15
Orthodontic Materials
Introduction
Orthodontists and orthodontic technicians use a variety of materials, equipment, and procedures as they treat patients and construct appliances. Only a brief survey of some materials will be included in this chapter. For an excellent in depth coverage of the topic, we recommend the book written by Brantley and Eliades (2001).
Orthodontic Wires
Four basic types of wires are used by orthodontists: (1) austenitic stainless steel, (2) cobalt-chromium-nickel, (3) beta-titanium, and (4) nickel-titanium. The wires have different uses during orthodontic treatment based on their physical properties (Table 15.1).
Table 15.1. Physical Properties of Major Orthodontic Wire Types1
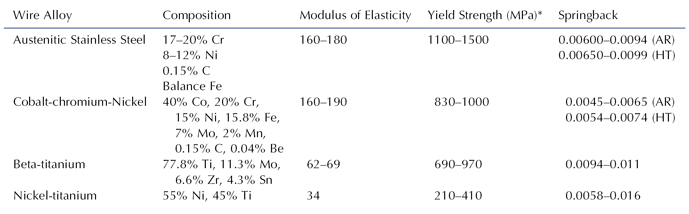
*Values correspond to 0.1% permanent tensile strain.
AR, as received; HT, heat-treated conditions.
1Brantley et al. 1997; Brantley 2001.
Stainless Steel Wires
The 18-8 stainless steel wires are composed of iron (73.75%), chromium (18%), nickel (8%), and carbon (0.25%). The 18-8 alloys are strong and ductile at room temperature. They resist tarnish and corrosion because of the presence of chromium. A thin, transparent layer of chromic oxide, known as a passavating film forms on the surface of the wire and prevents corrosion. If the oxide layer is disturbed by mechanical or chemical actions the wire can corrode. The nicks and rough places left on a wire after it is bent with pliers may produce localized electric couples that allow corrosion cells to form in the presence of an electrolyte solution such as saliva. If bits of carbon steel are incorporated into the surface of a stainless steel wire that comes into contact with carbon steel pliers, cutters, or burs, the embedded carbon steel becomes a couple that starts the process of corrosion. Chlorine cleansers attack 18-8 steels and cause corrosion. Acetic and lactic acids do not attack the passavating film and can be used in the cleaning of Hawley retainers containing stainless steel wires.
Sensitization
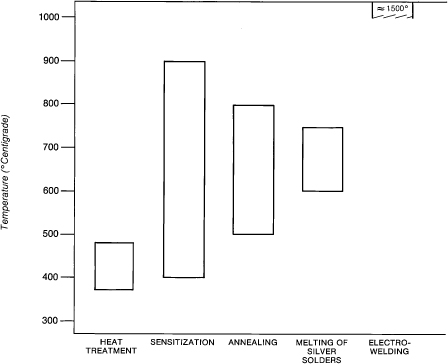
Depending on their carbon content, the 18-8 alloys lose resistance to corrosion when heated from 400°C to 900°C (Fig. 15.1). The formation of chromium carbide at the grain boundaries causes the alloy to lose its resistance to corrosion. Chromium carbide most rapidly forms at 650°C and begins to decompose above that temperature. The loss of chromium from the alloy that occurs with heat is known as sensitization. The corrosion along the grain boundaries where chromium carbide forms weakens the structure of the wire. Stainless steel can be stabilized against the formation of chromium carbide by adding an element such as titanium that precipitates as carbide in preference to chromium. Very few of the stainless steel wires used by orthodontists are stabilized.
Figure 15.1. Effects of heat on 18-8 stainless steel.
Cold Working
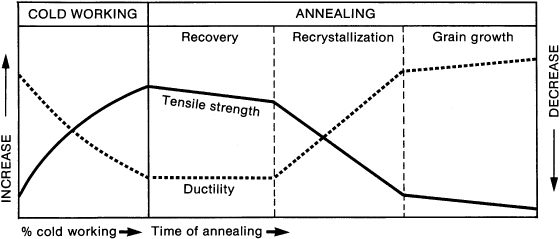
As a stainless steel wire is bent, tensile strength increases and ductility decreases (Fig. 15.2). Greatly strain-hardened wires are brittle and break more easily. Put as few bends in a wire as possible, so that it will function without breaking. Wires continue to work harden as they move teeth, are adjusted, and altered during mastication.
Figure 15.2. How cold working, recovery heat treatment, and annealing affect the tensile strength and ductility of stainless steel wires. (Redrawn from Richman 1967.)
Recovery Heat Treatment
The physical properties of cold-worked, strain-hardened stainless steel wires can be improved significantly with recovery heat treatment. When 18-8 steel wires are heated at moderately low temperatures of 370°C to 400°C after cold working (bending), the heat removes residual stresses, stabilizes the shape of the wire, and increases its elasticity, yield strength, and resilience (Fig. 15.2). Recovery heat treatment in a welder or oven should last from 1 to 2 minutes. The wire will turn to a light straw color. Sensitization occurs above 400°C; the wire darkens to dark brown at 500°C and a dark bluish brown at 600°C.
Annealing
If 18-8 stainless steel is heated above 500°C, the metal will begin to anneal and lose its elasticity and resilience (Fig. 15.2). Between 700°C and 800°C, a stainless steel wire becomes fully annealed. Annealing eliminates strain hardening and distorted grains associated with cold working. Orthodontic ligature wires are made from annealed stainless steel because ductility is important for that use. Figure 15.2 illustrates the relationships between tensile strength and ductility during cold working, recovery heat treatment, and annealing. The ends of an arch wire may be annealed and bent at the distal ends of the molar tubes to keep the wire from migrating mesially out of one of the tubes between appointments. A wire is annealed with an open flame or by passing an electric current through it using attachments on a welding machine. Arch wires, clasps, labial bows, and finger springs are not annealed, because these parts of an appliance must have tensile strength and elasticity.
Cobalt-Chromium-Nickel Wires
The cobalt-chromium-nickel wires were developed in the 1950s (see Table 15.1). They come in four levels of resilience: (1) blue, soft; (2) yellow, ductile; (3) green, semiresilient; and (4) red, resilient. The wires are protected from corrosion by a film of chromium oxide that forms on the exterior of the wires, similar to the 18-8 stainless steel wires. The softer wires can be bent easily and after bending can be heat treated to greatly increase their resilience and yield strengths. Heat treatment with an electric welder for 1 or 2 minutes or an oven for 10 minutes is effective at 500°C. The performance of cobalt-chromium-nickel and stainless steel wires is similar.
Beta-Titanium Wires
Beta-titanium wires were developed by Burstone and Goldberg (1980). The developers recognized that this alloy could deliver lower forces than the stainless steel and cobalt-chromium-nickel alloys and thus be more compatible with periodontal and alveolar bone tissues during orthodontic treatment. Compared to stainless steel, the modulus of elasticity of beta-titanium is favorably lower, about 40% of stainless steel, yield strength is less, about 60% of stainless steel, but their spring back that increases working range is much better than stainless steel (see Table 15.1). Beta-titanium wires can be easily bent. The beta-titanium wires should not be heat treated by the orthodontist, but they can be welded. These wires have a surface roughness that is associated with arch wire-bracket sliding friction.
Nickel-Titanium Wires
The nickel-titanium alloy wires were developed at the Naval Ordinance Laboratory by Buehler, Gilfrich, and Riley (1963). Andreasen and associates (1971, 1972, 1978) developed the alloy for use in orthodontic treatment. The nitinol wires have a very low modulus of elasticity and high flexibility, very low yield strengths, and wide elastic working range (see Table 15.1). Nickel-titatanium arch wires have two major phases: austenitic and martensitic (Brantley and Eliades 2001). The martensitic stabilized alloys do not have shape memory or superelasticity, an example of which is Nitinol Classic (3M Unitek Orthodontic Products, Monrovia, CA). The martensitic-active alloys achieve shape memory through a thermoelastic effect. As the wire warms to mouth temperature, the deformed martensitic phase is transformed into the austenitic phase that moves teeth toward the intended shape of the wire. Neo Sentalloy (GAC International, Islandia, NY) is an example of a thermoelastic shape memory nickel-titanium wire. The austenitic-active alloys are transformed through stress into the martensitic phase. These alloys are super elastic wires; one example is Nitinol SE (3M Unitek Orthodontic Products). Further details about these unique wires are discussed by Brantley and Eliades (2001) and Khier, Brantley, and Fournelle (199/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


