liver and renal tubular cells of the kidney. Non-dividing cells cannot be stimulated to proliferate, and include cardiac muscle fibres and neurons. Intuitively, labile cells are the most susceptible to disorders of growth, whereas non-dividing cells are unaffected.
Figure 16.1 A section of a liver showing metastatic deposits.
Disorders of growth
Within specific tissues and organs, alteration in growth takes place as an adaptive response following a change in demand (physiology) or underpins part of a disease process (pathology). Growth as a consequence of increased cell number is termed hyperplasia. Hyperplasia is mainly a consequence of increased mitosis, but reduction of apoptosis will also result in accumulation of cells. In contrast, growth as a result of increased cell size, without cell division, is termed hypertrophy. In most tissues, increased growth is a combination of hyperplasia and hypertrophy.
A good example of physiological hyperplasia is the increase in the number of erythrocytes when an individual moves to a high altitude location. The reduced partial pressures of oxygen at high altitude stimulates the body to produce more erythrocytes in order to optimise oxygen delivery to the tissues. Athletes sometimes use ‘altitude training’ as a performance-enhancing strategy. Following a period of training at high altitude, which results in erythrocyte hyperplasia, when the individual competes at sea level the body has an increased oxygen-carrying capacity, with greatly increased delivery of oxygen to the peripheral tissues. The illegal use of erythropoietin (EPO) by athletes, a hormone that stimulates the production of erythrocytes, is another method of inducing erythrocyte hyperplasia. Another example of physiological hyperplasia is the increase in the amount of breast tissue observed during the development of the mammary glands in puberty and the increase in breast size during pregnancy in preparation for lactation. Hyperplasia is also characteristic of some diseases, e.g. a generalised increase in the size of the thyroid gland is called goitre. In some circumstances, the hyperplasia is a consequence of inappropriate stimulation of the thyroid gland by an autoantibody that simulates the action of thyroid-stimulating hormone, which is called Graves’ disease. The hyperplastic thyroid tissue produces excess thyroxine (hyperthyroidism) and the patient develops clinical symptoms that include weight loss, tremor, sweating, tachycardia and heat intolerance.
Physiological hypertrophy of skeletal muscle occurs following increased usage and physical demand. Muscle hypertrophy observed in ‘body builders’ and athletes is a striking example. Hypertrophy of the cardiac muscle fibres in the left ventricle is an adaptive response to sustained peripheral resistance (hypertension) and is also a feature of hypertrophic cardiomyopathy, a cause of unexpected death in young adults (see Chapter 5A).
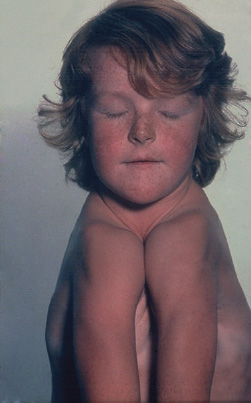
Terms used to describe inadequate growth include aplasia, hypoplasia and atrophy. Aplasia is a complete failure of an organ or tissue to develop. Aplasia of organs is uncommon, and in most cases it is incompatible with life. Examples of viable infants with aplasia include unilateral aplasia of a kidney and thymic aplasia in DiGeorge syndrome, which results in severe immunodeficiency. Cleidocranial dysostosis is an inherited skeletal defect characterised by abnormalities of the skull and clavicles. Some of these individuals have aplasia of the clavicles and are able to adduct the shoulders to the midline (Fig. 16.2).
Hypoplasia is used to describe the partial failure of development of an organ or tissue, such that the normal size is not attained. Hypoplasias are more common than aplasia and are seen in a variety of inherited diseases, e.g. hypoplastic left heart syndrome, testicular hypoplasia in Klinefelter’s syndrome, segmental renal hypoplasia and optic nerve hypoplasia.
The term atrophy is used to describe a decrease in size of an organ or tissue. Atrophy may be the result of a reduction in cell size or cell number, or a combination of both. Confusingly, the terms hypoplasia and atrophy are often used synonymously; however, hypoplasia should be used to indicate a failure in attainment of normal size in the context of development, whereas atrophy should be used to indicate a reduction in the size of an organ or tissue following complete development. Examples of physiological atrophy include atrophy of the ductus arteriosus in the neonate and thymic atrophy following T cell development in early adulthood. Disuse atrophy of muscles and osteoporosis can occur during prolonged periods of immobility, and the loss of the use of a limb following a fracture or denervation of a muscle group. Disuse atrophy is usually reversible following a return to normal activity. Cachexia is a ‘wasting syndrome’ seen in chronic disease, particularly cancer, which is typified by weight loss and attendant muscle atrophy. A summary of growth disorders is given in Table 16.1.
Figure 16.2 Cleidocranial dysostosis.
Disorders of differentiation
Differentiation is the process whereby cells become specialised and are assigned specific functions within the organism. Disorders of differentiation and morphogenesis are important when considering abnormalities that arise during embryogenesis. In the adult, changes in the differentiation of cells are classified as metaplasia and dysplasia.
Table 16.1 Disorders of growth
|
|
|
|
|
|
Metaplasia is a change in the normal pattern of differentiation to another type of differentiation. Metaplasia is usually an adaptive response to changes in the environment. Changes in the differentiation of specialised epithelia are common. For example, squamous metaplasia can be induced in respiratory-type epithelium (ciliated pseudostratified epithelium) during chronic exposure of the respiratory tract to tobacco smoke. Squamous metaplasia of ductal epithelium is seen in chronic sialadenitis and sialolithiasis (salivary gland stones). Squamous epithelium can be induced to undergo glandular metaplasia, sometimes called mucous metaplasia. Chronic gastric reflux into the lower part of the oesophagus causes the original squamous epithelium to be replaced by specialised columnar epithelia that contains mucus-secreting goblet cells, which resembles the lining of the colon. These changes are called Barrett’s oesophagus, which is associated with an increased risk of developing oesophageal adenocarcinoma.
Metaplasia can also occur in mesenchymal tissues, e.g. the development of bone in anatomical structures composed of cartilage, such as the larynx, trachea and bronchi, is called osseous metaplasia. Osseous Metaplasia may also develop in the complicated plaque of atherosclerosis.
Dysplasia is a term used to describe disordered tissue architecture. It develops as a consequence of increased cell growth and altered cellular differentiation. The term is most frequently used to describe the histological changes in the epithelia that comprise skin and mucous membranes; epithelial dysplasia. The term is also used to describe changes in other tissues, e.g. the myelodysplastic syndromes of the haemopoietic system and fibrous dysplasia of bone. Epithelial dysplasia is commonly detected in biopsy material from the mucosa of the upper aerodigestive tract and uterine cervix. In the upper aerodigestive tract there is strong correlation between epithelial dysplasia and chronic exposure to tobacco smoke, whereas, in the cervix, exposure to human papilloma virus (HPV) infection is the strongest risk factor (Table 16.2).
Pathologists grade epithelial dysplasia based on assessment of the architectural and cytological changes (Table 16.3). Grading systems vary, but the changes are most commonly classified into low and high grade dysplasia or termed mild, moderate and severe dysplasia. It is known that lesions with epithelial dysplasia have an increased risk of developing into cancer; this has led to a variety of terms for dysplastic lesions that include potentially malignant lesions, pre-malignant lesions, precursor lesions and pre-cancer.
Table 16.2 Disorders of differentiation
|
|
Table 16.3 Parameters used to render a diagnosis of epithelial dysplasia
| Architecture | Cytology |
| Irregular stratification of the epithelium | Abnormal variation in cell size (anisocytosis) |
| Loss of basal cell polarity | Abnormal variation in cell shape (cellular pleomorphism) |
| Drop-shaped rete processes | Abnormal variation in nuclear size (anisonucleosis) |
| Increased number of mitotic figures | Abnormal variation in nuclear shape (nuclear pleomorphism) |
| Suprabasal mitotic figures | Increased nuclear:cytoplasmic ratio |
| Single cell dyskeratosis | Increased nuclear size |
| Keratin pearls within rete processes | Hyperchromasia |
| Increased number and size of nucleoli | |
| Atypical mitotic figures |
Neoplasia
Definitions and terminology
Neoplasia means new growth (Greek: neo new, plassein to form). The most widely cited definition is that devised by the eminent British pathologist Professor R.A. Willis in the 1950s. ‘A neoplasm is an abnormal mass of tissue, the growth of which exceeds and is uncoordinated with that of the normal tissues, and that persists in the same excessive manner after the cessation of the stimulus which evoked the change’. This precise definition separates neoplasia from the other abnormalities of growth and differentiation, described above, and emphasises the inherent growth potential of a neoplasm. Perhaps a contemporary definition should also take account of the genetic basis of neoplasia, e.g. a neoplasm is a growth disorder characterised by genetic alterations that lead to loss of the normal control mechanisms that regulate cell growth and differentiation.
The term tumour is used synonymously with neoplasm, but literally means swelling and was originally used to describe swellings caused by inflammation (tumor: one of the cardinal signs of inflammation). Hamartomas can also produce a tumour-like mass of tissue that mimics neoplasia. Hamartomas represent an overgrowth of tissue that is typically indigenous to the site, grows with the individual and has a limited growth potential. A common hamartoma is the melanocytic naevus or ‘mole’. Another example is the pulmonary hamartoma, which appears as a coin-shaped radiodensity on chest radiography and is composed of respiratory-type epithelium admixed with mature cartilage. The vascular abnormalities, haemangioma and lymphangioma, are also best considered as hamartomas rather than true neoplasms.
Neoplasms are classified as benign or malignant depending on their clinical presentation, histological appearance and biological behaviour. A general term for malignant neoplasia is cancer, which is an emotive term that patients associate with suffering and death.
General features of neoplasia
Benign neoplasms tend to be small; however, occasionally, large benign neoplasms develop because they do not cause any significant symptoms. In other circumstances, a patient may not have sought treatment or been able to access appropriate healthcare services, e.g. in underdeveloped countries. Benign tumours are invariably well circumscribed and often delineated by a rim of compressed fibrous tissue, called a capsule (encapsulated). Benign tumours are composed of mature tissue that resembles the tissue of origin. The growth rate is slow and mitotic figures are infrequent. The majority of benign tumours do not show any evidence of local tissue invasion, although there are a few exceptions to this rule, most notably ameloblastoma, which is a benign odontogenic tumour that shows bone invasion. By definition, benign tumours remain localised and do not spread to form new neoplasms at other sites (Table 16.4).
The majority of malignant neoplasms tend to be rather large at diagnosis. They invade and destroy local tissues and, as a consequence, have ill-defined borders and are fixed to the adjacent tissues (Fig. 16.3). Malignant neoplasms show varying degrees of resemblance to the tissue of origin and are graded depending on the degree of differentiation, namely: well differentiated, moderately differentiated and poorly differentiated. The growth rate is variable, but usually more rapid than that of benign neoplasms. Mitotic figures are usually easily identified and some may look abnormal. In rapidly growing neoplasms, the growth may exceed the development of an adequate blood supply, leading to necrosis. Malignant neoplasms invade and destroy the surrounding tissues. There may be invasion along peripheral nerves, termed neural invasion.
Table 16.4 Typical features of benign and malignant neoplasms
| Benign | Malignant | |
| Size | Small | Large |
| Borders | Circumscribed/encapsulated | Ill defined |
| Differentiation | Resembles tissue of origin | Variable |
| Growth rate | Slow | Rapid |
| Mitotic figures | Rare |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


