Polymers in Prosthodontics
Objectives
After reading this chapter, the student should be able to:
< ?mpslid E1?>< ?mpslid S2?>
< ?mpslid E2?>< ?mpslid S3?>
3. Explain how free-radical polymerization is initiated for dental polymers.
< ?mpslid E3?>< ?mpslid S4?>
< ?mpslid E4?>< ?mpslid S5?>
< ?mpslid E5?>< ?mpslid S6?>
< ?mpslid E6?>< ?mpslid S7?>
< ?mpslid E7?>< ?mpslid S8?>
< ?mpslid E8?>< ?mpslid S9?>
< ?mpslid E9?>< ?mpslid S10?>
< ?mpslid E10?>< ?mpslid S11?>
11. Explain how acrylic polymers are bonded to alloys to form combination prostheses.
< ?mpslid E11?>< ?mpslid S12?>
< ?mpslid E12?>< ?mpslid S13?>
13. Explain why ceramic denture teeth are rarely used today.
< ?mpslid E13?>< ?mpslid S14?>
< ?mpslid E14?>
Key Terms
Acrylic polymers
Addition polymerization
Camphorquinone
Copolymers
Cross-linking
Curing
Denture bases
Fillers
Free-radical polymerization
Homopolymers
Initiator
Methyl methacrylate
Monomers
Plasticizer
PMMA, poly(methyl methacrylate)
Polymer
Polymerization
Record bases
Residual monomer
Residual stress
Wax rim
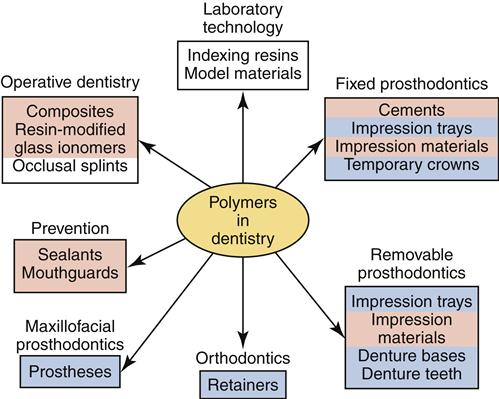
Polymers are nearly ubiquitous in dentistry and widely used in prosthodontics, primarily for removable prostheses such as complete or partial dentures (Figure 13-1). However, polymers also are used for denture teeth, impression trays, temporary crowns, and maxillofacial prostheses. Prosthodontic polymers have specific compositions and physical and chemical properties to ensure appropriate clinical performance, but the properties of these polymers overlap to some extent with those used for direct esthetic restorative materials (Chapter 4), mouth protectors (Chapter 3), impression materials (Chapter 8), and cements (Chapter 7). This chapter focuses on the properties and manipulation techniques that are most critical to polymers used in fixed, removable, and maxillofacial prosthodontics.
Some prosthodontic polymers may not be formed or manipulated in the dental office. For example, many of the fabrication steps of a removable complete or partial denture occur in a dental laboratory. Yet all members of the dental team need a basic knowledge of these materials. This knowledge ensures effective communication among dental office staff, patients, the dentist, and the laboratory personnel—resulting in optimal patient care and optimal care of polymer-based dental prostheses. Furthermore, the dental auxiliary may be asked to manipulate other polymers for temporary crowns, impression trays, orthodontic appliances, preventive restorations, or cements. Thus, knowledge of prosthodontic polymers is paramount.
Polymerization
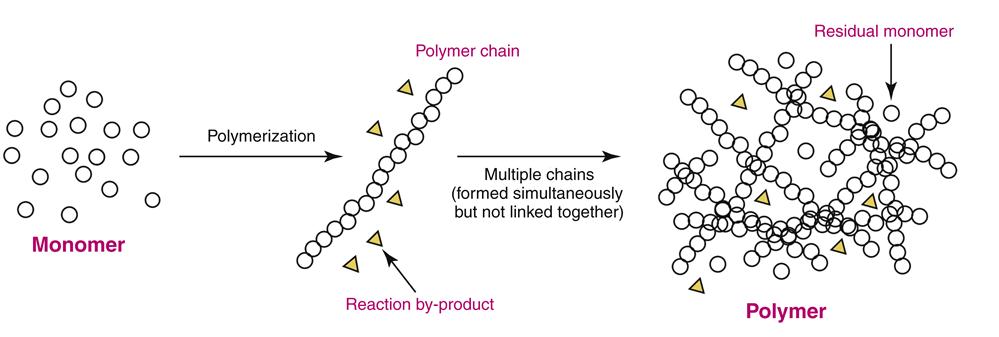
Polymerization chemically links small organic compounds called monomers into long chains of repeating monomer (“mer”) subunits (Figure 13-2). A single polymer chain commonly contains 10,000 to 100,000 linked monomer subunits. Because polymerized chains have molecular weights thousands of times greater than their parent monomers, the physical and chemical properties of polymers are always distinct from monomers. For example, methyl methacrylate, a common monomer used to make complete dentures, is a liquid that boils at about 100° C. Yet poly(methyl methacrylate) (PMMA) polymers (composed of methyl methacrylate monomer) never boil, but are rigid solids that decompose before they even melt! The simplest polymers consist of many long chains that behave much like long strands of spaghetti. In the simplest case, each polymer strand is not chemically linked to the other, but the strands entangle to create a polymer network that gives the polymer its particular physical properties (see Figure 13-2).
The nature of a monomer greatly influences the properties of its resulting polymer. Each monomer has at least one chemical group that participates in the polymerization reaction but may have other chemical groups that influence the polymer to be flexible, rigid, or water absorbing. For a number of complex reasons, not all monomers may be able to participate in the polymerization reaction, and any unreacted residual monomer in the polymer may have significant clinical consequences. For example, residual monomer may cause allergic reactions or chemical “burns.” The extent to which all monomer is converted to polymer is the degree of conversion. Polymers with high degrees of conversion have low levels of residual monomer; a high degree of conversion is generally desirable clinically.
The chemical reactions during polymerization may produce by-products such as water, hydrogen gas, or alcohols (see Figure 13-2). Because these by-products are at least temporarily trapped in the polymer network, they are important to the physical nature of the polymer. For example, polymers that form volatile alcohol by-products commonly shrink as the alcohol evaporates from the polymer network over a period of hours to days. Shrinkage may or may not be important to the performance of the dental prosthesis. The nature of the polymerization reaction also regulates dimensional changes during polymerization. If a monomer expands as it links to other monomers, then the polymer may not shrink as much as a monomer that loses mass by formation of a by-product. Most prosthodontic polymers used in dentistry shrink significantly as polymerization proceeds, and shrinkage generally has negative clinical consequences.
All chains in a polymer network are not of identical length. As polymerization proceeds, monomer is consumed by thousands of growing chains, each adding monomer units independently of the other. Each polymer chain will grow until monomer is no longer accessible, but the number of monomer units will be somewhat different for each chain. Thus, in a polymer network, the chains will have a distribution of sizes. The extent to which monomer is converted into polymer is called the degree of polymerization (to be distinguished from the degree of conversion, discussed previously). Polymers with a high degree of polymerization have fewer polymer chains that are longer (on average); those with a lower degree of polymerization have more polymer chains, but each is shorter. The degree of polymerization influences the physical properties of a polymer because shorter strands interact differently than longer strands in the polymer network. As a rule of thumb, high degrees of polymerization lead to more rigid, less soluble polymer networks, which is desirable in clinical applications.
Methyl Methacrylate, Acrylic Polymers (PMMA), and Free-Radical Polymerization
In prosthodontics, acrylics are a major class of polymers used for complete dentures, portions of removable partial dentures, maxillofacial prostheses, temporary crowns, custom impression trays, and denture teeth. Acrylics also are used in orthodontic appliances and in several laboratory procedures in casting and soldering (for example, indexing resins). Acrylic polymers are formed through free-radical polymerization. Free-radical polymerization uses monomers that have a carbon–carbon double bond as their reactive group. The most common acrylic monomer is methyl methacrylate, but hydroxyethyl methacrylate and butyl methacrylate also are common in dentistry. Methyl methacrylate is a sweet-smelling liquid that boils at 100° C. The odor of methyl methacrylate characterizes many dental laboratories, but chronic inhalation should be avoided because of potential toxicity to the liver. Methyl methacrylate also is highly flammable and allergenic. The careers of many dentists and dental auxiliaries have ended when they developed a severe allergy to this chemical.
Addition polymerization of methyl methacrylate to form poly(methyl methacrylate) (PMMA, acrylic) begins when a free radical (a highly reactive molecule with a free electron) is formed. Free radicals can be formed in a number of ways (Figure 13-3). The free radical attacks the carbon double bond of the monomer and initiates a series of reactions of monomer additions. For this reason, free radicals are commonly referred to as initiators. The free-radical reaction rapidly adds methyl methacrylate molecules in this manner (thousands per second!), one after the other. In this fashion, the polymer chain grows until no more monomer is available. When accessible monomer is depleted, the last free radical “terminates” via one of several complex reactions, and polymerization is complete. Some monomer cannot be added to the polymer chains and becomes trapped in the polymer as residual monomer.
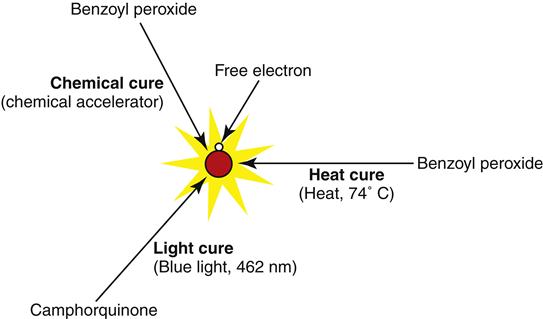
Acrylic free-radical polymerization is distinctive because it produces no reaction by-products. Because no by-product is formed, this type of polymerization is called addition polymerization. Addition polymerization reactions are used to form other dental polymers in impression materials (Chapter 8), restorative composites (Chapter 4), and cements (Chapter 7). Addition polymerization is preferred clinically because without reaction by-products, there is no risk of leaching of the by-product, which can increase shrinkage of the polymer or toxic reactions to the patient. In spite of the absence of reaction products, acrylics have significant polymerization reaction shrinkage, approaching 6 vol %. This degree of polymerization shrinkage poses significant problems for the fit of dental prostheses and temporary crowns (see later sections). The free-radical polymerization reaction also is highly exothermic, which means that heat is generated during polymerization. Heat generation is so great that it can boil unreacted methyl methacrylate, creating porosity in the final polymer. In the laboratory, steps must be taken to avoid this porosity. Heat generated during the fabrication of temporary crowns may burn adjacent tissues or even damage the pulp of the tooth!
Cross-Linking, Copolymers, Plasticizers, and Fillers
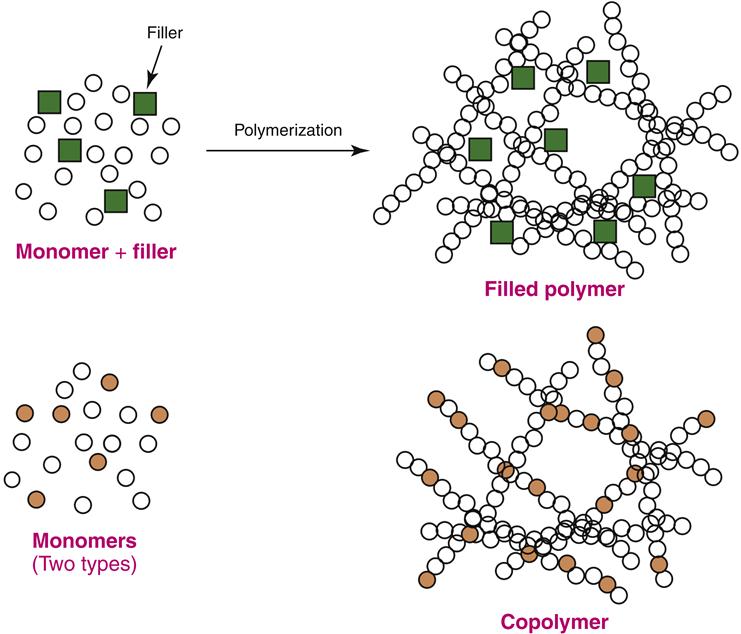
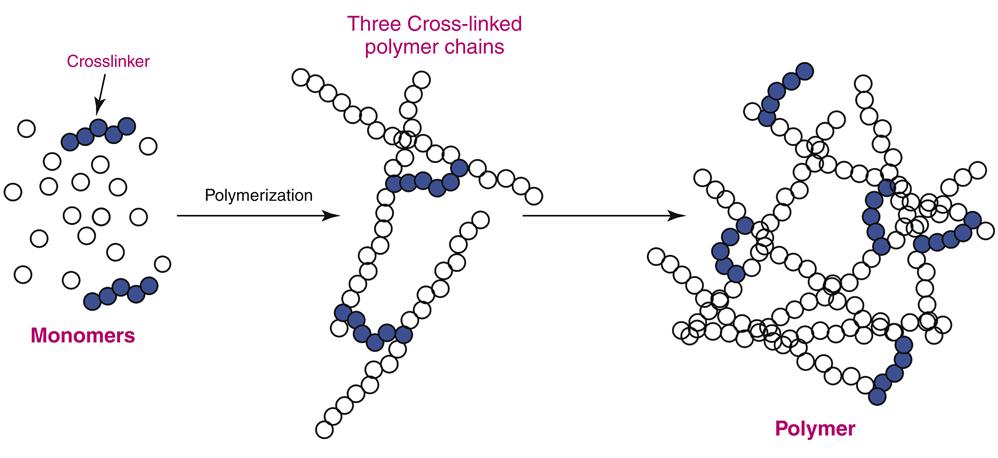
In PMMA (acrylic) polymers, each polymer strand is chemically unbonded to the other (see Figure 13-2). However, the physical and chemical properties of the polymer can be markedly changed if the strands are linked together. Such linkages are called cross-links (Figure 13-4). The cross-links are created by adding a cross-linking agent to the monomer before polymerization occurs. The degree of cross-linking may be controlled by the amount of cross-linking agent added to the monomer. Cross-linked acrylic polymers are more rigid, more temperature resistant, and less soluble than their uncross-linked counterparts. In dentistry, cross-linking improves wear resistance and susceptibility to organic solvents in foods containing ethanol (e.g., wine). Cross-linked acrylics also are easier to grind and polish. Cross-linked polymers are used in acrylic denture teeth and direct esthetic restorative materials (Chapter 4). They also have been used occasionally in some dentures to increase fracture resistance in patients prone to denture failure because of heavy biting forces or occupational activities (e.g., contact sports).
The acrylics discussed so far are homopolymers—that is, the polymer strands contain only one type of monomer. However, it is possible to use more than one type of monomer in a free-radical polymerization reaction (Figure 13-5, lower diagram). Polymers that have two or more types of monomer in their polymer network are called copolymers. When polymerization proceeds, the two monomers are randomly incorporated into the polymer strands. There also are methods to control the order of monomer addition (such as block polymer formation), but these strategies are beyond the scope of this discussion.
Copolymers are fabricated to modify the physical and chemical properties of the polymer network. For example, in denture bases, copolymerization of the monomers methyl methacrylate and butyl methacrylate results in a copolymer that is more fracture resistant. Addition of comonomers such as octyl methacrylate makes the copolymer soft and flexible, and these copolymers are used for denture soft liners. Addition of hydroxyethyl methacrylate increases the wettability of the polymer by water and improves complete denture retention in a patient with xerostomia (dry mouth). Copolymers also are common in direct esthetic restorative materials (Chapter 4) or mouth protector materials (Chapter 3). For example, a vinyl acetate-ethylene copolymer is used for mouth protectors to maximize energy absorption and prevent the energy of facial trauma from being transferred to the oral structures.
On occasion, chemicals called plasticizers are added to a polymer that do not participate in the polymerization reaction and do not become part of the polymer strands, yet are trapped in the polymer network. These chemicals are used to modify the physical properties of the polymer. For example, liquid oils such as dibutyl phthalate will cause a PMMA to become rubbery. Plasticizers dissolve into the polymer network and modify the interactions between the polymer strands. Plasticizers are most often used in polymer networks that are not cross-linked. Often, the polymer will expand when plasticized as the chemical dissolves into the network. Plasticizers are used in dentistry to soften polymers for denture soft liners. However, because plasticizers are not chemically linked to the polymer network, they leach out of the polymer over time, and their effect on the polymer network is lost. For this reason, denture soft liners harden with time, requiring periodic replacement. The effect of the released plasticizer on oral tissues must also be considered.
Fillers are solid particles added to polymers to change their optical or physical properties (Figure 13-5, upper diagram). Denture polymers are sometimes filled with small particles of butadiene-styrene rubber (itself a copolymer) to increase the resistance of the PMMA to fracture. Because filled denture bases have impact strengths of twice that of unfilled bases, these modified polymers are used in patients who have a history of denture base fracture. Inorganic fillers such as glass particles are added to PMMA to give the polymer color or change its handling properties during fabrication. For example, impression tray materials are filled to give the setting polymer a heavy doughlike consistency. The shape and thickness of the tray is then controllable as the material is formed onto a working cast to make the impression tray. Without the filler, the setting polymer would be too runny and sticky. Filler also improves the working consistency of elastomeric dental impression materials (Chapter 8), and it is used to modify the esthetic and wear properties of direct restorative materials (Chapter 4).
Fillers may be chemically linked into the polymer network. This strategy is used to maximize the ability of the filler to strengthen the polymer network, and these polymers have better wear resistance. The advantage of chemically linked fillers is that they cannot easily be dislodged from the polymer network by oral forces such as chewing. This strategy also minimizes polymerization shrinkage. Chemicals called coupling agents are used to link the filler to the polymer. Chemically linked fillers are used in light-activated impression tray materials and denture repair materials.
Complete Dentures
Complete dentures and partial dentures use filled PMMA (acrylic), uncross-linked homopolymers for the denture base, chemically bonded to denture teeth made of filled, cross-linked acrylic homopolymers. In this section, we first describe the process of complete denture fabrication and the options available for processing the denture. We then discuss the physical and chemical properties of acrylic polymers that influence clinical performance of the denture and how inappropriate care of the denture by the patient or the dental auxiliary may adversely affect dentures. Knowledge of this process enhances the ability of the auxiliary to aid chairside as well as in the laboratory and in communication to the patient.
Fabrication of Complete Dentures: An Overview
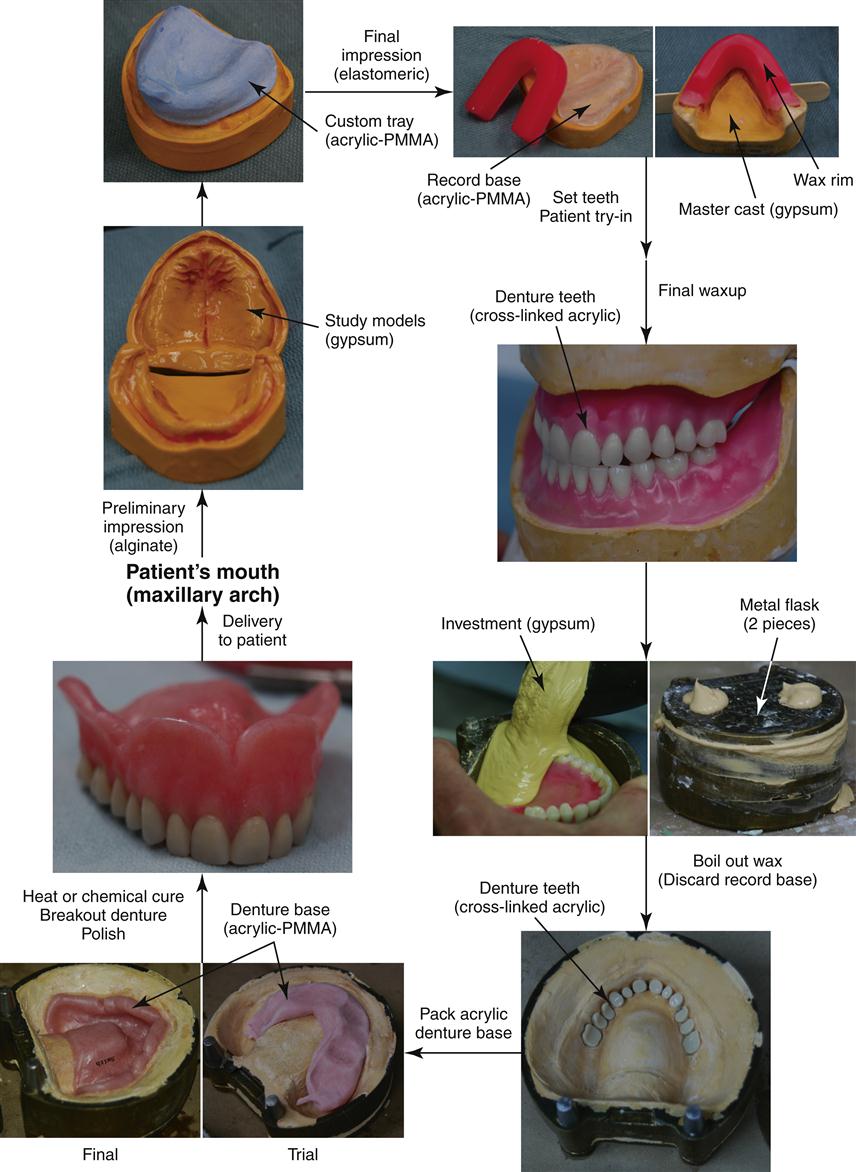
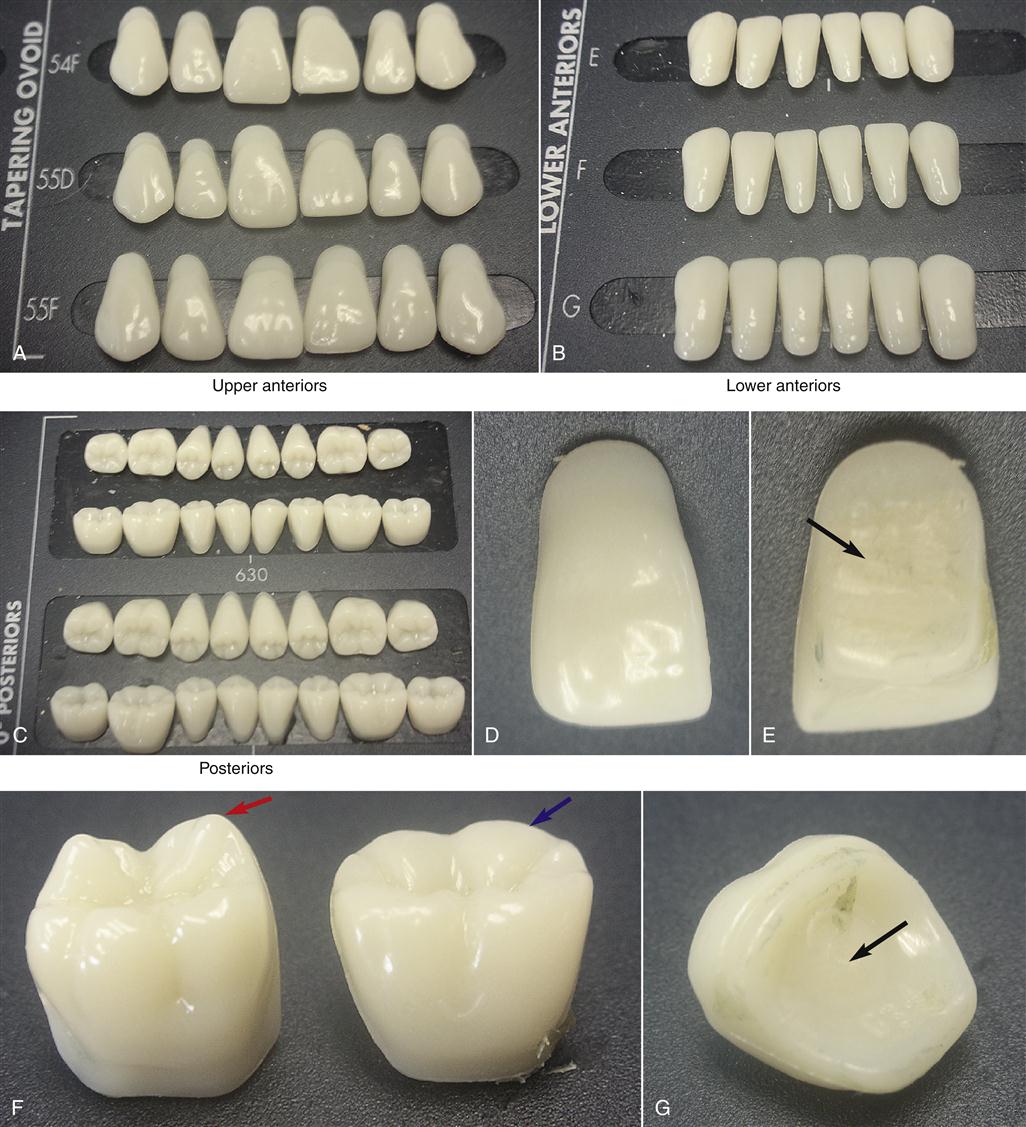
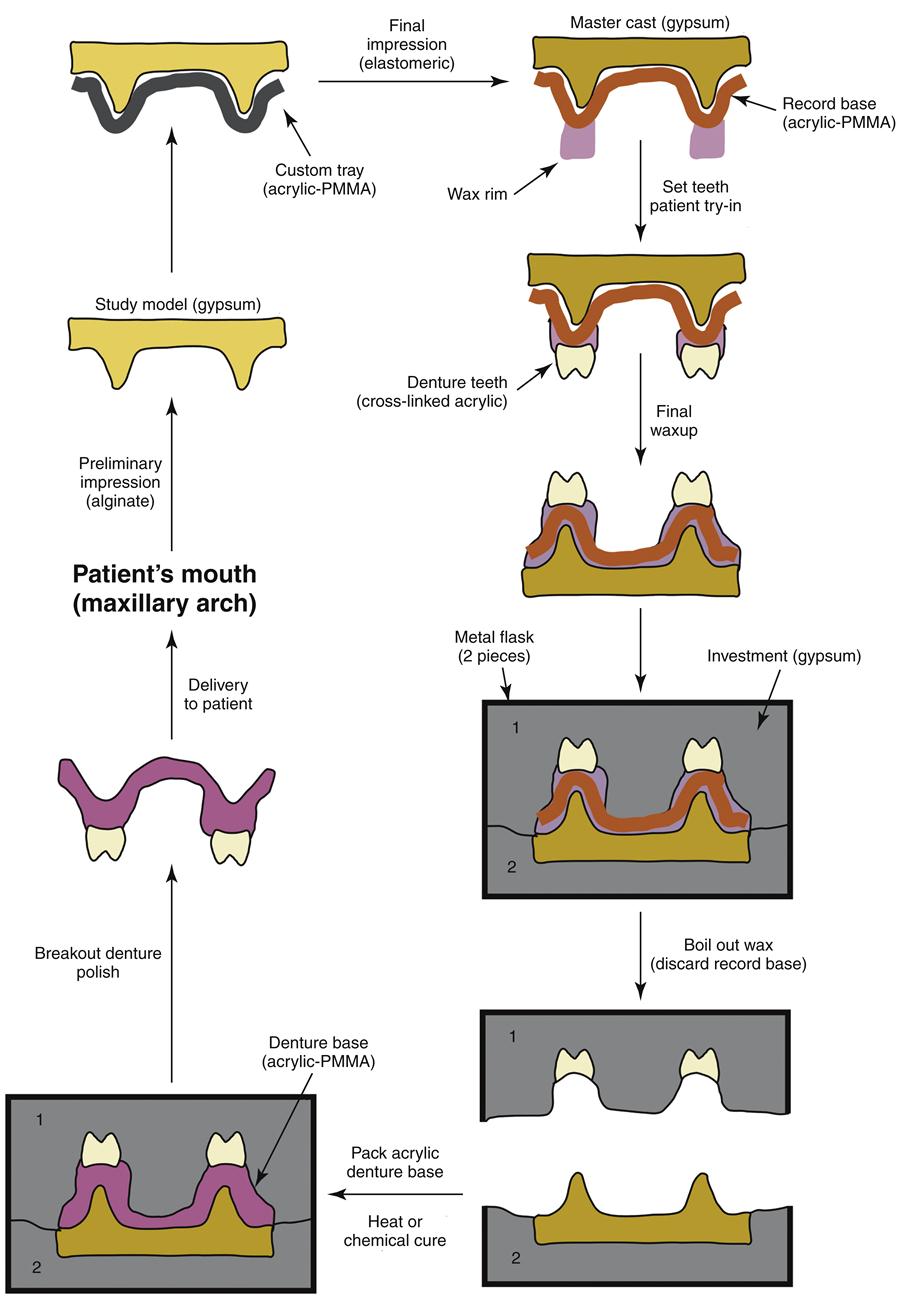
Complete dentures are fabricated in a rather complex series of steps over several appointments with the patient (Figures 13-6 and 13-7). Initially, a preliminary alginate impression is made, and preliminary working models are poured in dental stone. From these preliminary models, a custom impression tray is fabricated from a filled PMMA polymer (custom impression trays, often light-cured), and this tray is used to obtain the final impression using an elastomeric material in a second appointment. The final impression is poured in dental stone to obtain the master cast on which the denture will be constructed. Record bases are then made from unfilled PMMA, and a wax rim is added. This record base–wax rim combination serves as a trial denture that is used to determine jaw relations and tooth position for the final denture. The master casts are mounted on an articulator to capture the patient’s upper and lower jaw relationship, and the acrylic denture teeth are selected (Figure 13-8) and set into the wax rim. The esthetic and functional positions of the teeth are verified by inserting the record bases–wax rims into the patient’s mouth, and the teeth are moved as necessary to finalize esthetics, speech, and occlusion.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses