Chapter 13 Materials used in endodontics
Learning Objectives
From this chapter, the reader will:
• Appreciate the basic principles underpinning endodontics
• Be aware of the types of rubber dam and the necessity for its use during conventional endodontic therapy
• Be aware of the various types of endodontic file and how these should be used during root canal instrumentation
• Understand the importance of thorough disinfection of the root canal system by chemical irrigation and other more novel means
• Be aware of the materials used to temporize a tooth undergoing root canal therapy
• Be aware of the materials used to fill the root canal system and restore the tooth postoperatively
• Know the names of currently available commercial products.
Introduction
Endodontology is the branch of dental science concerned with the form, function, health, trauma and disease of the dental pulp. Endodontics is the practice of endodontology. Many patients will report that they have had root treatment, which is a misleading term. The correct technical term is root canal therapy (RCT), which more accurately describes the process, i.e. of cleaning and filling all of the root canals in the tooth. The term root filling is often used synonymously with root canal therapy but this only refers to the material that is used to fill the root canal system.
This chapter discusses the materials and the instrumentation used during endodontic procedures.
Rubber Dam
Reasons for use
The battle in endodontics is against the bacterial infection. To achieve a sterile root canal system the tooth should be isolated to prevent contamination with oral bacteria. The only predictable and effective method is the use of rubber dam, which is also referred to as dental dam (see Figures 13.1B, 13.2 and 13.4 below). The benefits of using rubber dam in relation to endodontics include:
• Protection of the oropharynx and prevention of inhalation or ingestion of instruments or medicaments
• Isolation of the operating field, rendering it aseptic
• Prevents ingress of saliva, and reinfection of the root canal system, by oral bacteria
As well as being used in endodontics, rubber dam also has many applications in general operative dentistry, particularly when the dentist is using hydrophobic materials, hence the many references to it in this text.
The rubber dam system
The rubber dam system consists of a sheet of an elastic material held taut over a frame. Some products have a frame incorporated in them, e.g. OptraDam (Ivoclar Vivadent). A hole or holes are punched into the sheet corresponding to the teeth that need to be isolated. The dam is retained by the use of a clamp or clamps or stabilizing cord such as Wedjets (Hygienic) (Figure 13.1).
Types and presentation of rubber dam
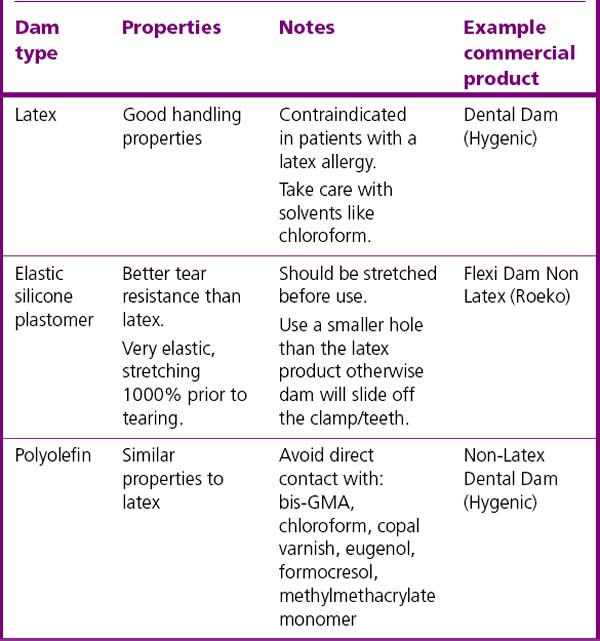
Rubber dam is available in latex and non-latex (elastic plastomer or polyolefin) presentations (Figure 13.2 and Table 13.1). It should be noted that the words ‘rubber’ and ‘dental’ in relation to dam are used synonymously although the elastic plastomers and polyolefins are polymers.
Rubber dam is either presented in precut squares of material (Figure 13.2) or in rolls where the dentist cuts off the desired amount. Different sheet thicknesses are available, the thicker sheets may be more difficult to place but they have a greater tear resistance and will retract the tissues more. Products are also available in different colours. Darker colours contrast with the operative site better so decreasing eye strain. Lighter shades, however, can naturally illuminate the area due to their greater translucency. Some products are supplied flavoured or scented in an attempt to improve the patient’s experience.
Rubber dam sealers
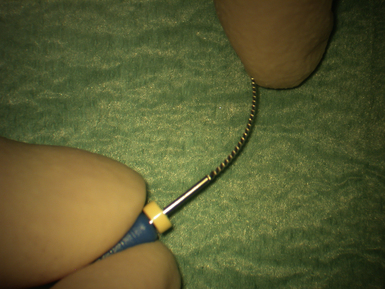
When rubber dam is placed for endodontics, it is advisable to create a seal around the dam using a caulk material. This prevents irrigants reaching the mouth and equally prevents saliva from leaking back into the operating field. The caulk material used is hectorite clay, which belongs to a group of materials called smectitec clays. In these materials platelets of a metal oxide are sandwiched between plates of silicon dioxide. This layered structure allows the clays to swell in contact with water. The metallic oxide in hectorite clay is magnesium (Figure 13.3). On taking up water the clay will expand to up to 35 times its original size effectively sealing any gaps adjacent to the dam.
An example of such a product on the market is OraSeal (Ultradent). The rubber dam is applied and the sealer is syringed around the now isolated tooth (Figure 13.4).
Endodontic Files
File preparation of root canals is done either by hand or using rotary files in a speed-reducing, torque-controlled handpiece (Figure 13.5). Rotary systems are much more efficient than those used by hand; however, hand files may be preferred to negotiate very curved root canals as rotary instruments may not be able to get round the curve due to lack of sufficient flexibility. The file may also break (separate) during use. The sharper the curve of the canal, the greater the incidence of fracture of the file due to cyclic fatigue.
Fracture may also occur due to flexural fatigue (i.e. overuse) or torsional fatigue, that is, forces placed on the instrument in rotation while the instrument is prevented from moving. The use of a torque controller is important so that the rotation of the file is stopped prior to receiving excessive torque, which may cause fracture. For further information on speed-reducing handpieces and torque, see Chapter 19. From this brief description, it can be seen that the selection of the material of which the file is made and its method of construction is critical in determining the performance of the instrument.
Anatomy of endodontic files
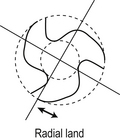
Hand and rotary files both have a cutting edge along the length of the file and at the other end is either a handle or an attachment that fits into the handpiece, respectively (Figure 13.6). The instrument cuts when its radial lands are in contact with the canal wall, the lead angle determining its cutting efficiency. Radial land areas are required for conventional helically fluted files because they prevent the file from over-engagement in the canal (Figure 13.7). Lack of radial land areas reduces friction. If a file becomes suddenly engaged or self-threaded, it may fracture. Radial lands are especially important for files that have positive rake angles. That is, the angle of action of the cutting blade is similar to a snow plough which is forced downward towards the surface of the road. Many files are designed so that the radial lands cannot screw themselves into the canal wall. Thus, the dentinal debris is directed towards the coronal part of the canal so that it is not compacted apically.
Stainless steel files
Work hardening: is the strengthening of a metal by plastic deformation. This strengthening occurs because the crystal structure of the material is dislocated by the movement. Any material with a fairly high melting point such as metals and alloys can be strengthened using this technique. Alloys such as low-carbon steel and stainless steel which are not amenable to heat treatment are often work hardened.
Stressing
The mechanical properties of the file will depend on the composition of the material, the geometry of the file and the way in which it is loaded. Anticlockwise twisting of a file is inadvisable as this increasing the twisting of the file, which may result in brittle fracture. Files should therefore not be stressed in this way and definitely not when they are bound in the canal. Some irrigants such as sodium hypochlorite and ethylene diamine tetra-acetic acid (EDTA) can reduce the cutting ability of stainless steel files and so they should be rinsed immediately after use. The risk of this occurring has been substantially reduced as the current recommendation is that files should only be used once and discarded. However, if canal preparation is extended over a long period of time at one appointment, prolonged immersion of these instruments in these solutions in the canal will start the process of degradation.
![]()
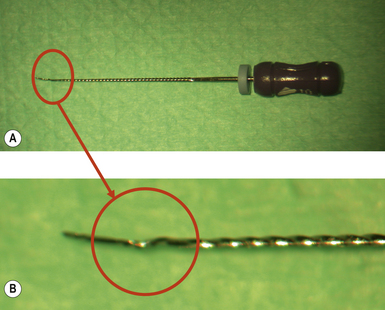
All files should be regularly inspected for damage after their removal from a root canal. If any damage is seen then the file should be discarded and a new file used. Continued use of a stressed instrument (Figure 13.8) has a high risk of instrument separation with the fractured fragment being retained in the root canal system. If the file cannot be removed, or the canal negotiated beyond it, the success of the treatment will be compromised. Unfortunately, fracture of files can also occur without any visible signs of previous permanent deformation. In order to minimize file breakage, files should only be used in a wet canal and in accordance with the manufacturer’s instructions.
Nickel-titanium files
An alloy which has been more recently used to produce endodontic files is nickel-titanium (NiTi). This material is extremely flexible (Figure 13.10), some 500% more than stainless steel and has been termed super-elastic. This means that it is more likely to maintain the canal shape during preparation as it can memorize its original shape. It is also less likely to straighten curved canals as occurs with stainless steel. With nickel-titanium instruments, after a finite number of rotations cyclic fatigue leads to failure. However, they are three times stronger and have a superior resistance to torsional fracture compared with the equivalent stainless steel file. They are also corrosion resistant. It is the alloy of choice for rotary systems although hand files are also available (Figure 13.6).
When the alloy is stressed during use, its structure changes from an austenitic crystalline structure to a martensitic crystalline structure. This happens progressively and as a result the stress on the file reduces even though the strain may be increased. The modulus of elasticity is much higher for an austenitic structure than for the martensitic form, meaning that the latter state is more brittle than the former. When the stress on the alloy decreases, spring back occurs without permanent deformation and the alloy returns to the austenitic phase. This allows an 8% strain deformation with complete recovery compared with less than 1% with stainless steel.
Root Canal Disinfection
Endodontic irrigants
• Lubricate endodontic files so facilitating their passage in the canal
• Wet canal walls and remove debris by flushing
• Contain any dentinal shavings in suspension, so facilitating their removal from the canals. This prevents impaction of debris in the apical portion of the canal which may prove difficult to renegotiate due to the blockage
• Facilitate dissolution of organic matter
• Remove smear layer and soften dentine
• Aid cleaning of areas inaccessible to mechanical cleansing methods
• Disinfect the canal system: mechanical root canal preparation alone is ineffective at microbial removal. It is essential that solutions used during root canal preparation can disinfect the root canal system.
The irrigants available can be divided into two groups based on their mode of action:
Biofilm and smear layer
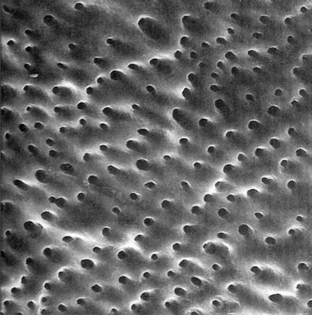
Bacteria colonize not only on the necrotic material in the lumen of the root canal but also infect its walls. Microorganisms can penetrate into the dentinal tubules as well as forming complex, highly developed ecologies on the surface of the canal wall. This has been termed a biofilm. In order to kill these organisms the biofilm must be disrupted and penetrated. Furthermore, during instrumentation organic material is left behind on the dentinal surface, termed the smear layer, which must also be removed to allow the disinfectant to gain access to lateral canals (Figure 13.11). If this is achieved, then the disinfection process will be enhanced.
Irrigants for cleaning
EDTA
EDTA may also be used as a chelating agent as an alterative to citric acid, its action being more aggressive. It is used as either a synthetic amino acid or sodium salt of EDTA and is highly effective at removing the smear layer and emulsifying soft tissue. EDTA demineralizes and softens root canal wall dentine by 20–50 μm. Although it is neither bactericidal nor bacteriostatic, EDTA-containing compounds (Figure 13.12) will eventually kill bacteria by starving them of the metallic ions needed for growth as the chemical chelates the ions. It is non-toxic and non-corrosive to instruments.
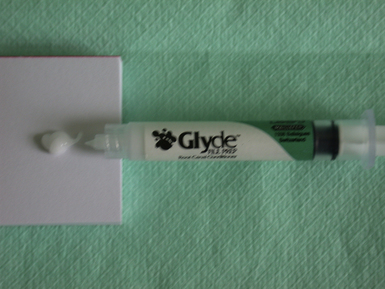
EDTA gel
EDTA is used in liquid form for endodontic irrigation but is also available in gel form (Figure 13.13). These products are used to coat endodontic files. They act as lubricants as well as helping to remove smear when the file is used within the root canal, making canal preparation easier and faster. The increased viscosity of this presentation is better for holding debris in suspension.
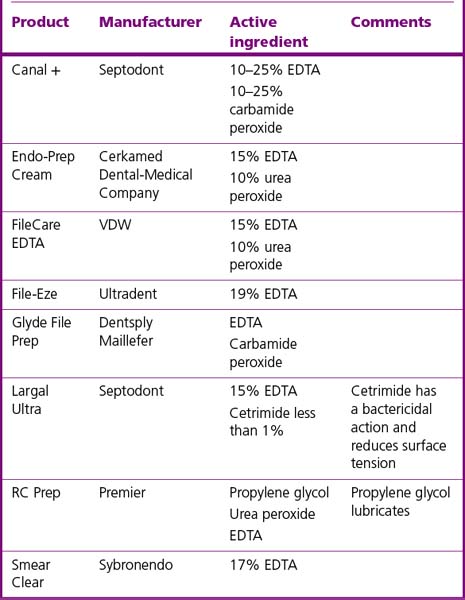
Commercially available products
Commercially available materials are shown in Table 13.2.
Table 13.2 Some EDTA-containing gels used during root canal preparation currently available on the market
![]()
• Chelating materials should be thoroughly washed from the root canal system as their retention for any length of time will continue to soften the dentine. This effect is eventually self-limiting as the chelator is used up. For best results, these irrigants should:
• Chelating materials soften the dentine within the tooth and so should not be initially used to negotiate the canals as a false canal may be cut iatrogenically.
Irrigants for disinfection
Sodium hypochlorite solution
• Commercial sodium hypochlorite endodontic irrigant products include Parcan Solution (Septodont), which is a 3% solution, or Chlorax (which is available as a 2% or 5.25% sodium hypochlorite solution from Cerkamed Dental-Medical Company). It can also be obtained from a pharmacy. The advantage of using these products is that they do not have to be made up at the chairside and they are buffered. Buffering maintains the solution’s properties throughout its shelf-life and extends this time. There are also products containing other chemicals to enhance the effect of the solution. For example, Chlor-Xtra (Vista) is a combination of sodium hypochlorite and Triton-X. This latter chemical is a surfactant that lowers the viscosity of the irrigant so improving its penetration in the dentinal tubules and narrow canals.
• An alternative is household bleach bought from a supermarket. This is usually supplied as a 1% solution in which case should be used neat or it can be diluted with tap water if required. However, the clinician must be satisfied that any material being used on a patient is approved for contact with humans or has been certified with a CE mark for dental or medical applications. This may preclude the use of supermarket-bought household bleaching products.
Many endodontists use sodium hypochlorite solution in combination with an ultrasonic instrument as they believe that the acoustic streaming that occurs enhances the cleaning effect.
Extracanal extrusion of endodontic irrigants
1. Stopping the irrigation, and considering giving additional local anaesthesia
2. Irrigating with sterile water or saline
3. Closure of the root canal with no medication and dressing the tooth
5. Considering prescription of an antibiotic, a steroid (such as dexamethasone) and an antihistamine.
6. Considering referral to an oral and maxillofacial surgical unit for specialist care and monitoring.
Chlorhexidine
Chlorhexidine digluconate (Figure 13.14) is used by many endodontists as it has a number of beneficial properties. This chemical is a cationic bis-biguanide that is bacteriostatic at low (0.2%) and bactericidal at higher (2%) concentrations. Its mode of action is to cause cell wall decomposition, leading to the loss of cellular components. It does not, however, dissolve any organic tissue. It is active against a wide spectrum of microorganisms, with its antibacterial properties similar or greater to that of sodium hypochlorite. It is known that the bacterial flora and ecology of endodontic cases which have failed is different. In these cases, chlorhexidine is thus preferred because it may have an effect on microorganisms resistant to sodium hypochlorite. Other clinicians use it in preference to sodium hypochlorite as it is a safer alterative (see below).
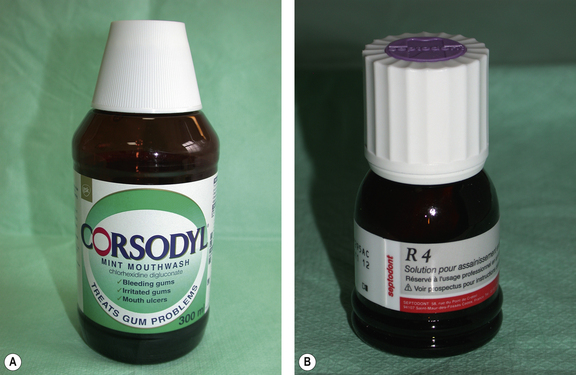
Fig. 13.14 Examples of products containing chlorhexidine that can be used for endodontic irrigation: (A) Corsodyl (GlaxoSmithKline) and (B) R4 (Septodont). The former product is also very effective against periodontal pathogens and is thus widely used in the treatment of periodontal disease (see Chapter 17).
Ozone
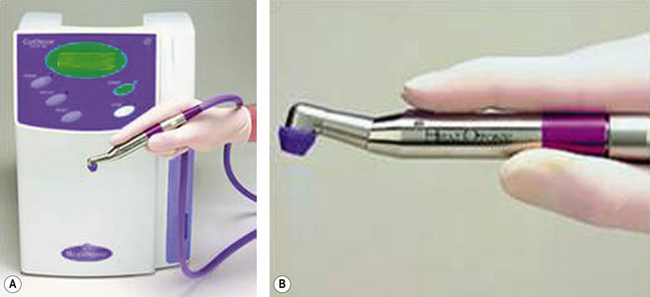
Ozone is used in the food and environmental industries for large-scale sterilizing of water supplies. The gas is a very effective sterilizing agent but at levels close to which it has toxicological effects can damage normal tissue. Devices are now available whereby the gas can be produced and delivered down a handpiece to the operating site in the mouth (Figure 13.15). In one device the area is sealed from the surroundings. The residual gas is then drawn back to the generator and neutralized using a platinum filter. More recent devices deliver the ozone down a fine tip directly into the canal for very short exposure times. This system does not require a seal around the tooth.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses