3
Principles of infection and infection control
3A Sterilisation, disinfection and antiseptics
Sterilisation and disinfection
Sterilisation has been defined as the killing or removal of all viable organisms. Concern about transmissible spongiform encephalopathies such as Creutzfeldt–Jakob disease (CJD) and particularly variant-CJD has generated research which has resulted in a much greater understanding of prions, the transmissible agents of the disease. The debate about whether prions are organisms probably reflects similar discussions decades ago regarding the nature of viruses, but the absence of even nucleic acid puts prions in a class apart, at least for now. The fact that prions have been identified as a transmissible agent of disease means that any definition of sterilisation has to take them into account. Sterilisation is therefore more accurately defined as: the inactivation or removal of all self-propagating biological entities.
Disinfection is a less precise concept and has been variously defined. The best definition is the reduction in viable organisms to the point where risk of infection is acceptable.
A related term that is in common use is antisepsis, which is usefully defined as the disinfection of skin or wounds.
General principles
The efficacy of a sterilisation or disinfection method is described by the relationship
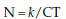
where N is the number of surviving organisms, C is the concentration of the sterilising agent (temperature in the case of heat) and T is the time for which the agent is applied.
The constant k depends on many factors including the species of organism present, its physiological state and many other environmental variables not least among which is the presence of contaminating organic material such as blood or saliva.
For a given value of C, a graph of Log10 viable count versus time can be useful to predict when sterility is achieved (Fig. 3A.1). The slope of the curve is known as the ‘death rate’ and D is the time taken to achieve a 90% reduction in viable organisms. Survival curves such as the one shown may have a shoulder, a tail or both, signifying that individual cells respond differently to the sterilising agent. In practice, this means that absolute sterility cannot be guaranteed. Therefore, by convention, an instrument is considered sterile if there is less than a one in a million chance of there being one viable organism present.
What should be sterilised?
Ideally, everything in a surgery should be sterile, but this is not practicably possible. The aim is to prevent infection and, importantly, cross-infection. Therefore, anything coming into direct contact with the surgical site should be sterile and everything else should be disinfected.
Decontamination and pre-cleaning
The presence of contaminating material especially organic matter such as saliva, blood, faeces and tissue can significantly degrade the sterilisation or disinfection process by either physically preventing access of the agent or chemical inactivation. In order to ensure proper sterilisation, instruments should therefore be precleaned. If the instruments have been in contact with infectious material the pre-cleaning should include adequate disinfection to render them safe before they are packaged for sterilisation.
Figure 3A.1 A graphical demonstration of viable organism reduction with time (see text).
Sterilisation and disinfection methods
Methods of sterilisation and disinfection include dry or moist heat, a wide variety of liquid or gaseous chemicals, β- or γ-emitting ionising radiation and filtration (Table 3A.1). The choice of method depends on the nature of the material being treated, the contaminating organisms and the degree of inactivation required. For example, it is possible to sterilise milk, but this effectively changes its character. On the other hand, it is possible to retain the character and reduce its microbial load to a level that increases the shelf-life of milk appreciably by raising its temperature for a period of time (the pasteurisation process).
Monitoring efficacy
It used to be the practice to include test organisms in every batch of material being sterilised; however, this results in a delay before sterility can be confirmed, which is often unacceptable. In modern practice, procedures are followed that are known to result in sterility for different materials and batch sizes, and the performance of the equipment in terms of the temperature and holding time is carefully monitored.
Table 3A.1 Methods of sterilisation
|
|
|
|
| gases |
| alcohols |
| aldehydes |
| bisbiguanides |
| chlorine compounds |
| iodophores |
| strong oxidising agents |
| phenols |
| quaternary ammonium compounds |
Figure 3A.2 Bowie–Dick tapes on a surgical pack. The brown cross-hatchings demonstrate that the autoclaving process has been carried out. The lower horizontal tape shows a tape which has not been successfully subjected to the autoclaving process.

The Bowie–Dick tape is a method of ensuring that an autoclave has been functioning appropriately (Fig. 3A2). When the cross-hatchings on the tape turn brown, the sterilisation process has been effective.
Heat
Heat is the most familiar of methods used because it is versatile, reliable, easy to control, cheap and efficient. It works by transferring energy to the organism and, depending on the conditions chosen, it can be used to sterilise or disinfect.
Dry heat
Dry heat sterilisation refers to conditions in which there is an absence of water in the environment being heated but this does not include any water present in the object being sterilised. Dry heat sterilisation therefore includes a wide variety of conditions in which the level of moisture present in the system as a whole varies between complete dryness and almost complete saturation. This relative humidity affects the efficiency of the process in a complex manner. For example, the time required to effect a 99.99% reduction in viability of Bacillus stearothermophilus spores at 120°C is approximately 6 min in the absence of any moisture, rising to a maximum of approximately 9 hours at a relative humidity of 30% and then dropping to 35 min near saturation. Moreover, different organisms vary in their susceptibility to dry heat. There are significant practical consequences of this relationship because in a closed system there may be pockets of air trapped in the material being sterilised. This will become more or less saturated with water vapour depending on a variety of conditions in the immediate vicinity, such as the porosity of the material, the overall amount of air and water present and the volume of the container. It is, therefore, imperative that these and other factors are taken into account if sterility has to be guaranteed.
Dry heat sterilisation is used to sterilise solid material such as glassware and some instruments, which are susceptible to corrosion, non-aqueous liquids and heat-stable powders such as drugs.
Moist heat
Sterilisation by moist heat depends on the use of saturated water vapour (steam) usually at a temperature between 121 and 134°C for various periods. Typical conditions are 15 min at 121°C; 10 min at 126°C; and 3 min at 134°C. Steam can only be heated above 100°C by increasing the pressure above normal atmospheric pressure. It is imperative that the system is fully saturated with water vapour so the equipment used to perform moist heat sterilisation (autoclave) is designed so that when operated properly, all other gases are excluded. This is achieved either by downward displacement by the steam which forces the less dense air out through a drain, by vacuum pumps or by steam pulsing.
Moist heat sterilisation is much more efficient than dry heat for two reasons. First, it takes less heat to denature fully hydrated proteins and, secondly, during the process, moist heat releases its latent heat of vaporisation which transfers considerably more energy than dry heat.
Irradiation
Ionising radiation is commonly used to sterilise large numbers of small items such as syringes, needles, catheters and gloves, but because of associated hazards it is only suitable for use in specially constructed commercial environments. γ-Rays, X-rays and accelerated electrons (β-rays) are all commonly used at a dose in the region of 2.5 Mrad. Irradiation is a very effective means of achieving sterilisation; however, there is inevitable damage to the material being irradiated so it is mostly used for single-use items. If an item is to be repeatedly irradiated then a careful log must be kept to ensure the damage is kept within known limits.
Radiation of each type works by producing radicals that damage cellular DNA. Common sources of radicals are water (OH· and H·), usually intracellular water, and oxygen (O·). Radiation is, therefore, normally carried out in an aerobic atmosphere. In the absence of oxygen, the dose must be increased.
Filtration
Filtration is different from other methods of sterilisation or disinfection because it works by physically removing microorganisms rather than killing or inhibiting them. It is used when heat, irradiation or chemical treatment would result in damage to the substance being sterilised. It is also used to sterilise products such as intravenous fluids when heat treatment would release breakdown products of microorganisms which, because they cause fever, are known as pyrogens.
Filtration is remarkably versatile and is widely used throughout the pharmaceutical and water industries. It also has a wide application in scientific work because the pore sizes of membranes can be controlled with such precision as to allow separation of macromolecules.
Chemicals
Gases
The gas most commonly used to sterilise medical equipment is ethylene oxide. In some cases formaldehyde gas is used, but this is less efficient and is only used when ethylene oxide is not available. More commonly formaldehyde is used as a fumigant to disinfect rooms or equipment such as workstations, which pose a biohazard threat.
Ethylene oxide is used to sterilise items that are sensitive to moisture or heat above 60°C. It works by alkylating microbial proteins and nucleic acids and must come into direct contact with the object being sterilised, so any packaging must be porous. After contact, sufficient time must elapse to allow removal of gas residue.
Antiseptics
Terminology
Antiseptics are substances used to destroy microorganisms on living tissues, such as the dentist’s hands. Disinfectants are used to destroy microorganisms on inanimate objects such as work surfaces. An antiseptic thus has to be more selective in its action.
Table 3A.2 Ideal properties of antiplaque agents
|
|
|
|
|
|
|
|
|
|
Antiseptics have several uses in dentistry which include disinfecting surfaces for cross-infection control, skin preparations prior to surgery and in the control of bacterial plaque. This section will focus on skin preparations and antiseptic mouthwashes.
Skin preparations
A variety of antiseptics are used to cleanse the skin prior to puncture wounds or surgical incisions. Preparations include alcohol, chlorhexidine, cetrimide, povidone-iodine and triclosan. Hydrogen peroxide and potassium permanganate solutions are also used for wound debridement. Some patients may be allergic to chlorhexidine or povidone-iodine, and this should be identified so that use can be avoided.
Antiseptic mouthwashes (antiplaque agents, plaque-inhibitory agents)
The extensive use of antiseptics in dentistry is in the control of bacterial plaque and subsequent effects on caries and periodontal disease. The ideal properties of an antiplaque agent are shown in Table 3A.2. A large number of agents have been used as antiseptic mouthwashes and these are classified in Table 3A.3.
The most widely used mouthwashes contain as their active ingredient either chlorhexidine, phenols or quaternary ammonium compounds (QACs). Only these agents will be considered in this section.
Chlorhexidine
Chlorhexidine is a cationic chlorophenyl bisbiguanide with outstanding bacteriostatic properties. It remains the number one antiplaque agent. The main use of chlorhexidine is as a mouthrinse, and several proprietary brands are now available to the public. The concentration of chlorhexidine varies from mouthwash to mouthwash, but the usual levels range from 0.12 to 0.2% w/v. Chlorhexidine is also incorporated into a dental gel (0.5–1% w/v), varnish (40%) and a local delivery device (a gelatin chip – Periochip®) for placement into periodontal pockets.
Table 3A.3 Classification of antiplaque agents
| Cationic surfactants |
|
|
|
|
| Phenolic compounds |
|
|
| Herbal extracts |
|
| Heavy metal salts |
|
|
|
| Enzymes |
|
|
|
|
| Anionic surfactants |
|
|
|
|
Pharmacological properties of chlorhexidine
Chlorhexidine’s mode of action is purely topical and the bactericidal action is mediated via the bacterial cell wall. Contact with the cell wall is therefore essential, and this is facilitated by electrostatic forces between the negatively charged bacterial cell and the net positively charged chlorhexidine molecule. Having gained access to the cell membrane, chlorhexidine disorientates its lipoprotein structure destroying the osmotic barrier of the bacteria. Cell permeability increases and intracellular components such as potassium ions leak through the damaged membrane.
The short-term use of chorhexidine causes a striking reduction in the number of bacteria in saliva by 85% after only 24 hours. A maximum reduction of 95% occurs after around 5 days. Cessation of chlorhexidine mouthrinse results in a rapid return of normal salivary bacterial counts. Long-term use of chlorhexidine has been associated with increased microbial resistance and reduced sensitivity.
Other properties of chlorhexidine as an antiplaque agent
Unwanted effects of chlorhexidine
Brown staining of teeth, restorations and the oral mucosa are the main unwanted effects of chlorhexidine. Dietary factors also enhance chlorhexidine’s staining potential, especially if the mouthwash is used where there is a high intake of food/beverages rich in tannins. These include tea, coffee and red wine. Staining is also related to the concentration of chlorhexidine in the mouthwash. Chlorhexidine-induced staining requires professional removal.
Many patients find the taste of chlorhexidine unpleasant, and prolonged use blunts further the taste acuity for sweet and salt. Chlorhexidine does bind to proteins on the taste buds and blunts their activity.
Regular use of chlorhexidine is also associated with increased supragingival calculus formation. The mechanism of this unwanted effect is uncertain, but may relate to chlorhexidine’s effect on the pellicle and/or calcium ions.
Rare unwanted effects of chlorhexidine include parotid swelling and hypersensitivity reactions. Parotid swelling may be due to over vigorous rinsing resulting in negative pressure in the gland and aspiration of the mouthwash into the parotid.
The efficacy of chlorhexidine appears to be less in the presence of blood. The compound binds to various proteins found in blood and such binding renders chlorhexidine inactive. There are obvious implications with the use of chlorhexidine in the subgingival environment where the tissues are often inflamed and blood may be present. The use of chlorhexidine in these circumstances will be more beneficial once the inflammation is brought under control. If a chlorhexidine chip is being used to supplement root surface instrumentation, then the chip should be placed some 7 days later when the risk of bleeding is significantly reduced.
Phenols
These are a group of antiseptic compounds that have been used in medicine for over 100 years. Preparations of phenols and their derivatives have widespread application as disinfectants and antiseptics. Most phenols exert a non-specific antibacterial action, which is dependent on the ability of the non-ionised form of the drug to penetrate the lipid component of the cell walls of Gram-negative organisms. The resulting structural damage affects the permeability control of the bacteria. Phenolic compounds also exhibit anti-inflammatory properties. This may result from their ability to inhibit neutrophil chemotaxis, neutrophil superoxide ion generation and prostaglandin synthetase production. All of these functions are important in the inflammatory response. Phenolic compounds are found in the proprietary mouthwash Listerine®. Triclosan is a further phenolic compound that is incorporated into mouthwashes and also toothpastes.
Quaternary ammonium compounds (QACs)
QACs are also cationic antiseptics and surface-acting agents. Thus the molecules have a net positive charge and react with the negatively charged bacterial cell wall. QACs tend to be more effective against Gram-positive than Gram-negative microorganisms. This may suggest that these agents would be more beneficial as antiplaque agents when used against early developing bacterial plaque. Examples of QACs include benzethonium chloride, benzalkonium chloride and cetylpyridinium chloride. QACs are mainly used as pre-brushing rinses. Despite many claims, the efficacy of such rinses is questionable.
3B Principles of infection and infection control, diagnosis and treatment of bacterial infections
Introduction
There are three main stages involved in establishing a diagnosis: history, examination and special investigations. Certain simple infections may be diagnosed clinically and managed empirically, but microbiological studies may be helpful in some infections and essential in others. In order to select the most appropriate management for a clinical infection, it is important to understand the nature of the disease and the pathogenic organism involved. If antimicrobial agents are indicated for treatment of an infection, then the microbiology laboratory can play a pivotal role in helping with their selection. Antimicrobial resistance, infections with multiresistant isolates and antibiotic-associated complications such as Clostridium difficile-associated diarrhoea are a developing problem and a major cause of public concern. The principles of judicious prescribing are therefore of fundamental importance to safe clinical practice.
When deciding on the optimal management for a particular condition, healthcare professionals need to be able to balance the potential benefits of antimicrobial agents against the potential risks to the individual and to public health from unnecessary prescribing. This section will concentrate on the role of the microbiology laboratory in aiding the diagnosis and treatment of bacterial infections.
Use of the laboratory in the diagnosis of bacterial infection
The main goals of a diagnostic microbiology laboratory are to provide guidance to clinicians about the prevention, diagnosis, management and control of infection. In order to do this, the laboratory must be able to identify the presence of potentially pathogenic organisms and provide guidance on appropriate therapy. Reliable identification of an organism is important for a number of reasons. It may help to confirm a clinical diagnosis, give some indication of the likely prognosis and guide appropriate management. Accurate identification is also essential for epidemiological purposes, including the recognition and control of outbreaks of infection.
Taxonomy is the systematic classification, naming and identification of living organisms. Systematic naming of an individual organism involves its classification into successively smaller groups starting with a specific Kingdom, followed by a Division, Family, Genus and finally the Species, e.g. Staphylococcus aureus (Table 3B.1). In practice, this identification may be made at a molecular level using an organism’s genetic make-up (genotype) or by its physical characteristics, i.e. the way in which it physically expresses its genes (phenotype). Although molecular techniques are becoming more widely available in routine microbiology, many of the standard methods still rely on recognising the phenotypic characteristics of organisms.
Bacteria belong to the kingdom Prokaryota. They are simple, unicellular microorganisms, usually <5 micrometres (µm) in size. They form a major part of the normal human flora, but may also act as pathogens or opportunistic pathogens under certain circumstances. The routine diagnostic laboratory therefore has to be able to identify the presence of potential bacterial pathogens, often from within mixtures of normal flora (commensal organisms). To do this, the laboratory will employ a number of different techniques, capitalising on the different phenotypic or genotypic characteristics of the organisms that are sought. Important phenotypic features of bacteria include their shape, size and appearance, staining characteristics, nutritional requirements, growth rate, enzymes and antigenic determinants.
Table 3B.1 Classification of Staphylococcus aureus
| Kingdom | Prokaryota |
| Division | Eubacteria |
| Family | Micrococcaceae |
| Genus | Staphylococcus |
| Species | Staphylococcus aureus |
Diagnostic microbiological techniques can be broadly classified into microscopy, culture, and non-cultural methods including molecular techniques and serology.
Microscopy
The discovery of optics in the 1500s paved the way for the visualisation of bacteria in the following century. Anton von Loewenhoek, a Dutch scientist, examined a variety of materials using a simple microscope based on a design by Robert Hooke. Studying the microscopic appearance of dental plaque, Loewenhoek described small motile particles, which he called ‘animalcules’. These later proved to be bacteria. Microscopy was first adopted in pathology laboratories in the 1800s and has remained a fundamental diagnostic method in microbiology ever since. There are now much more sophisticated techniques available.
Light microscopy
Specimens are commonly examined by light microscopy; they may be examined directly, or stained preparations may first be made from the samples. Liquid specimens such as urine samples and specimens of cerebrospinal fluid (CSF) may be examined directly for the presence of inflammatory cells. For example, in CSF samples, the number and type of white blood cells are used as important diagnostic pointers. In cases of bacterial meninigitis, such as meningococcal meningitis, the inflammatory cells are predominantly neutrophils, while in viral meningitis, such as enteroviral meningitis, the majority of the leukocytes present in the CSF will be lymphocytes. The ratio of the number of white blood cells to the number of red blood cells present is also important, as sometimes CSF samples may be contaminated by blood during collection and, unless the red blood cells are also enumerated, this might be mistaken for a CSF containing an elevated white cell count. A ratio of >1 : 500 white blood cells to red blood cells is suggestive of infection or inflammation. Direct microscopy is also used in the diagnosis of parasitic infections: emulsified faecal samples can be examined for the presence of the ova, cysts or trophozoites of intestinal parasites such as Giardia lamblia or Ascaris lumbricoides.
Phase contrast microscopy
Phase contrast microscopes may be used to improve the visualisation of transparent microorganisms suspended in fluids. They are set up so that particles present in the fluid examined scatter the transmitted light, which becomes out of phase and thereby interferes with the light waves that have simply passed through the clear fluid. This makes the microorganisms or particles and their structures stand out in contrast to their fluid background. Phase contrast microscopy can be used to perform white and red blood cell counts on urine samples.
Dark ground microscopy
Certain very fine bacteria may be more readily visualised when brightly lit against a dark background, using a dark field microscope. For example, this method may be used to provide a rapid presumptive diagnosis of syphilis. A freshly collected swab taken from a lesion of suspected primary or secondary syphilis, when smeared on a slide and examined by dark ground microscopy, may reveal the presence of fine, motile spirochaetes characteristic of Treponema pallidum. If oral lesions are suspected to be syphilitic, however, direct microscopy should not be used as it may give misleading results because of the presence of other spirochaetes in the mouth as part of the normal flora.
Fluorescent microscopy
The detection of certain microorganisms can be enhanced by staining samples directly with fluorescent dyes such as acridine orange, auramine or fluorescein, or with specific antibodies labelled with fluorescent markers. These stains or fluorochromes transform ultraviolet (UV) light into visible light. Using this technique, target organisms such as Mycobacterium species or Legionella species can be seen glowing against a dark background when stained specimens are examined with a fluorescent microscope.
Electron microscopy
Developed in the 1950s, the increased resolution available with the electron microscope allowed the visualisation of viruses. This technique, however, is not used in the routine detection or identification of bacteria and so it will not be discussed further here.
Staining techniques
In order to study the shape or morphology of bacteria they may be examined directly under a microscope, but examination of stained preparations of bacteria provides additional information.
Gram stain
A number of different staining methods may be used in microbiology, but the Gram stain is by far the most commonly used. It is quick and simple to perform (Table 3B.2) and is a fundamental test in the identification of bacteria. Developed in 1882 by Hans Christian Gram, a Danish bacteriologist, this technique has stood the test of time because Gram inadvertently discovered a stain which is able to distinguish between two important groups of bacteria, the Gram-positive and the Gram-negative. The principal difference between these two groups of bacteria lies in the structure of their cell walls. Gram-positive bacterial cell walls are composed of two layers, the inner cytoplasmic membrane and a thick outer layer of peptidoglycan-containing proteins, teichoic acids and lipoteichoic acids. Gram-negative cell walls, however, are made of three layers; the cytoplasmic membrane is covered by a thin layer of peptidoglycan, which in turn is surrounded by a thick fatty layer of lipopolysaccharide. In the first stages of the staining process, crystal violet and Gram’s iodine bind to peptidoglycan resulting in a blue-black colouration. The addition of acetone or ethanol dissolves the lipid in the lipopolysaccharide of Gram-negative cell walls, thereby allowing the crystal violet–Gram’s iodine to leach out and the pink carbol fuchsin or safranin counterstain to enter and stain the cell wall. Acetone or ethanol, however, have a dehydrating effect on Gram-positive cells walls, fixing the blue-black crystal violet–Gram’s iodine mixture in the thick layer of peptidoglycan and preventing the uptake of the pink counterstain. Hence using the Gram stain, bacteria can be divided into Gram-positive bacteria, which are stained blue-black, and Gram-negative bacteria that look pink or red (Table 3B.3). There are also some bacteria that fail to take up the Gram stain, including mycobacteria and mycoplasma species. It should be noted that yeasts will also take up the Gram stain, appearing blue-black, although, being fungi, they are much larger and their cell walls have a different structure from that of Gram-positive bacteria.
Table 3B.2 Gram stain method
|
1. Spread aliquot of sample thinly on clean glass slide. Heat-fix
|
|
2. Stain with crystal violet for 1 min. Rinse with sterile water
|
|
3. Decolourise with acetone or ethanol for a few seconds. Rinse with sterile water
|
|
4. Counterstain with carbol fuchsin or safranin for 1 min. Rinse with sterile water
|
|
5. Allow to dry and examine under a light microscope
|
Table 3B.3 Interpretation of Gram stain
| Gram-positive bacteria | Gram-negative bacteria | |
| Amount of peptidoglycan in cell wall | + + + + | + |
| Decolourisation with acetone or ethanol | Resisted | Easy |
| Final colour of Gram stain | Blue-black | Pink-red |
The Gram stain can be applied directly to clinical samples to detect the presence of microorganisms. Examples include the examination of spun deposits of CSF in cases of possible meningitis, or specimens of synovial fluid if septic arthritis is suspected. Gram staining may also be used as a preliminary test in the identification of bacteria isolated from clinical samples.
The Ziehl–Neelsen (ZN) stain is principally used to detect mycobacteria, including Mycobacterium tuberculosis, the cause of tuberculosis (TB). Mycobacteria cannot be stained using the Gram stain because of the high concentration of mycolic acids that make up their waxy cell walls. ZN staining involves the application of hot carbol fuchsin which stains bacteria pink, followed by the addition of 20% sulfuric acid, which decolourises bacteria apart from mycobacteria. This ability to retain the carbol fuchsin stain despite treatment with acid explains why mycobacteria are sometimes known colloquially as ‘acid-fast bacilli’. The final stage of the ZN stain is the addition of a counterstain, either methylene blue or malachite green, to reveal bacteria other than mycobacteria. On examination under a light microscope, therefore, mycobacteria will appear pink while other bacteria will look blue or green depending on the counterstain employed. ZN staining of a sputum sample can lead to a rapid presumptive diagnosis of TB in a patient with suspicious clinical symptoms and signs. This is important because Mycobacterium tuberculosis is a chronic and serious respiratory infection that is readily transmitted by the respiratory route. Early treatment of infectious cases helps to minimise the onward spread of this disease. M. tuberculosis grows slowly, however, taking a minimum of 2 weeks even using rapid culture techniques, and so rapid non-cultural methods including ZN staining are used to support a clinical diagnosis and allow the early institution of appropriate therapy.
Other stains may be employed for specific purposes within the laboratory. For example, Albert’s stain highlights intracytoplasmic volutin granules and is used in the diagnosis of Corynebacterium diphtheriae (the cause of the life-threatening infection diphtheria). Auramine is a fluorescent stain used for screening sputum samples for the presence of Mycobacterium tuberculosis.
The stains described above are all examples of positive stains where the organism that is sought takes up the stain. Negative staining is a different technique, where the stain is not taken up by the microorganism. Instead, the background is stained so that the organism stands out in relief. This technique is used to aid the diagnosis of cryptococcal meningitis. This is an opportunistic fungal infection, caused by Cryptococcus neoformans, which occurs in immunocompromised patients including pregnant women, patients infected with human immunodeficiency virus (HIV) and solid organ transplant recipients. India ink is added to a spun deposit of CSF, staining the background black and highlighting the very thick capsule that is characteristic of this yeast.
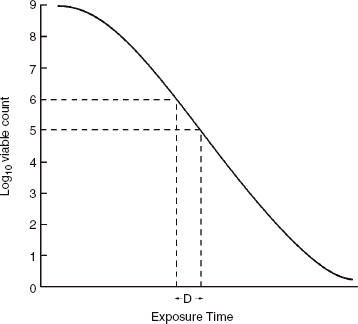
Bacterial morphology
In addition to the Gram reaction, bacteria may also be identified by their shape and the arrangement of their cells. The Gram stain is therefore used in the laboratory as one of the first steps in the identification of microorganisms cultured from clinical samples. Bacteria may be spherical (cocci), rod shaped (bacilli), comma shaped (vibrios) or spiral (spirochaetes) (see Fig. 3B.1). Cocci may be seen in characteristic arrangements. When they appear in pairs they are known as diplococci. The presence of Gram-negative diplococci in the Gram stain of a sample of CSF would suggest the diagnosis of meningococcal meningitis caused by the bacterium Neisseria meningitidis. The typical appearance of staphylococci is of Gram-positive cocci in clusters, while streptococci and enterococci are characteristically described as Gram-positive cocci in chains.
Figure 3B.1 Bacterial morphology.
Culture and identification
Most medically important bacteria can be cultured in the routine diagnostic laboratory. Some have only basic nutritional requirements, while others are fastidious, and will only grow on media containing a complex cocktail of supplements. For example, Staphylococcus aureus can be isolated on nutrient agar, a very simple medium. The growth of Haemophilus influenzae, however, is enhanced by using chocolate agar, so-called because of its rich brown colour. This is a very nutritious medium, made by heating blood agar to lyse the red blood cells and release additional growth factors.
There is now a wide variety of culture media available for different purposes. Culture media may be solid or liquid. Selective media contain inhibitors that prevent the growth of commensal organisms and thereby increase the detection of potential pathogens from within mixed cultures, while enrichment media generally encourage the growth of bacteria. Enrichment culture is useful for the detection of fastidious organisms, low numbers of bacteria or bacteria that have been inhibited by prior exposure to antibiotics. Indicator media, including chromogenic broths and agars, are now available. These contain specific enzyme substrates and are designed to change colour when the target organism is present. Laboratories will select appropriate combinations of media for the examination of different types of clinical specimen or the detection of specific pathogens.
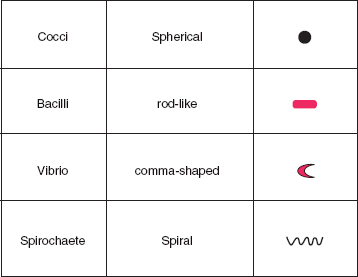
The optimal incubation requirements, both atmospheric and temperature, may be used to help identify bacteria. Organisms may be strict aerobes, only growing in the presence of oxygen, strict anaerobes, which will only grow in the absence of oxygen, or facultative anaerobes (also sometimes known as facultative aerobes) that can tolerate both conditions. Some bacteria are known as microaerophilic organisms, and these need to be incubated in an environment with a lower concentration of oxygen than is found in air. One example of a microaerophile is Helicobacter pylori, a spirochaete that is found in the stomach and duodenum and is associated with ulcer disease. Some bacteria also require the atmosphere to be enriched with additional carbon dioxide.
The majority of human pathogens will grow best at 37°C; however, some, such as Campylobacter jejuni, are able to tolerate higher temperatures. Others can multiply in colder conditions. For example, listeriosis, a serious systemic infection affecting pregnant women, foetuses and immunocompromised patients is caused by Listeria monocytogenes, a Gram-positive rod which is able to grow on contaminated foods such as soft cheeses and patés in the refrigerator at 4°C. The growth rate is also a useful feature when identifying bacteria. Many common organisms such as staphylococci, streptococci, Escherichia coli and Salmonella require 18–24 hours (overnight) incubation, while some take longer. For example, Pseudomonas aeruginosa and Bacteroides spp. often take 2 days, Legionella spp. take up to 10 days and Actinomyces israelii may take up to 2–4 weeks. At the far extreme, mycobacteria may take more than 6 weeks to grow using conventional solid media such as Lowenstein–Jensen agar, although more rapid liquid culture methods can now shorten this period to 2 weeks.
The macroscopic appearance of bacterial colonies growing on agar in the laboratory may also provide clues as to the identity of the organism. They may have a typical colour, size, shape or consistency. Some bacteria, including Proteus and Clostridia spp., can swarm over the surface of agar plates. Bacteria may be haemolytic if they are able to produce enzymes that lyse red blood cells. This feature may be seen when the bacteria are cultured on blood agar, and it is often enhanced by growth in an anaerobic environment. Streptococci are classified according to their haemolytic reactions. β-Haemolytic streptococci, including Streptococcus pyogenes (also known as β-haemolytic streptococcus Group A), an important cause of pharyngitis and cellulitis, produces a haemolysin that completely lyses red blood cells, resulting in a zone of clearing in the agar around the colonies. α-Haemolytic streptococci, including the viridans streptococci found in the mouth and Streptococcus pneumoniae, are only able partially to haemolyse red blood cells in blood agar, and this causes greening of the media. Streptococci that are unable to haemolyse blood agar are known as non-haemolytic streptococci. Some bacteria produce a typical odour, although care must be taken to prevent inhalation of aerosols of the organism when examining this characteristic.
Examination of basic phenotypic characteristics will usually provide a presumptive bacterial identification. For example, Table 3B.4 outlines a simple classification scheme for bacteria using their Gram-stained appearance and atmospheric requirements. Formal identification or speciation of bacteria, however, may require analysis of a collection of phenotypic characteristics. So, in practice, presumptive identifications are often confirmed by tests for additional features including biochemical characteristics such as the pattern of carbohydrate fermentation or the presence of enzymes such as urease or catalase, the presence of antigenic determinants or special structures such as spores or flagella. Manual and automated commercial tests are available for the identification of different types of bacteria.
Bacterial typing
There are occasions when identification beyond the species level is important. This is used to help determine whether organisms are likely to be the same strain, i.e. whether they are likely to have arisen from the same original source. This is helpful for epidemiological investigations, e.g. into possible outbreaks of infection, or to determine whether an individual patient is experiencing a relapsing infection with the same organism or a new infection with the same species of bacterium.
Table 3B.4 Simple bacterial classification scheme
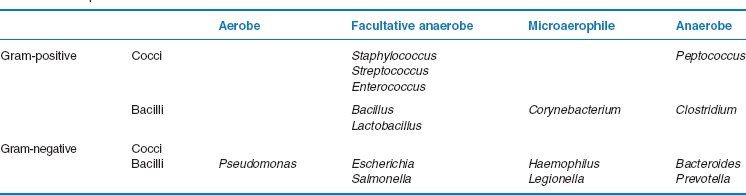
There are a variety of phenotypic and genotypic typing schemes available, based on different bacterial characteristics. Examples include simple tests such as the examination of the antimicrobial susceptibility pattern (antibiogram), determining different phenotypic or antigenic characteristics between bacteria from the same species (biotyping or serotyping, respectively) or examination of the proteins present in the organism (polyacrylamide gel electrophoresis (PAGE)). Other typing methods involve examination of the genetic make-up of the cell by molecular techniques. These are discussed later. They include restriction enzyme analysis, restriction fragment length polymorphism (RFLP), pulsed-field gel electrophoresis, DNA hybridisation studies and genetic sequencing. The latter provides the most definitive identification, but currently la/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


