Impression Materials
Objectives
After reading this chapter, the student should be able to:
1. Describe the function of an impression material.
< ?mpslid E1?>< ?mpslid S2?>
2. Describe the relationship between a tooth, an impression of the tooth, and the die.
< ?mpslid E2?>< ?mpslid S3?>
3. List the requirements for an ideal impression material.
< ?mpslid E3?>< ?mpslid S4?>
4. List the components in an alginate powder, and describe their function.
< ?mpslid E4?>< ?mpslid S5?>
5. List the five objectives for alginate impressions.
< ?mpslid E5?>< ?mpslid S6?>
< ?mpslid E6?>< ?mpslid S7?>
7. Describe how a tray may be modified for an alginate impression.
< ?mpslid E7?>< ?mpslid S8?>
8. Describe the proper dispensing and mixing of an alginate.
< ?mpslid E8?>< ?mpslid S9?>
9. Describe the proper loading of alginate into the tray.
< ?mpslid E9?>< ?mpslid S10?>
10. Describe the procedure for taking an upper and lower impression in alginate.
< ?mpslid E10?>< ?mpslid S11?>
11. Describe the proper handling and storing of an alginate impression.
< ?mpslid E11?>< ?mpslid S12?>
12. Compare the properties of hydrocolloid and elastomeric impression materials.
< ?mpslid E12?>< ?mpslid S13?>
13. Describe the advantages and disadvantages of alginate hydrocolloid impression materials.
< ?mpslid E13?>< ?mpslid S14?>
14. Describe the setting of alginate impressions.
< ?mpslid E14?>< ?mpslid S15?>
< ?mpslid E15?>< ?mpslid S16?>
16. List which die or model materials are compatible with the various impression materials.
< ?mpslid E16?>< ?mpslid S17?>
< ?mpslid E17?>< ?mpslid S18?>
18. List the various methods of disinfection of impressions and their impact on the accuracy.
< ?mpslid E18?>< ?mpslid S19?>
19. Describe the important properties of elastomeric bite registration materials.
< ?mpslid E19?>< ?mpslid S20?>
20. Describe the properties of alginate substitute impression materials.
< ?mpslid E20?>< ?mpslid S21?>
21. List the advantages of a hydrophilic elastomeric impression material.
< ?mpslid E21?>< ?mpslid S22?>
22. Describe the important characteristics of digital impression systems.
< ?mpslid E22?>
Key Terms
Addition silicone
Agar
Alginate
Computer-aided design/computer-aided manufacturing (CAD/CAM)
Condensation silicone
Elastomeric
Gel
Hydrocolloid
Hydrophilic
Hydrophobic
Hysteresis
Impression compound
Mercaptan
Monophase
Polyether
Polymer
Polysulfide
Retarder
Sol
Syneresis
The function of an impression material is to record accurately the dimensions of oral tissues and their spatial relationships. In making an impression, a material in the plastic state is placed against the oral tissues to set. After setting, the impression is removed from the mouth and is used to make a replica of the oral tissues. The impression gives a negative reproduction of these tissues. A positive reproduction is obtained by pouring dental stone or other suitable material into the impression and allowing it to harden. The positive reproduction is called a model or cast when large areas of the oral tissues are involved or a die when single or multiple tooth preparations are recorded.
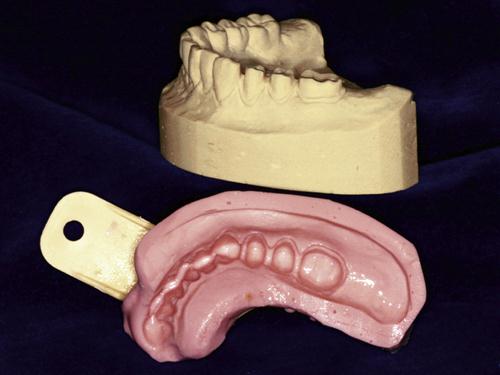
The relationships between a tooth, an impression of the tooth, and a die are illustrated in two dimensions in Figure 8-1. When examined from the anterior side, the various parts of the tooth and the impression are in the same relationship to each other, although the highest portions of the tooth are the deepest parts of the impression. When the maxillary die is examined with the occlusal portion downward, the die is a positive reproduction of the tooth preparation. If the die is examined with the occlusal surface upward, the buccal surface (B) is on the left rather than the right. A similar relationship is shown in Figure 8-1 for a cross section of a mandibular left molar, its impression, and the die made from it. The impression of the mandibular molar, when examined from the anterior side, is inverted for ease of examination, and the buccal (B) and lingual (L) surfaces are reversed with respect to the tooth. The cusp (C) is the highest area on the tooth and the lowest area of the impression, and the position of C for the tooth is on the left but on the right for the impression. The die of the mandibular molar, however, is a positive duplication of the tooth.
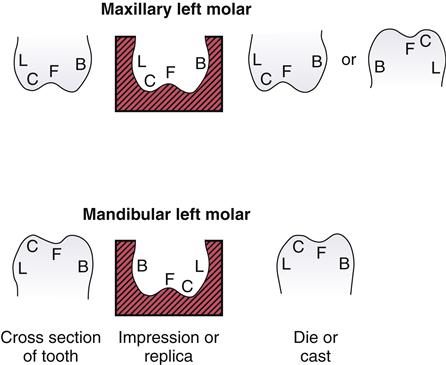
(B, buccal surface; C, cusp; F, fissure; L, lingual surface.)
Impressions may be taken of portions of a tooth, a single tooth, several teeth, a quadrant of the mouth, or an entire dentulous or edentulous arch. Examples of some of these types of impressions and the corresponding dies or casts are shown in Figure 8-2.
A variety of impression materials are described in this chapter, which is indicative of the fact that no single material is ideal for all applications. The list of properties for an ideal impression material presented in Box 8-1 emphasizes the many demands placed on these materials. Not surprisingly, none of the current materials completely satisfies these requirements.
Impression materials can be classified as those that are flexible and those that are rigid at the time of removal from the mouth. A rigid impression material is restricted to applications in areas where no undercuts exist. A rigid impression material could not be used on the teeth shown in Figure 8-1 because, on setting, it would be locked in place and could not be removed over the bulge of the tooth without fracturing. A rigid material can be removed from a tooth prepared for a full crown or from an edentulous arch, as shown in Figure 8-2, C, because no undercut areas are present. However, a flexible impression material could make these two impressions, in addition to impressions of single teeth or a full dentulous arch. Not surprisingly, impression materials that are flexible when set are used most frequently. Rigid impression materials (dental compound, zinc oxide–eugenol impression paste) are described in Appendix 8-1.
Impression materials discussed in this chapter are listed in Table 8-1 and are classified as rigid or flexible. An estimate of the usage of impression materials in restorative procedures is 51% for addition silicones, 32% for polyethers, 9% for polysulfides, 6% for hydrocolloids, and 2% for condensation silicones.
TABLE 8-1
Classification of Dental Impression Materials
| RIGID | FLEXIBLE |
| DENTAL COMPOUND | AQUEOUS |
| Zinc oxide–eugenol | Alginate hydrocolloid∗ |
| Elastomeric | |
| Addition silicone (PVS)∗ | |
| Polyether∗ | |
| Addition silicone–polyether hybrid | |
| Condensation silicone | |
| Polysulfide (rubber base) |
Alginate Hydrocolloid Impression Material
Alginate is one of the most widely used aqueous dental impression materials. The wide use of alginates results from (1) the ease of mixing and manipulating them, (2) the minimum equipment necessary, (3) the flexibility of the set impression, (4) their accuracy if properly handled, and (5) their low cost. Their principal disadvantages are that they have low tear strength and they do not transfer as much surface detail to gypsum dies as elastomeric impressions do.
Alginates are used extensively to prepare study models of either the entire dental arch or a segment of it. They also are used to prepare gypsum models for the preparation of athletic mouth protectors. They are not recommended for making impressions of cavity preparations.
Note: Agar hydrocolloid impression materials are described in Appendix 8-1.
Packaging
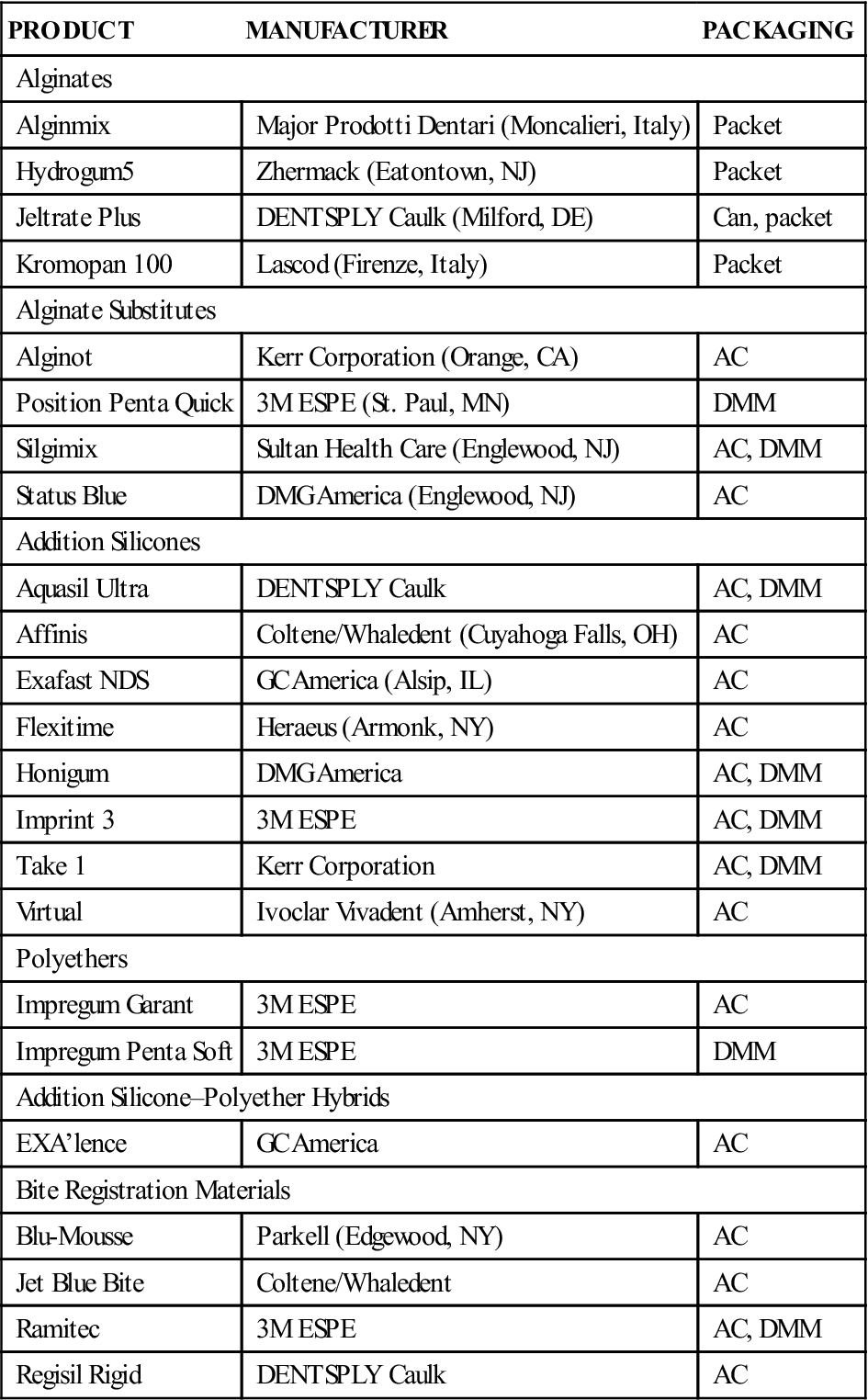
Manufacturers supply alginate as a powder that is packaged in bulk or in preweighed individual packets. Examples of alginate impression materials are listed in Table 8-2. The bulk material is packaged in a sealed screw-top plastic container; in a hermetically sealed metal can, such as that used to package coffee; or in foil packets as shown in Figure 8-3. The preweighed packages are constructed of plastic and metal foil and contain enough material for a single full-arch impression. These packages minimize moisture contact with the powder and extend the storage life of the alginate.
TABLE 8-2
Examples of Alginate Hydrocolloid and Elastomeric Impression Materials
< ?comst?>
| PRODUCT | MANUFACTURER | PACKAGING |
| Alginates | ||
| Alginmix | Major Prodotti Dentari (Moncalieri, Italy) | Packet |
| Hydrogum5 | Zhermack (Eatontown, NJ) | Packet |
| Jeltrate Plus | DENTSPLY Caulk (Milford, DE) | Can, packet |
| Kromopan 100 | Lascod (Firenze, Italy) | Packet |
| Alginate Substitutes | ||
| Alginot | Kerr Corporation (Orange, CA) | AC |
| Position Penta Quick | 3M ESPE (St. Paul, MN) | DMM |
| Silgimix | Sultan Health Care (Englewood, NJ) | AC, DMM |
| Status Blue | DMG America (Englewood, NJ) | AC |
| Addition Silicones | ||
| Aquasil Ultra | DENTSPLY Caulk | AC, DMM |
| Affinis | Coltene/Whaledent (Cuyahoga Falls, OH) | AC |
| Exafast NDS | GC America (Alsip, IL) | AC |
| Flexitime | Heraeus (Armonk, NY) | AC |
| Honigum | DMG America | AC, DMM |
| Imprint 3 | 3M ESPE | AC, DMM |
| Take 1 | Kerr Corporation | AC, DMM |
| Virtual | Ivoclar Vivadent (Amherst, NY) | AC |
| Polyethers | ||
| Impregum Garant | 3M ESPE | AC |
| Impregum Penta Soft | 3M ESPE | DMM |
| Addition Silicone–Polyether Hybrids | ||
| EXA’lence | GC America | AC |
| Bite Registration Materials | ||
| Blu-Mousse | Parkell (Edgewood, NY) | AC |
| Jet Blue Bite | Coltene/Whaledent | AC |
| Ramitec | 3M ESPE | AC, DMM |
| Regisil Rigid | DENTSPLY Caulk | AC |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>

A plastic scoop is provided for dispensing the bulk powder, and a plastic cylinder is supplied for measurement of the water (see Figure 8-3). A wide-bladed, reasonably stiff spatula is used to mix the powder and water. Mechanically driven mixers (Alginator II; DUX Dental, Oxnard, California) are also available.
Composition
Table 8-3 shows the ingredients in alginate powder and their functions. When water is mixed with the alginate powder, a smooth viscous mass is formed, which becomes an irreversible gel a few minutes after mixing. The overall simplified reaction is as follows:
< ?xml:namespace prefix = "mml" ns = "http://www.w3.org/1998/Math/MathML" />
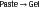
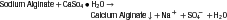
TABLE 8-3
Ingredients and Their Function in Alginate Powder
| INGREDIENT | FUNCTION |
| Sodium or potassium alginate salt | To dissolve in water |
| Calcium sulfate | To react with dissolved alginate to form insoluble calcium alginate |
| Sodium phosphate | To react preferentially with calcium sulfate and serve as a retarder |
| Diatomaceous earth or silicate powder | To control consistency of mix and flexibility of impression |
| Potassium sulfate or potassium zinc fluoride | To counteract the inhibiting effect of alginate on the settling of gypsum model or die material |
| Organic glycol | To coat the powder particles to minimize dust during dispensing |
| Pigments | To provide color |
| Quaternary ammonium compounds or chlorhexidine | To provide self-disinfection |
| Aspartame | To function as a sweetener |
The manufacturer controls the time of setting by the amount of sodium phosphate present in the alginate powder. As long as any sodium phosphate is present, it reacts preferentially with the soluble calcium ions. After all the sodium phosphate has reacted, the soluble sodium alginate reacts with the remaining calcium ions, and calcium alginate precipitates. The sodium phosphate therefore is called a retarder. The calcium alginate precipitates into a fibrous network; water occupies the intervening capillary spaces. This type of structure is called a gel. At least one of the dimensions of the network is colloidal (0.5 µm), and this material traditionally has been named alginate hydrocolloid. The reaction is driven by the lower solubility of calcium alginate compared with sodium alginate. These materials frequently are referred to as irreversible hydrocolloids, because once the paste sets to a gel, the process cannot be reversed.
Properties
American National Standards Institute–American Dental Association (ANSI-ADA) Specification No. 18 (ISO 1563) for alginates established requirements for odor, flavor, lack of irritation, uniformity, mixing and setting times, permanent deformation (alteration in shape) at the time of removal from the mouth, flexibility at the time of pouring the model or die, compressive strength, reproduction of detail, compatibility with gypsum, and deterioration of the packaged powder during storage.
Mixing and Setting Times
Alginates, when properly mixed by hand, should develop a smooth, creamy consistency free of graininess in less than 1 minute for the normal set material and should be suitable for making impressions in the mouth. The setting time of the alginate is indicated as normal or fast by the manufacturer. An alginate sold as a normal-setting material should set in no less than 2 minutes or more than 4½ minutes after the start of the mix and be workable for up to 2 minutes. The setting time of fast-setting alginate is between 1 and 2 minutes and workable for at least 1¼ minutes. The mixing time of fast-setting alginate is 30 to 45 seconds. In general, the setting time should be no less than that listed by the manufacturer and at least 15 seconds longer than the stated working time.
Because the setting occurs as a result of a chemical reaction, an increase in the temperature of the water used to prepare the mix shortens the working and setting times. The proportions of powder and water also affect the setting times. Thinner mixes increase the time required for the material to set. The usual range of setting times for normal-setting mixes of commercial alginate impression materials is from 2½ to 5 minutes.
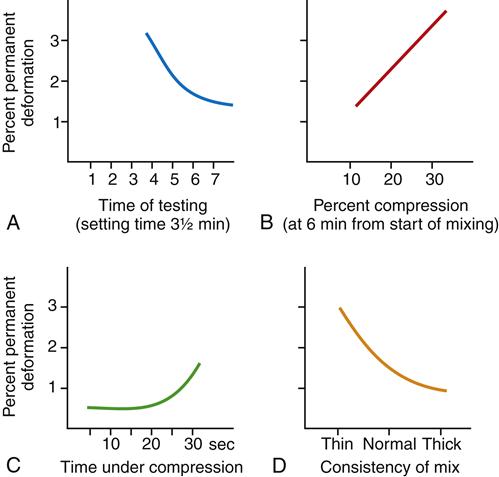
Elastic Recovery
Because the set alginate is held between the impression tray and the tissues, knowledge of the extent of any permanent deformation during the removal of the impression is important (Figure 8-4). The ANSI-ADA specification requires at least 95% elastic recovery (no more than 5% permanent deformation) when the alginate is compressed 20% for 5 seconds, which simulates removal of the impression from the mouth. Many commercial alginates have actual values of 96% to 98% elastic recovery (2% to 4% permanent deformation). Thus, alginate impression material is flexible but not perfectly elastic (Box 8-2).
Flexibility
The ANSI-ADA specification sets limits of 5% to 20% in compression at the time a model or die is prepared in the impression (10 minutes after the start of mixing). The compression is measured between a stress of 0.01 and 0.10 MPa. Typical values for commercial alginates are between 12% and 18%, but a few manufacturers supply hard-set alginates, which have values of about 5% to 8%.
The relative amounts of water and powder influence the flexibility of the set alginate. Thicker mixes result in lower flexibility.
Strength
The strength of alginates in compression and resistance to tearing are important requirements, although the tear strength is the more critical. The ANSI-ADA specification requires a minimum compressive strength of 0.37 MPa at a time when the material is removed from the mouth. Most commercial products have compressive strengths substantially higher than this limit with values ranging from 0.5 to 0.9 MPa.
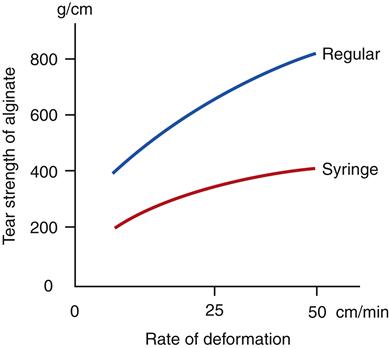
The tear strength of alginates varies from 3.7 to 6.9 N/cm. Because many sections of an impression are thin, tearing may result from a rather small applied force.
The strengths of alginates are a function of the rate at which the impression is deformed; higher rates of deformation (removal) result in higher compressive and tear strengths. An example of the effect of the rate of deformation on tear strength is shown in Figure 8-5, which illustrates that alginate impressions are less likely to tear during removal from the mouth when they are removed rapidly.
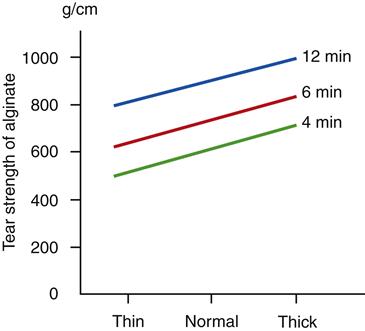
The strength of alginate impression materials increases if thick rather than thin mixes are used. The advantage of the use of increasingly thicker mixes is somewhat limited because the consistency becomes too thick, and the flow during seating of the impression is so low that an adequate impression cannot be obtained.
The tear and compressive strengths at the time the impression is removed increase if the time of removal is delayed. The effect of the consistency of the mix and of the time of removal on the tear strength is shown in Figure 8-6. Again, only limited advantage can be taken of the increase in tear strength with time because of the inconvenience of leaving the impression in the mouth for longer periods.
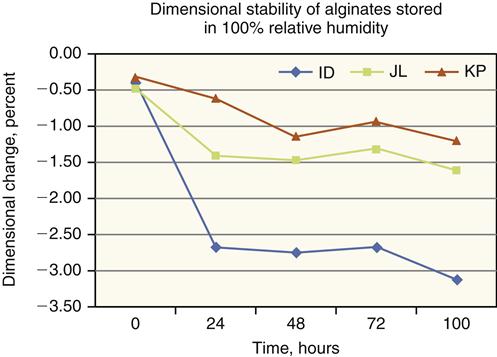
Dimensional Change
The accuracy of an impression material is important, and alginates are no exception. A problem with alginate impressions is loss of accuracy with increased time of storage (Figure 8-7). The set alginate is a hydrocolloid gel that contains large quantities of water. This water evaporates if the impression is stored in air, and the impression shrinks. If the impression is placed in water, it absorbs water and expands. Therefore, storage in either air or water results in serious changes in dimensions and a loss of accuracy. Storage in humid air approaching 100% relative humidity results in the least dimensional change. Alginate gels, however, shrink even under conditions of 100% relative humidity as a result of a process called syneresis, in which water forms on the surface of the impression. Fortunately, syneresis occurs rather slowly, and alginate impressions prepared from modern products (see Table 8-2) usually can be stored under conditions of 100% relative humidity for up to 5 days without serious dimensional changes.
Studies of the dimensional change of alginates during storage emphasize that they should be stored for as short a period as possible and that the preparation of the model or die should proceed directly after the impression has been made. If the immediate preparation of the model or die is not possible, the next most appropriate procedure is to store the impression in an atmosphere of 100% relative humidity for the shortest possible time.
Reproduction of Detail
The impression material must record the detail of the oral tissues, but this detail also must be transferred to the model or die. The ADA specification recommends that alginates must have the minimum capabilities of transferring a line only 0.075 mm wide to a gypsum model or die material. A number of products have properties that exceed this minimum value.
Disinfection
Guidelines recommend that alginate impressions be rinsed and disinfected. Sodium hypochlorite, iodophor, glutaraldehyde, and phenylphenol solutions have been used, and some manufacturers have added disinfectants to the alginate powder. Studies have shown that test viruses have been inactivated in alginate impressions by (1) a 10-minute soak in 0.5% sodium hypochlorite or a 10-minute wait after spraying the impression with this solution; (2) a 10-minute immersion in an iodophor solution diluted 1:213; (3) a 20-minute immersion in 2% glutaraldehyde diluted 1:4; and (4) a 20-minute immersion in phenylphenol diluted 1:32. The incorporation of disinfectants in the alginate powder also was found to be effective.
Measurements of dimensional changes showed that immersion of alginate impressions for 30 minutes did not affect their clinical accuracy.
Objectives for Taking Alginate Impressions
The objectives are to record the following:
• All teeth in the upper and lower arch
< ?mpslid E33?>< ?mpslid S34?>
< ?mpslid E34?>< ?mpslid S35?>
• The retromolar area of the lower arch
< ?mpslid E35?>< ?mpslid S36?>
• The area of the hamular notch in the upper arch
< ?mpslid E36?>< ?mpslid S37?>
• A detailed, undistorted, and bubble-free reproduction of the oral tissues
< ?mpslid E37?>
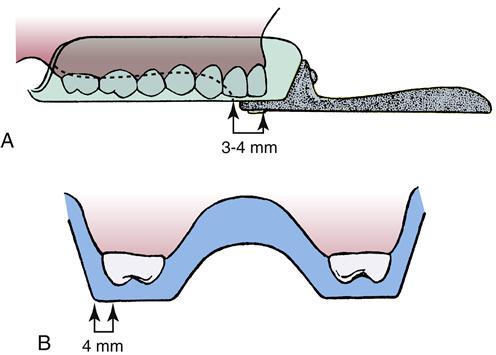
Selection of the Tray
Criteria for the maxillary tray:
• Completely cover the tuberosity
< ?mpslid E38?>< ?mpslid S39?>
• Be 4 mm wider than the most apical portion of the alveolar process at the molar region
< ?mpslid E39?>< ?mpslid S40?>
• Cover the anterior teeth with the incisors contacting the flat arch portion of the tray about 4 mm from the raised palatal part of the tray (Figure 8-8)
< ?mpslid E40?>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses