Respiratory medicine
Key Points
• Upper respiratory infections are commonplace, especially in young people, and are often contagious
• Lower respiratory infections are often contagious and some are potentially fatal
• Asthma is common and may be life-threatening
• Chronic obstructive pulmonary disease is common and disabling
The respiratory tract consists of the upper respiratory tract (URT – nose, paranasal sinuses, pharynx and larynx; discussed in Ch. 14) and the lower respiratory tract (LRT): the respiratory airways (trachea, bronchi and bronchioles) and lungs (respiratory bronchioles, alveolar ducts, alveolar sacs and alveoli), discussed in this chapter.
Protective mechanisms in the respiratory tracts include a mucociliary lining. Particles or pathogens are trapped in the mucus and driven by ciliary action (the ciliary elevator) to the pharynx. Mucociliary transport declines with age but any effect on clinical infection has not been proved. Lymphoid tissues of the Waldeyer ring (adenoids, palatine and lingual tonsils) are important in developing an immune response to pathogens. However, the best respiratory defence mechanism is the cough reflex, the components of which include cough receptors, afferent nerves, the cough centre, and efferent nerves and effector muscles. Impairment of any of these – as may be seen in older patients or those with conditions associated with lowered consciousness (e.g. sedative use and neurological disease) – can weaken protection. Dysphagia or impaired oesophageal motility may exacerbate the tendency to aspirate foreign material. The alveolar defence mechanisms include macrophages, immunocytes, surfactant, phospholipids, immunoglobulin G (IgG), IgE, secretory IgA, complement components and factor B; many immune defects manifest with recurrent respiratory infections.
Lung function is vital to gas exchange – the blood absorbs oxygen and releases carbon dioxide. Normal gas exchange requires adequate alveolar ventilation, normal ventilation/blood flow relationships and adequate alveolar–capillary membrane surface area. Breathing (ventilation) depends on respiratory drive, which reacts to the respiratory load. This process requires work and results in gas exchange.
Oxygen is transported in combination with haemoglobin in erythrocytes and a small amount dissolved in plasma. The oxyhaemoglobin dissociation curve is sigmoidal; once the oxygen saturation falls below 95%, the amount of O2 transported to the tissues and brain falls rapidly. High temperatures, acidosis, raised CO2 and raised 2,3- diphosphoglycerate (2,3-DPG) levels encourage oxygen offloading, whereas fetal haemoglobin and carboxyhaemoglobin have the contrary effect. Chronic hypoxaemia (e.g. at high altitudes) stimulates release of erythropoietin from the kidneys, with a rise in red cell production, and raised 2,3-DPG. Athletes have abused erythropoietin to gain competitive advantage (Ch. 33).
Lower Respiratory Disease
The most common LRT disorders are asthma and chronic obstructive pulmonary disease (COPD).
General aspects
Respiratory disorders are common, and are often caused or aggravated by tobacco smoking. They may significantly affect general anaesthesia (GA) and conscious sedation (CS), since they are often a contraindication to use of benzodiazepines, opioids, GA agents and other respiratory depressants.
Clinical features
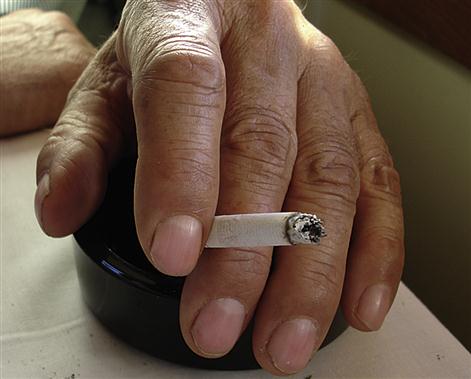
Impaired gas exchange leads to laboured breathing and can cause significant incapacity. Features include cough, sputum production, wheeze, dyspnoea, chest pain, cyanosis, finger-clubbing (Fig. 15.1), use of accessory muscles of respiration with indrawing of the intercostal spaces (hyperinflation), and abnormalities in chest shape, movements, respiratory rate and breath sounds.
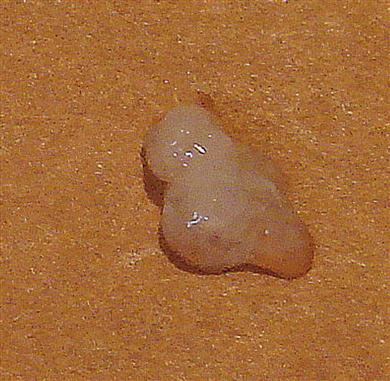
Cough may be a feature of any respiratory problem but, if chronic, may herald serious disease – for example, COPD, cancer or infection such as tuberculosis. Mucoid or mucopurulent sputum is often a feature (Fig. 15.2); purulent sputum indicates acute bronchitis, bronchiectasis or lung abscess. Blood (haemoptysis) or blood-stained sputum, though common in acute infections (especially in pre-existing COPD), bronchiectasis and pulmonary embolism, may herald an even more serious condition – for example, possibly one due to carcinoma or tuberculosis. Wheezing is caused by airways obstruction and is a typical sign of asthma or COPD. Breathlessness (dyspnoea) is distressing, and may be caused by respiratory or cardiovascular disease, or by anaemia, and is particularly ominous if it persists at rest.
Excessive resistive load, such as in asthma, COPD and cystic fibrosis, impairs airflow. Elastic load increases because of, for example, interstitial fibrosis, muscle paralysis and obesity.
General management
Diagnosis of respiratory disorders is from the clinical features supported by imaging (especially chest radiography). Spiral computed tomography (CT) can now scan the lungs in a quick 20–30-second breath-hold and therefore, instead of producing a stack of individual CT slices, which may be misaligned due to patient movement or breathing in between slices, provides high-resolution three-dimensional images.
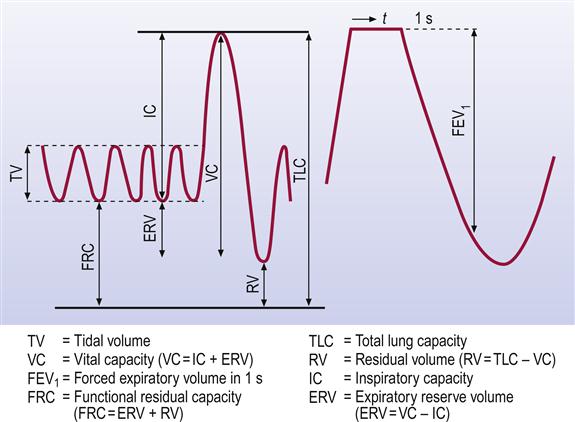
Respiratory function tests can measure individual components of the respiratory process. Spirometry is the basic screening test for assessing mechanical load problems, the quantification involving determination of the vital capacity (VC) – slow vital capacity (SVC) and/or forced vital capacity (FVC) – and the speed of maximal expiratory flow (MEF; Fig. 15.3). In health, about 75% of a normal-sized VC is expelled in 1 second (FEV1). The peak flow meter, which measures the peak expiratory flow rate (PEFR; the earliest portion of forced expiration), is a simple measure of airflow obstruction, when the FEV1 is a much smaller fraction of the VC. In lung restriction, the diminished VC can be mostly expelled in about 1 second. Serial measurements (e.g. in asthma) provide valuable information about disease progress. The reversibility of airways obstruction is usually assessed by spirometry before and after use of a bronchodilator agent.
Arterial blood gas analysis yields considerable information about gas exchange efficiency. Arterial hypoxaemia in adults is defined as PaO2 below 10.7 kPa breathing room air, although it is not usually treated as clinically important unless below 8 kPa, when oxygen saturation will be 90% or less (Table 15.1).
Table 15.1
Adult values for PaO2 and oxygen saturationa
| PaO2 (kPa) | SaO2 (%) | |
| Normal (range) | 13 (≥10.7) | 97 (95–100) |
| Hypoxaemia | <10.7 | <95 |
| Mild hypoxaemia | 8–10.5 | 90–94 |
| Moderate hypoxaemia | 5.3–7.9 | 75–89 |
| Severe hypoxaemia | <5.3 | <75 |
aFrom Williams AJ. Assessing and interpreting arterial blood gases and acid–base balance. BMJ 1998; 317:1213. http://www.bmj.com/cgi/content/full/317/7167/1213 (accessed 30 September 2013).
Arterial carbon dioxide tension (PaCO2) is used as an inversely proportional index of ‘effective’ alveolar ventilation. Hence, a high PaCO2 is taken to indicate poor alveolar ventilation. Alveolar hypoventilation (raised PaCO2) with a normal pH probably represents a primary ventilatory change present long enough for renal mechanisms to compensate, as in chronic ventilatory failure. Ventilation/blood flow relationships are most simply assessed by considering the size of the difference between the amounts of oxygen and carbon dioxide in the blood and in the air; the differences are small if the lungs are working efficiently. Disparity between ventilation/blood flow ratios results in abnormally wide differences – and then alveolar–arterial PO2 and arterial–alveolar PCO2 gradients will be abnormal.
Alveolar capillary surface area is assessed by measuring the uptake of carbon monoxide – usually abnormal in diffuse interstitial inflammatory and fibrotic processes and in emphysema.
Assessing bronchial reactivity and the exercise response can help evaluate breathlessness. Simple exercise testing provides information about overall fitness and the appropriateness of cardiorespiratory responses. Radionuclide lung scanning, blood gas analysis and sputum cytology or culture are sometimes needed in addition.
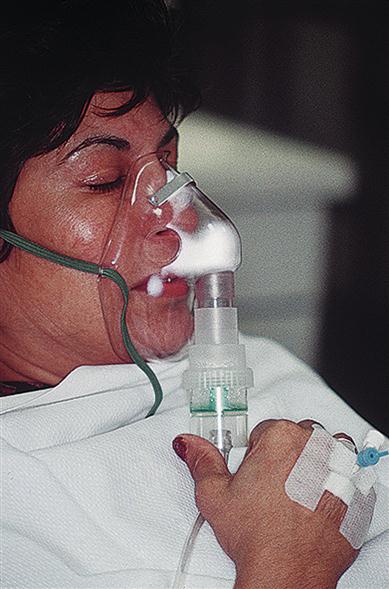
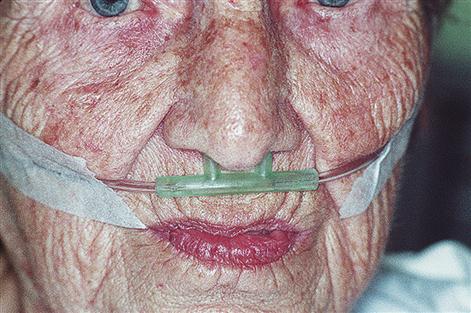
Management can include oxygen administration by mask or nasal cannula (Figs 15.4 and 15.5).
Dental aspects
LRT disorders can cause significant incapacity and are often a contraindication to GA, and even to CS.
Asthma
General aspects
Asthma is common, affecting 2–5% of the overall population; it is on the increase, particularly in childhood, with a frequency of up to 20% in some high-income countries. Asthma usually begins in childhood or early adult life; about half the patients with asthma develop it before age 10 years.
Bronchial hyper-reactivity causes reversible airway obstruction from smooth muscle constriction (bronchospasm), mucosal oedema and mucus hypersecretion. There are two main types, extrinsic (allergic) and intrinsic asthma (Table 15.2).
Table 15.2
| Extrinsic asthma | Intrinsic asthma | |
| Frequency | Most common | Least common |
| Aetiopathogenesis | Allergens causing IgE-mediated mast cell degranulation | Mast cell instability and airway hyper-responsivity |
| Association with atopy | + | − |
| Typical age of onset | Child | Adult |
Extrinsic (allergic) asthma, the main childhood type, may be precipitated by allergens in animal dander, feathers or hair, drugs (e.g. non-steroidal anti-inflammatory drugs [NSAIDs] and some antibiotics), food (e.g. eggs, fish, fruit, milk, nuts), house dust (mite allergens) or moulds. Patients frequently have or develop other allergic diseases, such as eczema, hay fever and drug sensitivities. Extrinsic asthma is associated with IgE overproduction on allergen exposure, and release of mast cell mediators (histamine, leukotrienes, prostaglandins, bradykinin and platelet activating factor), which cause bronchospasm and oedema. About 75% of asthmatic children lose their asthma or improve by adulthood.
Intrinsic asthma is usually of adult onset and not allergic, but appears rather to be related to mast cell instability and airway hyper-responsivity. Triggers include emotional stress, gastro- oesophageal reflux or vagally mediated responses.
Either type of asthma can be triggered by: infections (especially viral, mycoplasmal or fungal); irritating fumes (e.g. traffic or cigarette smoke); exercise (possibly due to cold air); weather changes; emotional stress; foods (e.g. nuts, shellfish, strawberries or milk) or additives (such as tartrazine); and drugs (e.g. aspirin and other NSAIDs, beta-blockers and angiotensin-converting enzyme inhibitors [ACEIs]).
Clinical features
In well-controlled patients with asthma, clinical features may be absent. During an asthmatic episode, symptoms may include dyspnoea, cough and paroxysmal expiratory wheeziness with laboured expiration. The frequency and severity of attacks vary widely between individuals (Table 15.3). Patients may become distressed, anxious and tachycardic, have reduced chest expansion and be using accessory respiratory muscles to increase their ventilatory effort. Nasal polyps are common, especially in aspirin-sensitive asthmatics. Children with asthma initially suffer from repeated ‘colds’ with cough, malaise and fever, often at night.
Table 15.3
< ?comst?>
| Severity of asthma | Symptom duration | Attack frequency per week | Other comments | Typical therapy |
| Mild | <1 h | <2 | Attacks follow exercise or exposure to trigger | Beta-agonist as required |
| Moderate | Days | >2 | Activity restricted | Beta-agonist plus steroid |
| Severe | Persistent | Persistent | Audible wheezing. Tachypnoea. Activity and sleep severely restricted | Beta agonist plus steroid plus theophylline |
< ?comen?>< ?comst1?>

< ?comst1?>
< ?comen1?>
Asthma is typically diagnosed when the patient has more than one of the following – wheeze, cough, difficulty breathing and chest tightness – particularly if these are frequent and recurrent; are worse at night and in the early morning; occur in response to, or are worse after, exercise or other triggers, such as exposure to pets, cold or damp air, or with emotions or laughter; or occur without an association with colds. There is often:
■ a personal history of atopic disorder
■ a family history of atopic disorder and/or asthma
■ widespread wheeze, heard on chest auscultation
■ a history of improvement in symptoms or lung function in response to adequate therapy.
A prolonged asthmatic attack, which is refractory to treatment, may lead to life-threatening status asthmaticus (persisting for more than 24 hours). Failure of the patient to complete a sentence, indrawing of the intercostal muscles, a rapid pulse, a silent chest and signs of exhaustion are suggestive of impending respiratory arrest.
General management
Diagnosis of asthma is from the clinical history and presentation, based on recognizing a characteristic pattern of episodic symptoms in the absence of an alternative explanation. Investigations include a chest radiograph (to exclude other diagnoses, such as a pneumothorax), spirometry (serial PEFR), skin tests and blood examination (usually eosinophilia, raised total IgE and specific IgE antibody concentrations, which may help identify allergens). Occasionally, a histamine or methacholine challenge is used if the diagnosis is unclear.
In children with an intermediate probability of asthma, who can perform spirometry and have evidence of airways obstruction, assess the change in FEV1 or PEFR in response to an inhaled bronchodilator (reversibility) and/or the response to a trial of treatment for a specified period; if there is significant reversibility, or if a treatment trial is beneficial, a diagnosis of asthma is probable.
Management includes patient education, smoking cessation advice, avoidance of identifiable irritants and allergens, and use of drugs. Home use of peak flow meters allows patients to monitor progress and detect any deterioration that may require urgent modification of treatment. Treatment should be based on the amount by which peak flow is reduced (a PEFR diary should be kept).
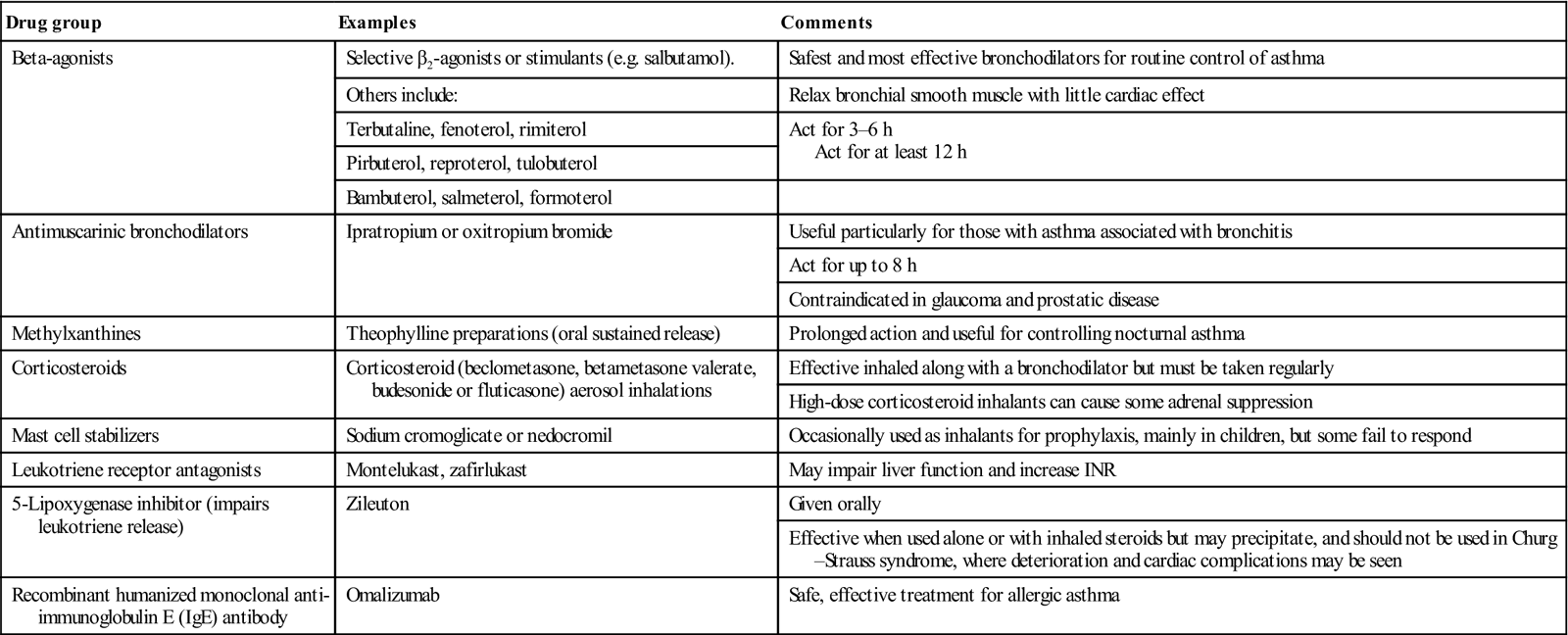
Drugs used for asthma management (Table 15.4) include oxygen, short-acting β2 agonists (SABAs; such as salbutamol), corticosteroids, leukotriene receptor antagonists and omalizumab (a recombinant humanized monoclonal anti-IgE antibody that reduces the antigen-specific IgE).
Table 15.4
< ?comst?>
| Drug group | Examples | Comments |
| Beta-agonists | Selective β2-agonists or stimulants (e.g. salbutamol). | Safest and most effective bronchodilators for routine control of asthma |
| Others include: | Relax bronchial smooth muscle with little cardiac effect | |
| Terbutaline, fenoterol, rimiterol | Act for 3–6 h Act for at least 12 h |
|
| Pirbuterol, reproterol, tulobuterol | ||
| Bambuterol, salmeterol, formoterol | ||
| Antimuscarinic bronchodilators | Ipratropium or oxitropium bromide | Useful particularly for those with asthma associated with bronchitis |
| Act for up to 8 h | ||
| Contraindicated in glaucoma and prostatic disease | ||
| Methylxanthines | Theophylline preparations (oral sustained release) | Prolonged action and useful for controlling nocturnal asthma |
| Corticosteroids | Corticosteroid (beclometasone, betametasone valerate, budesonide or fluticasone) aerosol inhalations | Effective inhaled along with a bronchodilator but must be taken regularly |
| High-dose corticosteroid inhalants can cause some adrenal suppression | ||
| Mast cell stabilizers | Sodium cromoglicate or nedocromil | Occasionally used as inhalants for prophylaxis, mainly in children, but some fail to respond |
| Leukotriene receptor antagonists | Montelukast, zafirlukast | May impair liver function and increase INR |
| 5-Lipoxygenase inhibitor (impairs leukotriene release) | Zileuton | Given orally |
| Effective when used alone or with inhaled steroids but may precipitate, and should not be used in Churg–Strauss syndrome, where deterioration and cardiac complications may be seen | ||
| Recombinant humanized monoclonal anti-immunoglobulin E (IgE) antibody | Omalizumab | Safe, effective treatment for allergic asthma |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
Inhaled long-acting β2 agonists (LABAs) may be needed (Fig. 15.6). Deaths from asthma are usually a result of failure to recognize deterioration or reluctance to use corticosteroids.

Other factors that have been studied include:
■ air pollution – There is an association between air pollution and aggravation of existing asthma
■ allergen avoidance – There is no consistent evidence of benefit
■ breast-feeding – There is evidence of a protective effect in relation to early asthma
■ electrolytes – There is no consistent evidence of benefit
■ fish oils and fatty acid – There is no consistent evidence of benefit
■ house dust mites – Measures to reduce the numbers of house dust mites do not affect asthma severity
■ immunotherapy – Allergen-specific immunotherapy is beneficial in allergic asthma
■ modified milk formulae – There is no consistent evidence of benefit
■ pets – There are no controlled trials on the benefits of removing pets from the home
Dental aspects
Elective dental care should be deferred in severe asthmatics until they are in a better phase; this can be advised by the patient’s general practitioner.
Asthmatic patients should be asked to bring their usual medication with them when coming for dental treatment. Local anaesthesia (LA) is best used; occasional patients may react to the sulphites present as preservatives in vasoconstrictor-containing LA, so it may be better, where possible, to avoid solutions containing vasoconstrictor. Adrenaline (epinephrine) may theoretically enhance the risk of arrhythmias with beta-agonists and is contraindicated in patients using theophylline, as it may precipitate arrhythmias.
Relative analgesia with nitrous oxide and oxygen is preferable to intravenous sedation and gives more immediate control. Sedatives in general are better avoided as, in an acute asthmatic attack, even benzodiazepines can precipitate respiratory failure.
GA is best avoided, as it may be complicated by hypoxia and hypercapnia, which can cause pulmonary oedema even if cardiac function is normal, and cardiac failure if there is cardiac disease. The risk of postoperative lung collapse or pneumothorax is also increased. Halothane or, better, enflurane, isoflurane, desflurane and sevoflurane are the preferred anaesthetics, but ketamine may be useful in children.
Allergy to penicillin may be more frequent in asthmatics. Drugs to be avoided, since they may precipitate an asthmatic attack (see later), include those listed in Box 15.1.
Acute asthmatic attacks may also occasionally be precipitated by anxiety; it is important to attempt to lessen fear of dental treatment by gentle handling and reassurance. Even routine dental treatment can trigger a clinically significant decline in lung function in approximately 15% of asthmatics. Acute asthmatic attacks are usually self-limiting or respond to the patient’s usual medication, such as a beta-agonist inhaler, but status asthmaticus is a potentially fatal emergency (Ch. 1). There may be complications caused by the anti-asthmatic drugs (Table 15.5).
Table 15.5
Management complications from anti-asthmatic drugs
| Anti-asthmatic drug | Possible complications |
| β2-agonists | Dry mouth |
| Corticosteroids | Steroid complications and adrenal crisis. Corticosteroid inhalers occasionally causes oral or pharyngeal thrush and, rarely, angina bullosa haemorrhagica |
| Ipratropium bromide | Dry mouth |
| Leukotriene antagonists | Bleeding tendency (and prolonged INR) because of impaired liver metabolism |
| Theophylline | Levels increased by epinephrine, erythromycin, clindamycin, azithromycin, clarithromycin or ciprofloxacin |
Gastro-oesophageal reflux is not uncommon, with occasional tooth erosion. Periodontal inflammation is greater in asthmatics than in those without respiratory disease. Persons using steroid inhalers may develop oropharyngeal candidosis or, occasionally, angina bullosa haemorrhagica.
Guidelines on the management of asthma may be found at: http://www.sign.ac.uk/guidelines/fulltext/101/index.html, http://www.nice.org.uk/guidance/qualitystandards/indevelopment/Asthma.jsp and http://www.brit-thoracic.org.uk/Portals/0/Guidelines/AsthmaGuidelines/qrg101%202011.pdf (all accessed 30 September 2013).
Churg–Strauss Syndrome (Allergic Granulomatosis or Angiitis)
General aspects
Churg–Strauss syndrome (CSS) is a rare, potentially fatal, systemic vasculitis similar to polyarteritis nodosa (PAN), characterized by severe asthma-like attacks with peripheral eosinophilia, and intravascular and extravascular granuloma formation with eosinophil infiltration and skin lesions in 70%. Cardiopulmonary involvement is the main cause of death.
Clinical features
CSS is diagnosed if at least 4 of the 6 criteria listed in Box 15.2 are positive.
General management
The 5-year survival of untreated CSS is 25%. Combination treatment with cyclophosphamide and prednisolone (prednisone) provides a 5-year survival of 50%.
Dental aspects
Management problems relating to patients with CSS may include respiratory impairment and corticosteroid treatment (Ch. 6).
Chronic Obstructive Pulmonary Disease (COPD)
General aspects
Chronic obstructive pulmonary disease (COPD; chronic obstructive airways disease, COAD) is a common, chronic, slowly progressive, irreversible disease (most frequently a combination of chronic bronchitis and emphysema), characterized by breathlessness and wheeze (airways obstruction), cough and sputum. Chronic bronchitis is defined as the excessive production of mucus and persistent cough with sputum production, daily for more than 3 months in a year over more than 2 consecutive years. It leads to production of excessive, viscous mucus, which is ineffectively cleared from the airway, obstructs and stagnates, and becomes infected, usually with Streptococcus pneumoniae, Moraxella catarrhalis and Haemophilus influenzae. Patchy areas of alveolar collapse can result. Emphysema is dilatation of air spaces distal to the terminal bronchioles with destruction of alveoli, reducing the alveolar surface area available for respiratory exchange. COPD is now the preferred term for conditions with airflow obstruction because of a combination of airway and parenchymal damage; patients were previously diagnosed as having chronic bronchitis or emphysema.
COPD is characterized by airflow obstruction – defined as an FEV1/FVC ratio reduced to less than 0.7. If FEV1 is 80% or more, a diagnosis of COPD should only be made if there are respiratory symptoms (e.g. dyspnoea or cough). The airflow obstruction is not fully reversible, does not change significantly over months, and is usually progressive in the long term.
The most important causes of COPD include cigarette smoking, environmental pollution, dusts, chemicals or occupational exposures to various substances. Exposure to smoke from home cooking or heating fuels may contribute. Deficiency of the antiproteolytic enzyme alpha1-antitrypsin is a rare cause of emphysema.
Clinical features
There is often significant airflow obstruction before the person is aware of it and so COPD typically remains undiagnosed until patients are in their fifties. Differentiation from asthma is important (Table 15.6).
Table 15.6
Clinical features differentiating COPD and asthma
| COPD | Asthma | |
| Smoker or ex-smoker | Nearly all | Possibly |
| Symptoms under age 35 | Rare | Often |
| Chronic productive cough | Common | Uncommon |
| Breathlessness | Persistent and progressive | Variable |
| Night-time waking with breathlessness and/or wheeze | Uncommon | Common |
| Significant diurnal or day-to-day variability of symptoms | Uncommon | Common |
A diagnosis of COPD should be considered in patients over the age of 35 who have a risk factor (e.g. smoking) and exertional breathlessness, chronic cough, regular sputum production, frequent winter ‘bronchitis’ or wheeze. Clinical judgment is based on history, physical examination, confirmation of airflow obstruction using spirometry (post-bronchodilator spirometry) and assessment of the severity of dyspnoea (Tables 15.7 and 15.8).
Table 15.7
Additional investigations in suspected COPD
| Investigation | Comments |
| Alpha1-antitrypsin | If COPD has an early onset, or there is a minimal smoking history or positive family history |
| CT scan of thorax | To investigate symptoms that seem disproportionate to spirometric impairment |
| To investigate chest radiograph abnormalities | |
| To assess suitability for GA and surgery | |
| Echocardiogram | To assess cardiac status, if cor pulmonale is present |
| Electrocardiogram | To assess cardiac status, if cor pulmonale is present |
| Peak flow measurements (serial domiciliary) | To exclude asthma, if any diagnostic doubt |
| Pulse oximetry | To assess need for oxygen therapy, if cyanosis or cor pulmonale is present, or if FEV1 is<50% predicted |
| Sputum culture | To identify organisms, if sputum is persistently present and purulent |
| Transfer factor for carbon monoxide (TLCO) | To investigate symptoms that seem disproportionate to spirometric impairment |
Table 15.8
| Grade | Degree of breathlessness related to activities |
| 1 | Not troubled by breathlessness except on strenuous exercise |
| 2 | Short of breath when hurrying or walking up a slight hill |
| 3 | Walks slower than contemporaries on level ground because of breathlessness, or has to stop for breath when walking at own pace |
| 4 | Stops for breath after walking about 100 metres or after a few minutes on level ground |
| 5 | Too breathless to leave the house, or breathless when dressing or undressing |
Adapted from Fletcher CM, et al. The significance of respiratory symptoms and the diagnosis of chronic bronchitis in a working population. Br Med J 1959; 2:257.
COPD is characterized by breathlessness and wheeze (airways obstruction), cough and an early morning mucoid sputum production. Progressive dyspnoea, low oxygen saturation, carbon dioxide accumulation (hypercapnia) and metabolic acidosis mean that patients may ultimately become dyspnoeic at rest (‘respiratory cripples’), especially when recumbent (orthopnoea), and eventually develop respiratory failure, pulmonary hypertension, right ventricular hypertrophy and right-sided heart failure (cor pulmonale).
Two clinical patterns of COPD are recognized:
■ ‘Blue bloaters’ – patients with chronic bronchitis who lose their CO2 drive, fail to maintain adequate ventilation and become both hypercapnic and hypoxic with central cyanosis, cor pulmonale and oedema (for these patients, the respiratory drive is from the low PO2 and thus oxygen administration is contraindicated) (Table 15.9).
Table 15.9
Comparison of chronic bronchitis and emphysema
| Chronic bronchitis | Emphysema | |
| Cough | Chronic | Minimal |
| Sputum | Copious and mucoid | Minimal |
| Infections | Common | Uncommon |
| Dyspnoea | Mild | Severe |
| PO2 | Low | Low |
| PCO2 | Raised | Normal |
| Forced expiratory volume (FEV) | Low | Low |
| Clinical appearance | Blue bloater | Pink puffer |
General management
The diagnosis of COPD is based upon clinical history and presentation. Investigations include a chest radiograph (which may show hyperinflated lung fields with loss of vascular markings); arterial blood gases (which should be measured if pulse oximetry shows oxygen saturation less than 92%); spirometry; and lung function tests. FEV1 is reduced in all cases (FEV1 of less than 40% signifies severe COPD) and the flow–volume curve shows a typical pattern, with reduced flow rates at mid- and lower-lung volumes. A ratio of FEV1:FVC of less than 70% confirms airways obstruction.
Patients with COPD and their family should be educated about the disease, and about required lifestyle changes and medication. Non-drug therapy includes: stopping smoking (nicotine replacement therapy or bupropion may help); exercise by pulmonary rehabilitation – of proven benefit; weight loss (improves exercise tolerance); and vaccination (pneumococcal and influenza vaccines). Drug therapy includes short-acting bronchodilators (anticholinergic drugs [ipratropium bromide]) and β2 agonists (salbutamol) to treat the reversible component of airway disease; corticosteroids (inhaled or systemic); and antibiotics (amoxicillin, trimethoprim or tetracycline). Mucolytics, such as carbocisteine, reduce acute exacerbations by almost one-third. Long-term oxygen therapy (LTOT) reduces mortality.
People with stable COPD who remain breathless or have exacerbations, despite using short-acting bronchodilators, should be offered the following as maintenance therapy:
Offer a LAMA in addition to a LABA plus ICS to people with COPD who remain breathless or have exacerbations, despite taking LABA plus ICS, irrespective of their FEV1.
Provide pulmonary rehabilitation for all who need it; non-invasive ventilation (NIV) is the treatment of choice for persistent hypercapnic ventilatory failure during exacerbations not responding to medical therapy. The frequency of exacerbations should be reduced by appropriate use of inhaled corticosteroids and bronchodilators, and vaccinations.
Inhaled therapy
Bronchodilators (short-acting β2 agonists [SABA] and short-acting muscarinic antagonists [SAMA]) should be the initial empirical treatment for the relief of breathlessness and exercise limitation. ICS have potential adverse effects (including non-fatal pneumonia) in people with COPD. Offer a once-daily LAMA in preference to four-times-daily SAMA to people with stable COPD who remain breathless or have exacerbations, despite using short-acting bronchodilators as required, and in whom a decision has been made to commence regular maintenance bronchodilator therapy with a muscarinic antagonist (see above).
Most patients – whatever their age – are able to acquire and maintain an adequate inhaler technique. Bronchodilators are usually best administered using a hand-held inhaler device (including a spacer device if appropriate).
Patients with distressing or disabling dyspnoea, despite maximal therapy using inhalers, should be considered for nebulizer therapy. They should be offered a choice between a face mask and a mouthpiece to administer their nebulized therapy, unless the drug specifically requires a mouthpiece (for example, anticholinergic drugs).
Oral therapy
Some patients with advanced COPD may require maintenance oral corticosteroids when these cannot be withdrawn following an exacerbation. These individuals should be monitored for the development of osteoporosis and given appropriate prophylaxis.
Theophylline should only be used after a trial of SABA and LABA, and only to those who are unable to use inhaled therapy, as there is a need to monitor plasma levels and interactions. The dose of theophylline prescribed should be reduced at the time of an exacerbation if macrolide or fluoroquinolone antibiotics (or other drugs known to interact) are given. There is insufficient evidence to recommend prophylactic antibiotic therapy in the management of stable COPD. Mucolytic drug therapy should be considered in patients with a chronic cough productive of sputum.
Combined inhaled and oral therapy
If patients remain symptomatic on monotherapy, their treatment should be intensified by combining therapies from different drug classes, such as:
Oxygen
Long-term oxygen therapy (LTOT).
Inappropriate oxygen therapy in people with COPD may depress respiration. LTOT is indicated in patients with COPD who have a PaO2 of less than 7.3 kPa when stable, or a PaO2 greater than 7.3 kPa and less than 8 kPa when stable, and one of: secondary polycythaemia, nocturnal hypoxaemia (oxygen saturation of arterial blood [SaO2] of less than 90% for more than 30% of the time), peripheral oedema or pulmonary hypertension.
To reap the benefits of LTOT, patients should breathe supplemental oxygen for at least 15 hours per day. To ensure that all those eligible for LTOT are identified, pulse oximetry should be available in all health-care settings. The assessment of patients for LTOT should comprise the measurement of arterial blood gases on two occasions at least 3 weeks apart in patients who have a confident diagnosis of COPD, who are receiving optimum medical management and whose COPD is stable.
Patients should be warned about the risks of fire and explosion and told not to smoke when using oxygen.
Ambulatory oxygen therapy.
Ambulatory oxygen therapy should be considered in patients on LTOT who wish to continue oxygen therapy outside the home, and who have exercise desaturation, are shown to have an improvement in exercise capacity and/or dyspnoea with oxygen, and are motivated to use oxygen.
Non-invasive ventilation (NIV).
Adequately treated patients with chronic hypercapnic respiratory failure who have required assisted ventilation during an exacerbation, or who are hypercapnic or acidotic on LTOT, should be referred to a specialist centre for consideration of long-term NIV. Advanced emphysema is occasionally treated with surgery – excision of large acquired bullae or, rarely, lung transplantation.
Dental aspects
Patients with COPD who need dental care can be classified as follows:
Patients with COPD are best treated in an upright position at mid-morning or early afternoon, since they may become increasingly dyspnoeic if laid supine. It may be difficult to use a rubber dam, as some patients are mouth-breathers and not able to tolerate the additional obstruction. LA is preferred for dental treatment, but bilateral mandibular or palatal injections should be avoided. Patients with COPD should be given relative analgesia only if absolutely necessary, and only in hospital after full preoperative assessment. CS with diazepam and midazolam should not be used, as benzodiazepines are respiratory depressants. Patients should be given GA only if absolutely necessary, and intravenous barbiturates are contraindicated. Secretions reduce airway patency and, if lightly anaesthetized, the patient may cough and contaminate other areas of the lung.
Postoperative respiratory complications are more prevalent in patients with pre-existing lung diseases, especially after prolonged operations and if there has been no preoperative preparation. The most important single factor in preoperative care is cessation of smoking for at least 1 week preoperatively. Respiratory infections must also be eradicated; sputum should first be sent for culture and sensitivity, but antimicrobials such as amoxicillin should be started without awaiting results.
The medical management of COPD should be optimized prior to surgery. The ultimate clinical decision about whether or not to proceed with surgery should rest with a consultant anaesthetist and consultant surgeon, taking account of co-morbidities, functional status of the patient and necessity for the surgery.
Composite assessment tools, such as the American Society of Anesthesiologists (ASA) scoring system, and not just lung function, are the best criteria for the assessment of patients with COPD before surgery. Those taking corticosteroids should be />
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses