Chapter 13
Problem-Solving Challenges in Compromised Roots, Root Canal Systems, and Anatomic Deviations
Problem-solving challenges and dilemmas in the total management of compromised roots and root canal systems and anatomic deviations addressed in this chapter are:
“It is seldom that we see canals in buccal roots of superior molars, or in roots of lower molars, in which a drill can be used. . . . There are canals that are constricted just at the chamber, sometimes so much so that they can scarcely be found. . . . There are canals in curved roots and canals obstructed by osseous growths that, if not properly opened, would most likely cause trouble. It is with this difficult class of root-canals that I wish to deal at this time.”< ?xml:namespace prefix = "mbp" />
“I have seen resorption of apical cementum progressing with such attending conditions, while on the opposite surface of the same root there was a marked hyperplasia of cementum. In fact, it seems quite common to find these processes going on at the same time.”
This chapter will focus exclusively on the total management of difficult or challenging root canal anatomies, such as immature root development and a necrotic pulp; teeth that have been altered due to caries (irritational dentin and calcification) or resorptive processes both internal and external; teeth with accentuated curvatures; and unusual root and pulp space development in C-shaped canals. The reader is encouraged to seek additional and supportive information from the other chapters in this text; however, the challenges addressed in this chapter seemed to warrant individual attention using the problem-solving format.
Problem-Solving Challenges in the Tooth With a Necrotic Pulp and Immature Apical Development: Apexification
Historically, when a dental pulp had undergone demise before full root formation, an apexification procedure was indicated and still is the treatment of choice.
The traditional apexification procedure requires complete canal cleaning, shaping, removal of smear layer, and disinfection (see
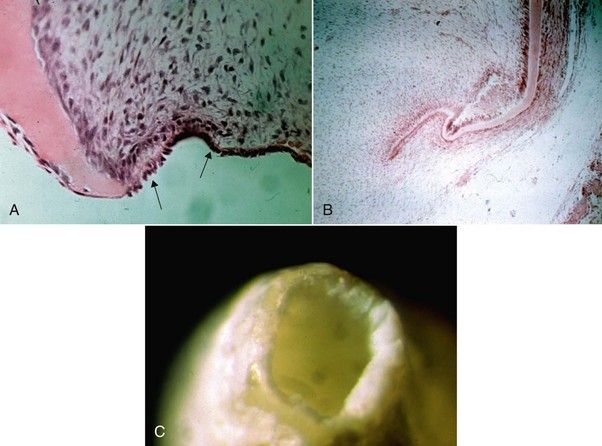
FIGURE 13-1 A, Developing root apex showing the Hertwig epithelial root sheath (arrows). B, Note how the sheath invaginates into the mesenchymal tissues (dental sac) on its way to root development. Once this sheath dies, the chance for normal root development ceases. C, View of tooth root that has not closed; perimeter of the opening represents the area of the root sheath.
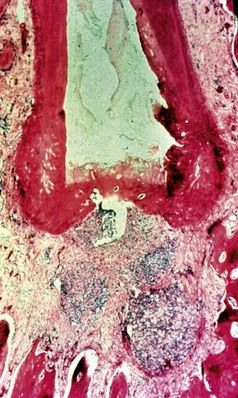
FIGURE 13-2 Apical closure of a primate’s tooth during apexification with calcium hydroxide, showing a gnarled-type of hard tissue has formed over the root canal. There is no root lengthening or root wall strengthening.
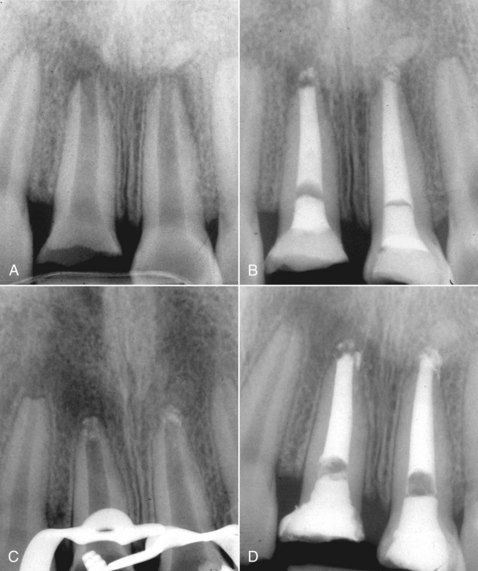
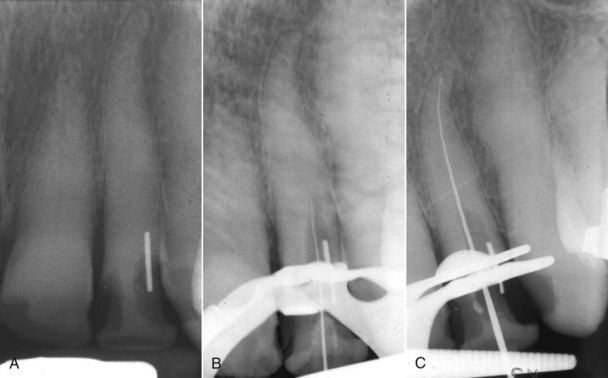
FIGURE 13-3 A, Two traumatized maxillary central incisors with immature root formation and necrotic pulps. B, Calcium hydroxide was placed. The patient is symptom free, and the teeth are functional. C, Removal of the calcium hydroxide at 1 year and verification of apical bridge formation. D, Both canals filled with gutta-percha and sealer. Note the porosity of the apical bridging.
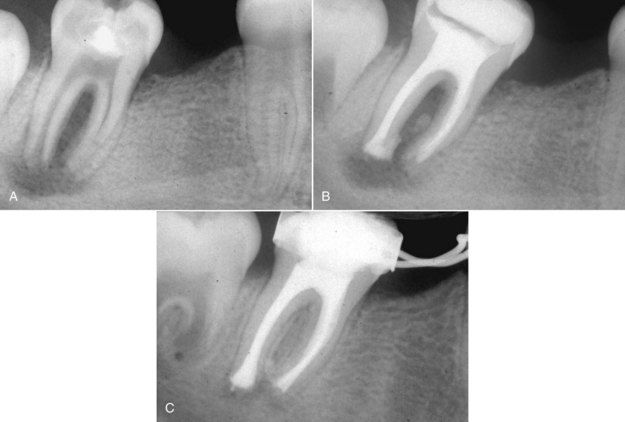
FIGURE 13-4 A, This 10-year-old male has significant decay and delayed root development on the mandibular first molar. Apical and furcation bone loss is evident, and the tooth is scheduled for extraction. B, The pulp chamber and canals are cleaned and shaped, and calcium hydroxide is placed. The open apices and slight extrusion of the material is noted. C, Six-month assessment shows great healing and the beginning formation of apical bridging. The roots are filled with gutta-percha and sealer. Slight porosity of the apical bridge is noted, but the healing response is excellent. A space maintainer is placed on the tooth.
From a contemporary standpoint, Ca(OH)2 is still used for bacterial control,
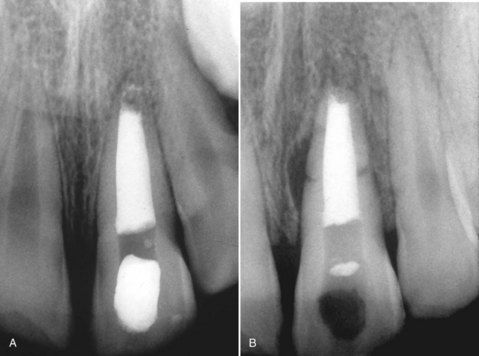
FIGURE 13-5 A, Maxillary central incisor that has undergone apexification and canal filling. B, Four months later, the patient had not had the tooth restored and complained of soreness in his gingival tissues. Radiograph shows a horizontal root fracture on the central incisor.
MTA, while effective in most cases in securing a positive tissue response and healing when used in a variety of applications,
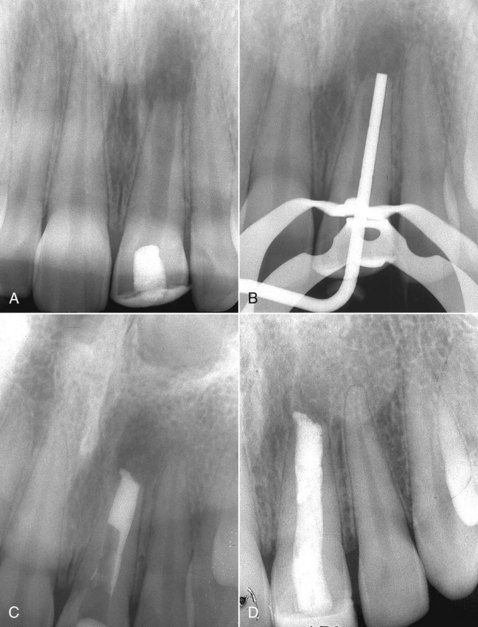
FIGURE 13-6 A, Maxillary lateral incisor with an open apex, thin root walls, and a large periapical lesion. The tooth was opened by her general dentist 2 months subsequent to a coronal fracture in which the pulp was exposed. B, The root canal was cleaned and a plugger (compactor) fitted into the canal for both length and width assessment. C, Mineral trioxide aggregate (MTA) is placed and compacted using the plugger. D, Canal is completely filled and a 6-month evaluation shows good bony response. Healing is not complete but is progressing.
The use of MTA in one-visit apexification treatment has received some significant attention,
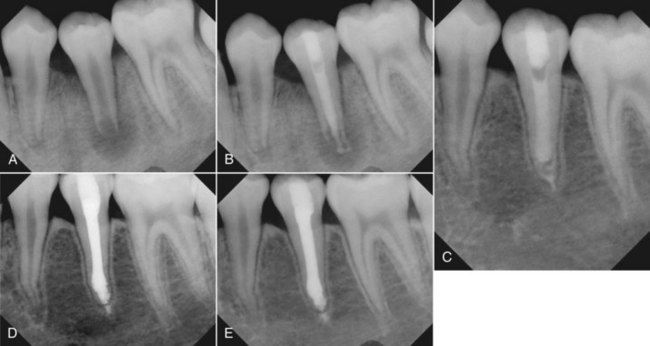
FIGURE 13-7 A, A 13-year-old male with a large periapical lesion on a mandibular second premolar. B, Calcium hydroxide was placed. C, Within 3 months, the apex appears to be closing. D, Entire canal filled with MTA. Note slight amount of material that is past the foramen, probably calcium hydroxide. E, Three months later, the tooth and periapical tissues are stable.
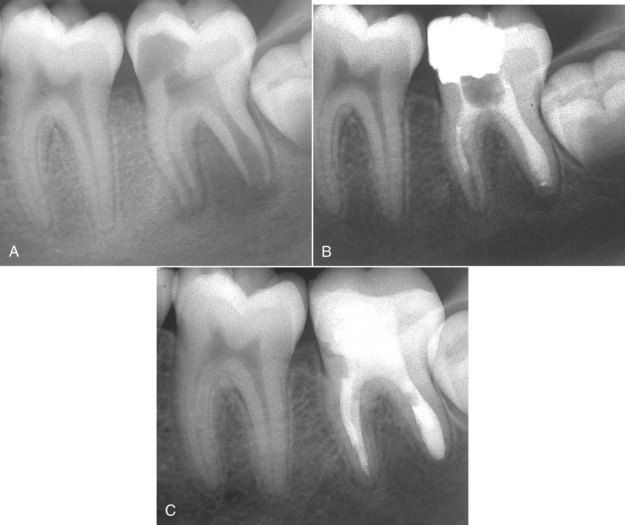
FIGURE 13-8 A, Mandibular molar with significant decay, periapical lesions, and open apices. B, Decay was excavated, and calcium hydroxide was placed. Four months later, the root apices appear to be closing. C, Mineral trioxide aggregate (MTA) was placed in the canal, and the tooth was restored.
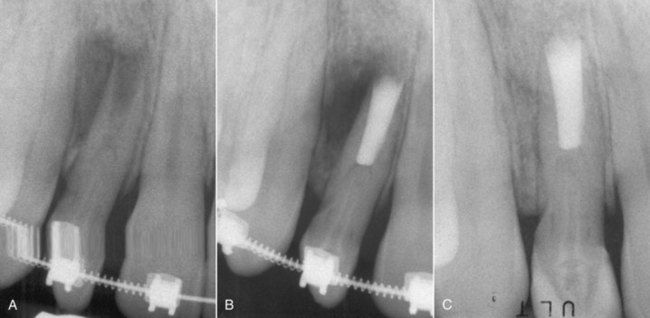
FIGURE 13-9 A, Maxillary lateral incisor with a dens in dente, open, immature apex, periapical lesion, and no response to sensibility testing. The dens in dente was intact, and surgery was chosen. B, Apical fill with mineral trioxide aggregate (MTA). C, 19-Month reevaluation healing is excellent.
Following placement of MTA in the canal, some options are available to the clinician for restoring the tooth, depending on whether only apical plugs were placed or the entire canal was filled.
The most contemporary approach to the management of teeth with immature root formation and either irreversible pulpitis and periapical periodontitis
Preliminary technique: The technique to achieve this goal consists of an access to the canal, drainage as necessary, rinsing the canal with sodium hypochlorite (NaOCl) and CHX, drying the canal, and placement of a mixture of ciprofloxacin, metronidazole, and minocycline with a lentulo spiral.
This initiative has even developed some of its own nomenclature in its claims, such as bioroot engineering, pulp revascularization, regenerative endodontic treatment or regenerative therapies, and stem cells from the apical papilla (SCAP).
It is not the intent of this text to espouse these theories or clinical innovations, but rather to raise some challenging problem-solving questions relative to the thought and direction behind them. The concept of stem cell identification and their potential applications is laudable, exciting, and should be pursued. However, there are issues that need distillation and clarification before these concepts and potential techniques can become meaningful and sought after by the clinician:
Problem-Solving Challenges in the Tooth With Fine and Calcified Canals
The problem of calcification was introduced in
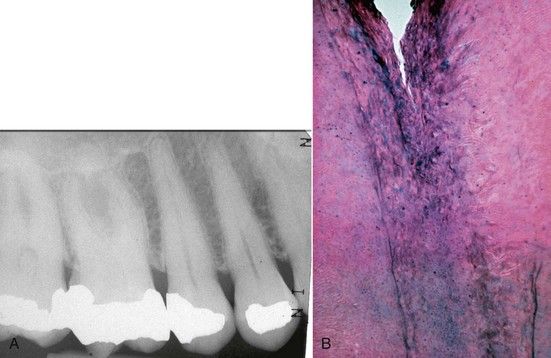
FIGURE 13-10 A, Maxillary molar with significant calcification present. B Histologic variations that may be noted in these types of calcified canals (H&E stain ×40).

CLINICAL PROBLEM
Problem
A 71-year-old female sought dental consultation because the porcelain fused to metal crown on her left maxillary lateral incisor fractured off at the gingival margin. There were no symptoms and the tooth had no clinical or radiographic signs of pathosis. A root canal space is evident on the examination film (
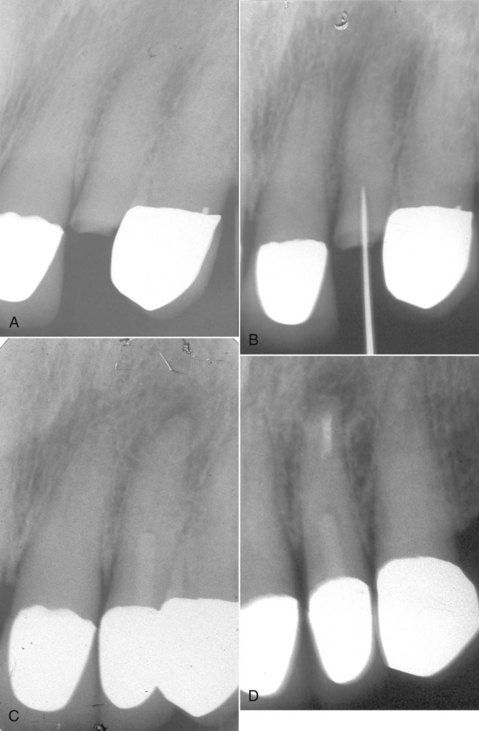
FIGURE 13-11 A, Complete crown fracture on a maxillary lateral incisor with extensive calcification. B, Unsuccessful attempt to negotiate canal. C, Two-year reevaluation following restoration with resin-fiber post retained crown. Apical pathosis is evident. Note: Conversion of access excavation into post space. D, Post operative radiograph. Apical surgery was indicated to clean and seal remaining canal space at the apex.
Solution
In the absence of symptoms or signs of pathosis, conversion of the excavation to a preparation for an intraradicular post is indicated. The tooth was subsequently restored with a resin-fiber type of post, followed by a new full crown. Two years later, the patient developed symptoms of periapical inflammation. A radiograph revealed an apical lesion(see

In these types of cases, the eventual development of periapical pathosis has been observed by the authors to be uncommon. It is also fortunate that only a small percentage of cases that radiographically exhibit fine or unidentifiable canals prove to be unmanageable using nonsurgical root canal techniques (
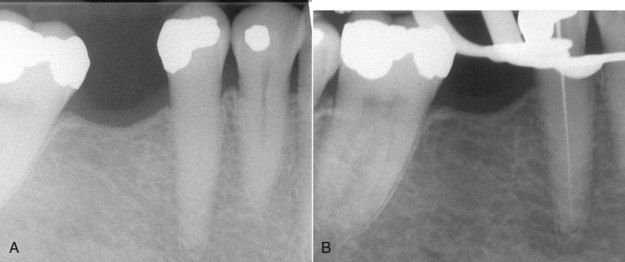
FIGURE 13-12 A, Mandibular premolar with apparent complete calcification of the entire root canal system.
B, Successful negotiation of the canal with a .08 file in combination with a solution containing EDTA.
This discussion will assume that the orifices have been located. Each tooth group will be somewhat different, but the concept and technique of gaining access to these canals are the same.
A 21-mm No. 8 K-file can be used initially to negotiate the calcified canal (
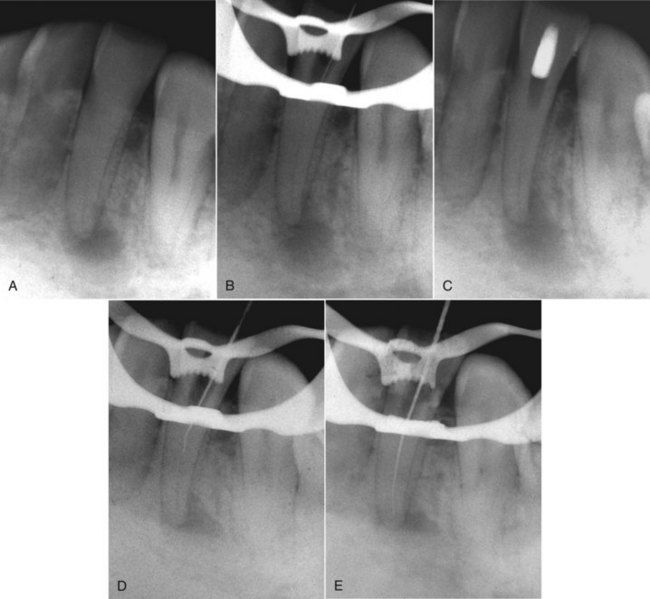
FIGURE 13-13 A, 38-year-old male presented with a history of tooth trauma to his mandibular anterior teeth. His chief complaint was periodic pain to pressure in the left lateral incisor. The tooth was nonresponsive to sensibility tests, with a slightly abnormal response to palpation and percussion. A radiograph showed significant pulp chamber and coronal canal calcification. B, Initial penetration of the pulpal space shows a mesial deviation of the file in the root and how the dental dam clamp can block direct vision of the file penetration. C, After failing to penetrate the canal, a temporary filling was placed and a radiograph was exposed without the dental dam in place to determine the angle of the access relative to the residual canal position. D, Reorientation and penetration with a small apically curved file. E, the canal was located, and penetration was achieved to the apex.
Before file insertion, a small curve is placed in its apical 1 mm. This can also be done with the C+ file. The precurved instrument must be directed along the pathway the canal is most likely to follow (see
During penetration, the chamber must be filled with NaOCl . A chelating agent can be used, but this will not dissolve loose debris, and it will negate the action of NaOCl. The calcified canal should never be negotiated without irrigant. This approach only serves to pack debris or calcifications into the canal and risks complete blockage. As the file is teased into the canal, try to establish a “sense of the patency” as this will provide a guideline for subsequent instrument use. Obtaining radiographs to verify the file position is exceedingly important
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses