Waxes
Objectives
After reading this chapter, the student should be able to:
1. Describe the difference between pattern waxes and processing waxes.
< ?mpslid E1?>< ?mpslid S2?>
< ?mpslid E2?>< ?mpslid S3?>
< ?mpslid E3?>< ?mpslid S4?>
4. Describe the common properties of pattern waxes that are important clinically.
< ?mpslid E4?>< ?mpslid S5?>
< ?mpslid E5?>
Key Terms
Burnout
Ester
Hydrocarbon
Pattern wax
Processing wax
Waxes are used in many aspects of dentistry in the clinic and the laboratory. Although not used in the final dental restoration, waxes are often important in the fabrication and success of the final metal or ceramic restoration. The use of wax in dentistry dates back 200 years to the taking of impressions of the teeth with beeswax.
Dental waxes are always a mixture of various components. These components may be natural waxes from minerals, plants, or animals, or they may be synthetic waxes. Gums, fats, fatty acids, oils, and various resins also may be added to modify the properties. Dental waxes may have pigments for color. The mixing of components allows manufacturers to create a wide variety of properties useful to dentistry, but it also makes waxes complex.
Component waxes in dental waxes are usually organic molecules with high molecular weights as shown in the following structures:
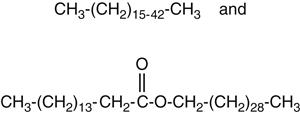
Organic molecules in waxes are composed primarily of long chains containing carbon, hydrogen, and oxygen. The top structure is a hydrocarbon (from paraffin), and the bottom structure is a high-molecular-weight ester. The parentheses in the structures indicate repeating units of carbon and hydrogen. The long chains allow waxes to be flexible at room temperature or sticky as solids or as liquids. Properties such as hardness, melting range, and flow depend on the amounts of the various waxes and the molecular structure of the organic molecules in the mixtures.
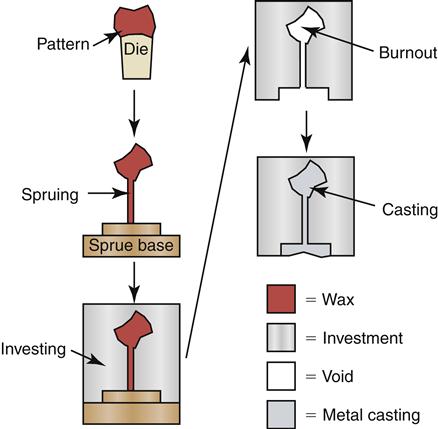
Waxes can be classified broadly as pattern waxes and processing waxes. Pattern waxes include casting waxes, baseplate wax, and inlay wax. They are used to fabricate a restoration using the lost-wax technique (Figure 10-1). Details of the lost-wax technique are described in Chapter 12. A model of an inlay pattern is shown in Figure 10-2. Processing waxes include boxing, beading, utility, and sticky wax. These waxes are used as auxiliary materials. For example, these waxes aid in the making of castings or impressions or during soldering. Self-cured and light-cured resins also have been used as pattern “waxes” or as “processing waxes.”
Important Properties of Waxes
Because of their complex composition, waxes have properties unlike any other dental material. Some properties are especially important for pattern waxes used to fabricate dental restorations. American National Standards Institute–American Dental Association (ANSI-ADA) Specification No. 122 (ISO 15854) describes minimum requirements. However, the following descriptions of properties apply to all waxes and are useful in understanding properties of most waxes.
Melting Range
Because waxes are mixtures of different components, they do not melt at a single temperature and do not have a melting point. Rather, they have a melting range. At the low end of the range, some but not all of the components melt, which causes the wax, still solid, to flow much more (see the discussion of flow). As the temperature increases through the melting range, more of the components melt, and the wax flows severely, and eventually all components become a liquid.
Excess Residue
Because wax patterns used in the lost-wax technique are melted or burned to remove them from the casting mold, the wax must not leave a residue, which would affect the quality of the final restoration (see Figure 10-1).
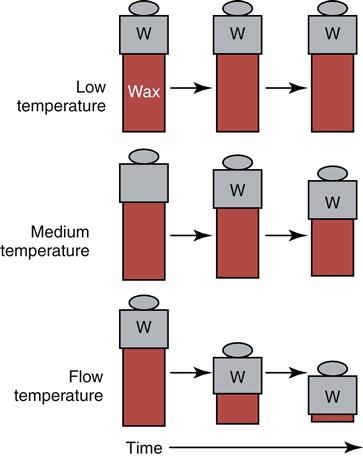
Flow
Flow is the change in shape under an applied force. It is caused by the slippage of the long-chained wax molecules over each other. Flow is highly dependent on temperature and time. At low temperatures, waxes hardly flow at all, but as the temperature approaches the melting range of the wax, the flow increases dramatically (Figure 10-3). For pattern waxes, flow is generally not desirable at room or mouth temperature, because it results in a permanent distortion of the wax pattern. For processing waxes, flow is a highly desirable property because these waxes need to be pliable at room temperature.
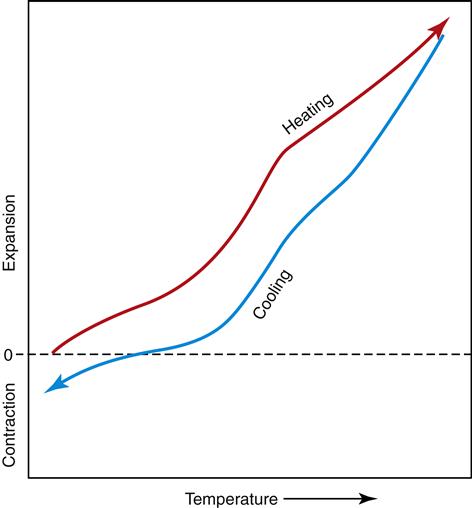
Thermal Expansion
When waxes are heated, they expand significantly. This expansion can be quantified as a percentage of the original dimension of the specimen. It usually is reported as parts per million of expansion per Celsius degree of the rise in temperature of the specimen (Figure 10-4). This number is called the “coefficient of thermal expansion of the wax,” and the higher this coefficient is, the greater the expansion as the wax is heated. In general, waxes have the highest coefficients of thermal expansion of any dental material. For example, the coefficient of thermal expansion of a typical pattern wax is 323 × 10−6/°C, but that of dental ceramic is 20 times less at about 14 × 10−6/°C. For pattern waxes, the thermal expansion is critical. Small changes in temperature can cause a sufficient change in dimension to make the pattern inaccurate.
Residual Stress
Residual stress is stress remaining in a wax as a result of manipulation during heating, cooling, bending, carving, or other manipulation. Manipulation of wax puts molecules of the wax into positions that they do not like but cannot change because of their solid state. These stresses that are present in wax generally are released as the temperature of the wax increases, and the wax molecules can move more freely.
The release of residual stress at higher temperatures causes an irreversible deformation that can destroy the fit of a wax pattern. For this reason, techniques that prevent the formation of the stresses and keep those that have formed from being released have been developed in the manipulation of wax. To prevent residual stresses from forming, waxes should not be carved or burnished at temperatures well below their melting range. Wax patterns are carved with warm (37 °C) instruments, and melted wax is added in small increments to prevent rapid or uneven cooling, which promotes residual stress. To preve/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses