Interpolated Paramedian Forehead Flaps
Introduction
Median forehead flaps were first described in an Indian medical treatise, the Sushruta Samita, in approximately 700 BC.1–3 The operation was performed by members of a caste of potters known as the Koomas. The need for this operation arose from the common Indian practice of amputating the tip of the nose as punishment for a variety of crimes ranging from robbery to adultery.4,5 The first reported use of the median forehead flap outside of India was by Antonio Branca of Italy. Based on an Arabic translation of the Sushruta Samita, Branca performed a nasal reconstruction using the midforehead flap in the 15th century.6,7 In the 16th and early 17th centuries, little advancement was made in the use of the median forehead flap because plastic and reconstructive surgery fell into disrepute.4,5,7 The flap had a revival in 1794, when J. C. Carpue read an editorial in the Gentleman’s Gazette of London describing the flap’s use for nasal reconstruction.8–10 Initially, Carpue practiced the median forehead flap operation on cadavers. Twenty years passed before he performed the operation on two patients. He reported his successful results in a monograph entitled “An Account of Two Successful Operations for Restoring a Lost Nose from the Integuments of the Forehead.” His article was widely circulated throughout Europe and served to popularize the operation.6,9 In the 1830s, Ernst Blausius (Chief of Ophthalmologic Surgery of Berlin), Johann Friedrich Dieffenbach (Chief of Surgery at Munich Hospital), and Natale Petrali of Milan simultaneously reported on uses and variations of the median forehead flap for reconstruction of the face and nose. On the basis of their influence as respected surgeons at large European teaching hospitals, the use and popularity of the forehead flap grew.6 In the late 1830s, use of the median forehead flap for nasal and facial reconstruction crossed the Atlantic when J. M. Warren performed the operation in the United States.4,11
By the early 1900s, the forehead flap was used to reconstruct losses of the nose secondary to battle, scrofula, syphilis, and cancer. Many American surgeons, such as Pancoast, Mutter, Buck, Davis, and Fomon, wrote about the use of the forehead flap in nasal reconstruction. Little modification of flap design, use, harvest, or donor site closure occurred until articles authored by Kazanjian appeared in the plastic surgery literature of the 1930s. This pioneer plastic surgeon was the first to determine that the primary blood supply of the flap is from the supratrochlear and supraorbital arteries. Kazanjian described a forehead flap designed precisely in the midline, allowing primary closure of the donor site. This technical modification minimized the forehead donor scar, which up to that point represented the major morbidity of the operation. Before Kazanjian’s modification, the donor site of the forehead flap had either been skin grafted or left open to heal secondarily by granulation and wound contraction. This practice commonly left the patient more scarred and disfigured than before the reconstructive surgery.12
Kazanjian carried incisions from the hairline to a point immediately above the level of the nasofrontal angle. By his time, surgeons had recognized that the use of unlined forehead flaps to repair full-thickness nasal defects predictably resulted in contraction of the flap and compromise of the nasal passage. The contour of the external nose also deformed as the airway constricted from progressive contracture of scar developing on the undersurface of the flap. In response to this, surgeons developed a number of modifications in the design of the forehead flap to achieve additional length so the flap could be folded on itself for internal lining. These designs included oblique and horizontally oriented flaps. Gillies13 described a U-shaped flap with an ascending and descending component that he called the up-and-down flap. The ascending portion of the flap was positioned over the axis of the supraorbital artery on one side and the descending portion over the contralateral supraorbital artery. Converse14 harvested lateral forehead skin based on a long pedicle of hair-bearing scalp, which became known as the scalping flap. The designs by Gillies and Converse circumvented the need to include hair-bearing scalp to provide additional length so that the flap could be folded on itself. Unfortunately, these modifications of the original design of the median forehead flap left marked deformities of the forehead. It also became apparent that folding the flap on itself created a great deal of tissue bulk that caused the nose to collapse. In addition, partial necrosis of the portion turned internally was common.
In the 1960s, Millard11,15 designed a large modified median forehead flap called the seagull flap, in which lateral extensions were used to cover the nasal alae. Incisions for the pedicle of the flap extended below the bony orbital rim to gain additional flap length. Millard also described methods of donor site repair and techniques for constructing nasal support, all of which improved the outcome of nasal reconstruction.
Labat7,16 was the first surgeon to design a median forehead flap with the base centered over a unilateral supratrochlear artery. He curved the incisions of the proximal pedicle so the base of the flap rested immediately above the medial brow and canthus on one side. This reduced the standing cutaneous deformity resulting from pivoting of the flap and increased the effective length, making more tissue available for reconstruction. Millard shifted the entire vertical axis of the central forehead flap to a paramedian position, demonstrating that the flap could survive without including the central glabellar skin in the pedicle.7 Menick modified Millard’s design of the paramedian flap by making the pedicle narrower.7 This offered a greater freedom of tissue movement, a smaller standing cutaneous deformity, and a longer effective length to the flap.
In the 1980s, Burget and Menick17–19 found that extending incisions for the pedicle of the paramedian flap below the bony orbital rim provided additional flap length. This often avoided the need to extend the flap to hair-bearing scalp for sufficient length to reach the nasal tip. They noted that the end arterioles of the supratrochlear artery are located immediately under the dermis, superficial to the frontalis muscle. The authors determined that the frontalis muscle could be safely removed from the distal flap without impairing the vascularity of the skin.1
Studies by Mangold, McCarthy, and Shumrick better defined the vascular anatomy of the forehead. In 1980, Mangold et al20 demonstrated that the blood supply to forehead skin is from the dorsal nasal (a terminal branch of the angular artery), supratrochlear, supraorbital, and superficial temporal arteries. Each of these vessels provides a primary blood supply to a particular region of the forehead, but all demonstrate numerous interconnecting anastomoses. Mangold’s injection studies and dissections of cadavers showed that the forehead could be divided into regions based on their predominant vascular supply (Fig. 13-1). On the basis of these vascular regions, Mangold determined that median and paramedian vertically oriented forehead flaps are nourished primarily by the supratrochlear artery and secondarily by the dorsal nasal and supraorbital arteries. McCarthy and others21,22 confirmed Mangold’s work in clinical experiences with patients. McCarthy injected the facial artery after ligation of the supraorbital and supratrochlear arteries and showed sufficient filling of the forehead vasculature to supply vertically oriented flaps in the region of the central forehead.21 This suggested that a paramedian forehead flap could survive even when the supratrochlear artery on the side of the flap is not present.
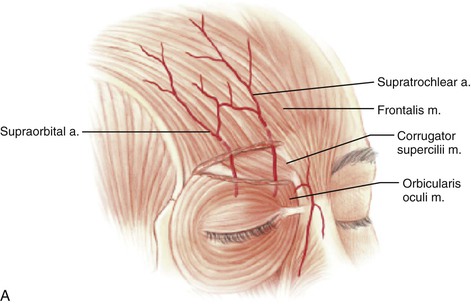
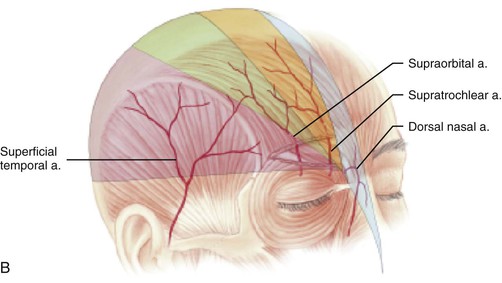
FIGURE 13-1 A, Supratrochlear artery exits orbit approximately 2 cm lateral to midline, passing under orbicularis oculi and over corrugator supercilii. At level of eyebrow, supratrochlear and supraorbital arteries pass through orbicularis and frontalis muscles and continue superiorly in superficial subcutaneous tissue plane. B, Vascular territories of arteries supplying forehead skin. (From Baker SR: Interpolated paramedian forehead flaps. In Baker SR, editor: Principles of nasal reconstruction, ed 2, New York, Springer, 2011.)
In 1992, Shumrick and Smith10 performed detailed anatomic studies of the forehead using techniques of latex injection, radiography, and microdissection to determine the precise vascular anatomy of the central forehead. Examination of the radiographic data confirmed a clinically apparent fact: the forehead region contains an intricate system of anastomosing vessels among the angular, supratrochlear, supraorbital, and superficial temporal arteries (Fig. 13-2). The paired supratrochlear arteries connected with each other by several horizontal unnamed arteries that cross the midline. Moreover, the supratrochlear arteries consistently demonstrated anastomotic branches with the angular and supraorbital arteries in the medial canthal region. Microdissection of the forehead vasculature confirmed these radiographic findings. The supratrochlear artery was consistently found to exit the superior medial orbit approximately 1.7 to 2.2 cm lateral to the midline and continued its course vertically in a paramedian position approximately 2 cm lateral to the midline. This position closely corresponds to the location of the medial border of the eyebrow. The supratrochlear artery was found to exit the orbit by piercing the orbital septum, passing under the orbicularis oculi and over the corrugator supercilii.10 At approximately the level of the eyebrow, the artery passes through the orbicularis and frontalis muscles and continues superiorly in the superficial subcutaneous tissue. This transition of the artery from a deeper to a more superficial tissue plane was confirmed by histologic examination of cross sections of forehead skin at various levels. Doppler examinations of healthy volunteers helped confirm the findings of the cadaver studies.10
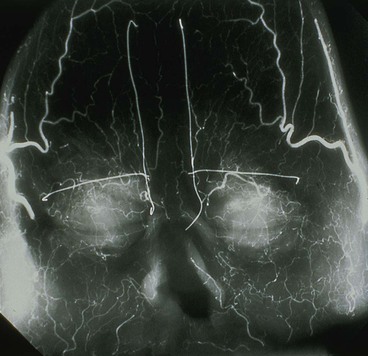
FIGURE 13-2 Radiograph showing skin vasculature after injection of contrast medium. Horizontal wire markers delineate superior bony orbital rims. Vertical wires mark medial end of eyebrows. Note intricate system of anastomosing vessels. (From Baker SR: Interpolated paramedian forehead flaps. In Baker SR, editor: Principles of nasal reconstruction, ed 2, New York, Springer, 2011.)
Studies of the vascular anatomy of the forehead confirm that the supratrochlear artery serves as the axial blood supply of median and paramedian vertically oriented forehead flaps. The studies also confirm a rich anastomotic network in the medial canthal region. Surgical techniques that preserve this regional blood flow have allowed surgeons to harvest paramedian forehead flaps based on pedicles narrower than those used for median forehead flaps. The narrower pedicle provides the flap with greater freedom of transposition about its pivotal point and greater effective length. The pedicle may be as narrow as 1.2 cm, reducing deformity of the inferior forehead area and always enabling the surgeon to achieve primary closure of the lower half of the donor defect (Fig. 13-3).7,23
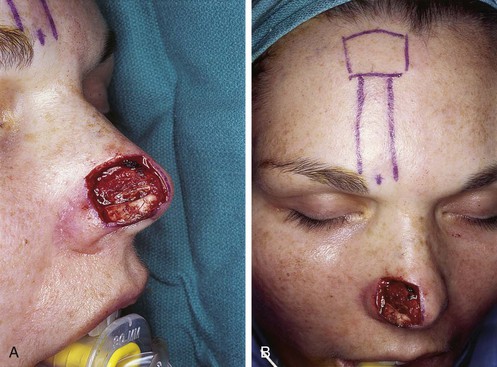
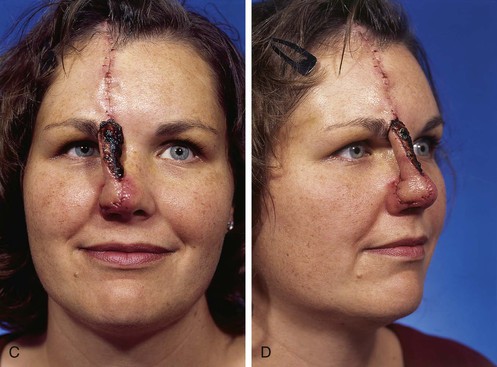
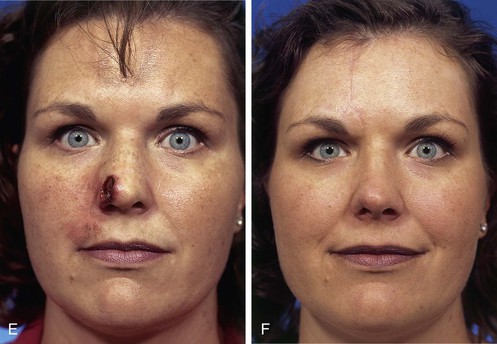
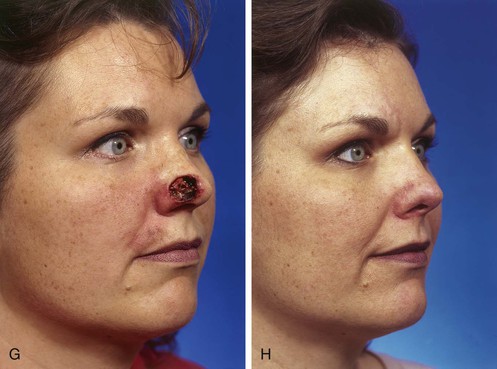
FIGURE 13-3 A, Heminasal cutaneous defect. Auricular cartilage graft placed along nostril rim for structural support. B, Paramedian forehead flap designed for reconstruction of nose. Centering of pedicle over vertical axis of supratrochlear artery enables narrow pedicle 1.2 cm wide. C, D, Narrow pedicle provides greater effective length and smaller standing cutaneous deformity than wider pedicle. E-H, Preoperative and 1.5-year postoperative views. No revision surgery performed.
Paramedian Forehead Flap
Midforehead flaps, which include the median and paramedian flaps and their many variations, have proved to be dependable flaps for midfacial reconstruction.2,3,24 However, the paramedian forehead flap based on a single supratrochlear artery has replaced the median forehead flap for nasal reconstruction because it has a more axial design, narrower base, and greater effective length. The design also enables the simultaneous or sequential use of two vertically oriented forehead flaps. The paramedian flap has an abundant blood supply, providing for revascularization of cartilage and bone grafts covered by the flap. Removal of muscle and subcutaneous fat from the distal portion can make the paramedian forehead flap thin, pliable, and easily contoured to fit any defect of the nose. Maintaining the attachment of the frontalis muscle to the flap is occasionally useful when more bulk is required to fill defects of considerable depth.25 In most instances, however, freeing the flap from the frontalis muscle and most of its subcutaneous fat is essential when the forehead flap is used as covering for the nose. Proper thinning of the flap will ensure that the shape and contour of the underlying nasal framework are visually manifested. Flap thinning may be accomplished without concern for compromise of the flap’s vasculature because the supratrochlear artery travels superiorly in the subcutaneous/subdermal tissue plane from a point 1 cm superior to the level of the eyebrow.10 The superficial location of the artery enables thinning of the distal portion of the flap by removal of the fascia and frontalis muscle and, if necessary, nearly all of the subcutaneous tissue. The ability to modulate the thickness of a flap so that it may exactly match the thickness of the nasal defect at the recipient site greatly enhances the aesthetic result.25
Paramedian forehead flaps used as interpolated flaps for nasal reconstruction require a second operation to separate the pedicle.26 The pedicle may safely be detached as soon as 10 to 14 days after flap transfer. The author prefers a 3-week interval between the time of flap transfer and flap inset for all patients, especially those who use tobacco products. At 3 weeks, the distal portion of the flap has developed sufficient collateral blood supply from the nose to allow thinning and sculpturing of the more proximal portion of the flap left attached to the recipient site. If the nasal defect is small, the portion of the forehead flap covering the defect may be completely thinned at the time of initial transfer.
A minor disadvantage of the paramedian forehead flap is the donor site scar and the necessity for a two-stage procedure. However, forehead scars are rarely unsightly because primary wound closure is easily achieved in the inferior portion of the forehead owing to the narrow pedicle required (Fig. 13-4). Although the narrow pedicle enables primary repair of the inferior aspect of the donor site wound, the width of the superior portion of the flap may be several centimeters, precluding complete wound approximation. In general, flaps wider than 4.5 cm are too large to allow complete primary closure of the donor site. In such circumstances, the superior portion of the donor site wound must, in part, be left to heal by secondary intention. Fortunately, the resulting midline or paramedian scar in the superior portion of the forehead is extremely forgiving and only occasionally requires revision. The acceptable scar is related to the immobile skin and convex contour of the superior central forehead (Fig. 13-5).
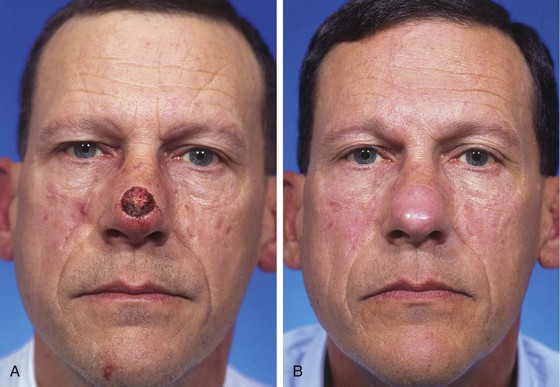
FIGURE 13-4 A, A 1.5 × 1.8-cm cutaneous defect of nasal tip. B, At 8 months after repair of defect with paramedian forehead flap. C, Donor site forehead scars are rarely unsightly. No revision surgery performed.
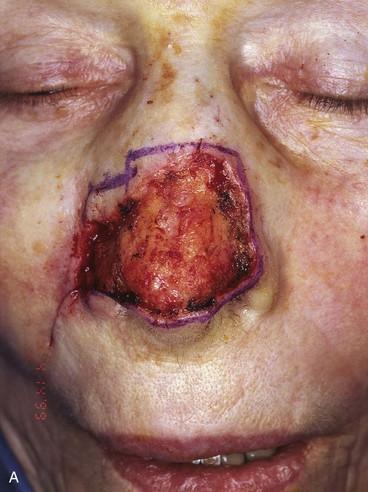
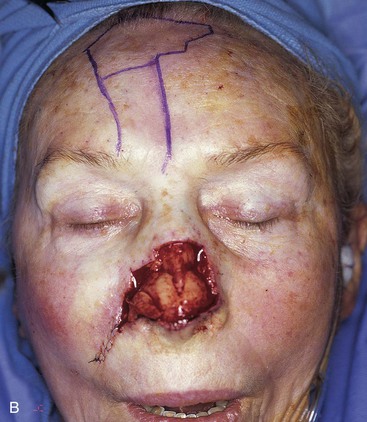
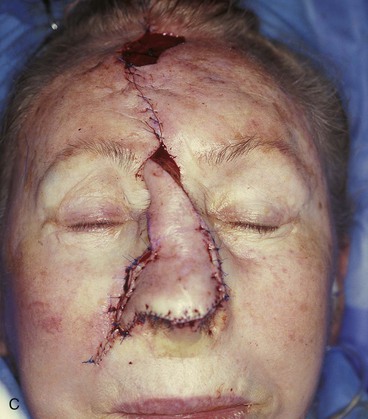
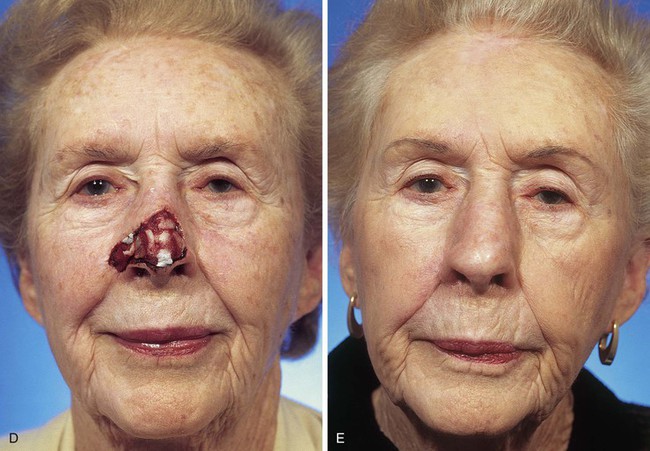
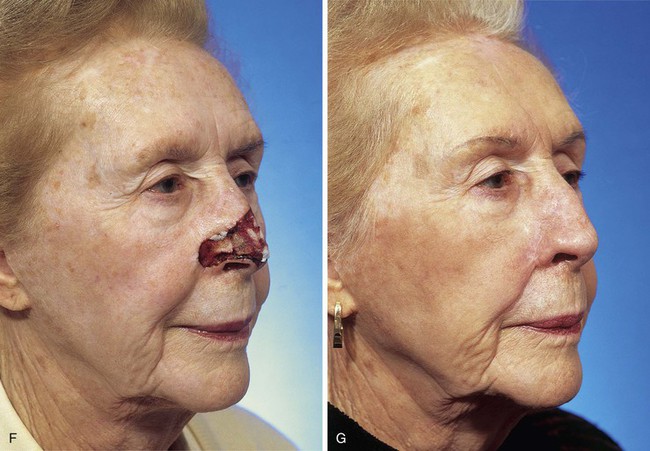
FIGURE 13-5 A, A 5 × 4-cm cutaneous defect of nasal tip, caudal dorsum, and right ala. B, Paramedian forehead flap designed for repair. C, Flap in place. Flaps wider than 4.5 cm are too large to allow complete wound closure of donor site. Portion of superior donor site wound left to heal by secondary intention. D-G, Preoperative and 9-month postoperative views. Contouring procedure to restore alar groove performed 6 months postoperatively. No revision surgery performed on forehead scar.
The fine vellus hair that is prominent in some patients just in front of the forehead hairline is even more difficult to eliminate from a forehead flap because the hair follicles are not visible by the human eye and are located in the dermis rather than in the subdermal plane. This hair may be treated by electrolysis with limited success. The best treatment is to have the patient periodically use a depilatory cream for removal. Laser hair removal may be used in place of electrolysis for dark-pigmented scalp hair, but it is not effective for vellus hair.
To avoid multiple procedures directed at depilation, paramedian forehead flaps should be designed not to include scalp hair whenever possible. Avoiding scalp hair may be possible by extending the incision for the pedicle through the eyebrow to the level of the bony orbital rim. The pedicle is skeletonized on the soft tissue surrounding the supratrochlear artery as it exits the orbit. This requires complete sectioning of the corrugator supercilii to achieve free tissue movement. If this step is unlikely to lend sufficient length to the flap, another helpful approach is to obliquely angle the flap from the midline laterally toward the temporal recession just beneath the hairline (Fig. 13-6).27,28 This modification in design of the paramedian forehead flap is possible only for smaller flaps measuring less than 3 cm in maximum width. Harvesting of flaps 3 cm or greater in width from the lateral portion of the forehead may occasionally cause more apparent scars than centrally located donor sites. It may also cause excessive upward displacement of the central portion of the eyebrow on the donor side. Thus, flaps wider than 3 cm are usually extended to the hair-bearing scalp rather than designed in an oblique fashion when the necessary length of the flap requires such extensions (Fig. 13-7). When the oblique forehead design is used, the distal portion of the flap does not have the advantage of an axial vascular pattern. However, the author has been successful in transferring forehead skin based on a supratrochlear artery but positioned several centimeters lateral to the axis of the artery. This success is related to the rich vascular anastomotic network of forehead skin.
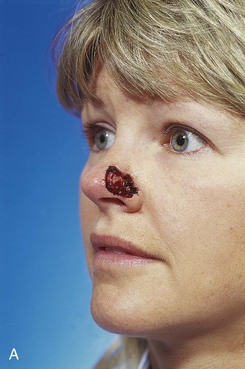
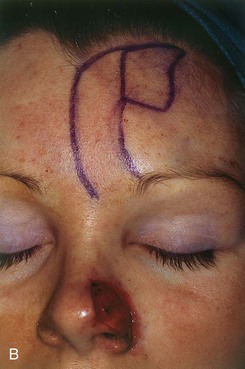
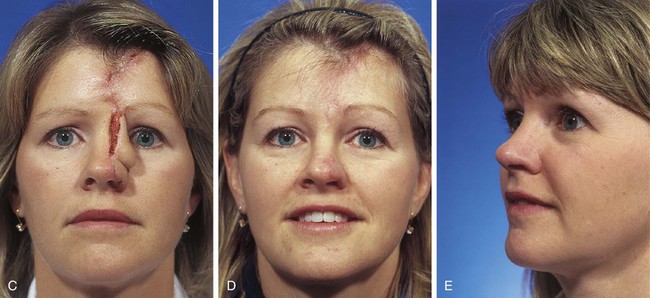
FIGURE 13-6 A, A 2 × 1-cm cutaneous defect of nasal tip and ala. B, Paramedian forehead flap designed for repair. Distal flap angled toward temporal recession beneath anterior hairline to provide additional length to flap while avoiding transfer of scalp hair to nose. C, Flap in place. D, At 2 months after flap inset. E, Postoperative view at 1.5 years. No revision surgery performed.
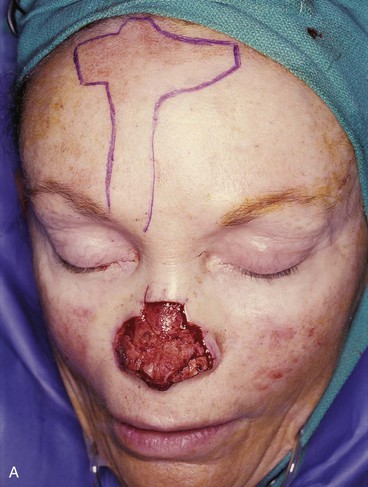
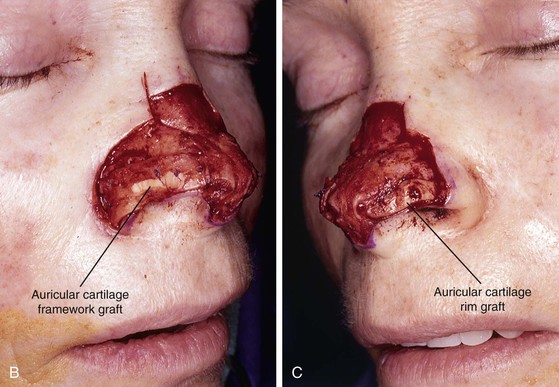
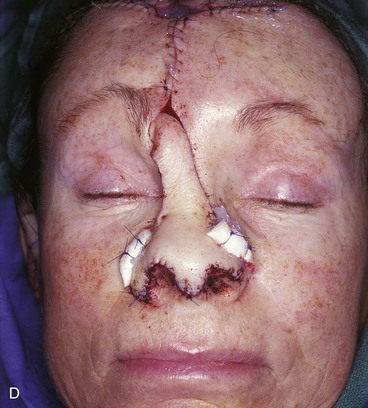
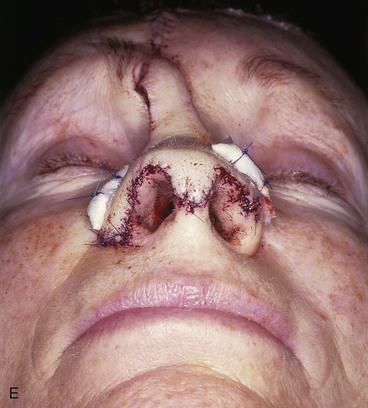
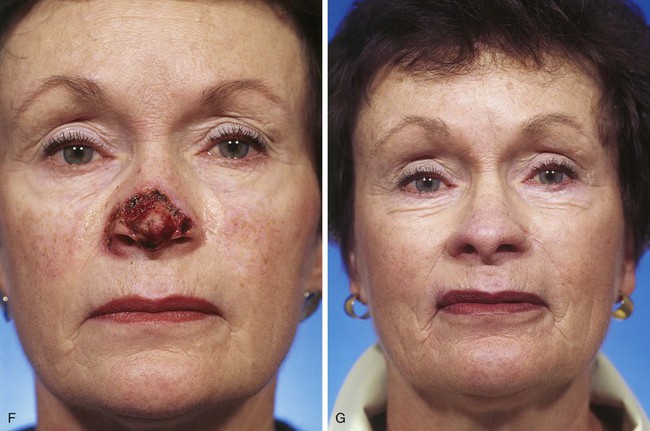
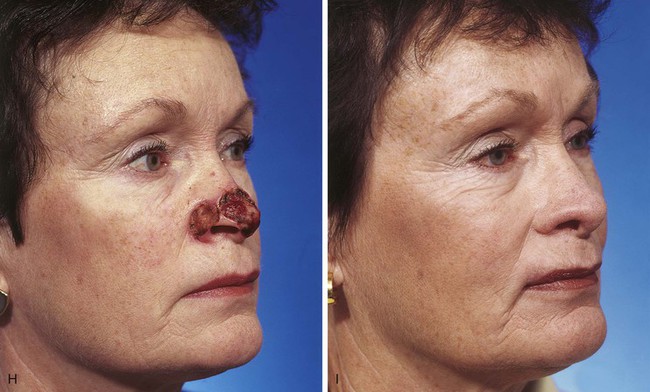
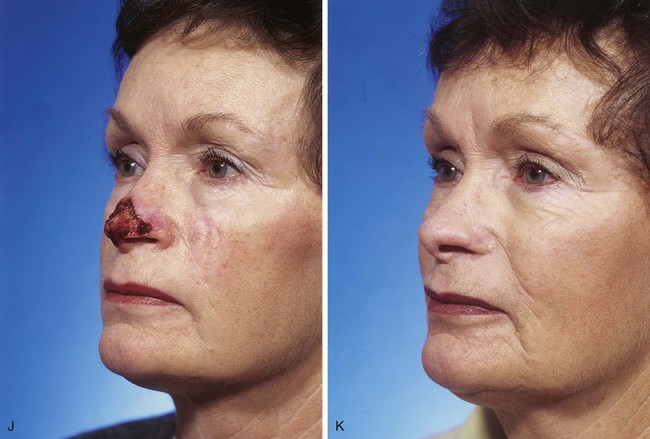
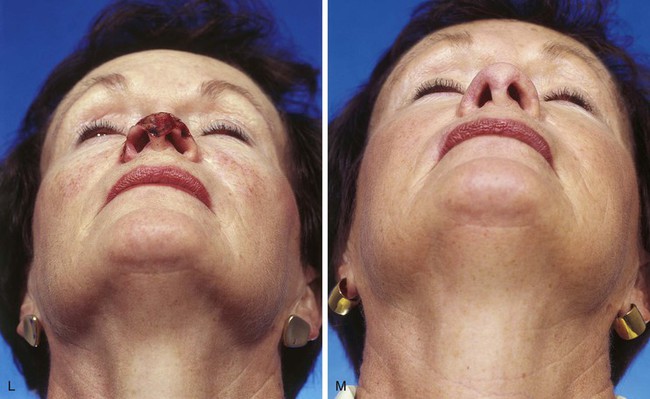
FIGURE 13-7 A, A 5 × 2-cm cutaneous defect of nasal tip, caudal dorsum, and right ala. Paramedian forehead flap designed for repair. Size of defect necessitated extending forehead flap into hair-bearing scalp. B, C, Auricular cartilage grafts in place to provide structural support to right ala and left nostril margin. D, E, Flap in place. Portion of superior donor site wound left to heal by secondary intention. Bolster dressings used to appose flap to cartilage grafts. F-M, Preoperative and 5-year postoperative views. Contouring procedure and nasal base reduction performed.
Historically, the paramedian forehead flap has been used for reconstruction of larger defects of the nose. It is the preferred method for covering nasal defects too large to be repaired with full-thickness skin grafts, nasal cutaneous flaps, composite auricular grafts, or interpolated melolabial flaps.4,5,12 In general, nasal defects larger than 2 cm in width in the horizontal axis are best repaired with a paramedian forehead flap (see Fig. 13-7).12 In addition, this flap is best for repair of nasal defects with exposed bone and cartilage deficient of periosteum or perichondrium and in instances in which the central face has been irradiated.5,12
Surgical Technique
The cutaneous defect is outlined by squaring off the corners of the defect with a skin marker. Giving the defect angular rather than curvilinear borders reduces the propensity for development of trap-door deformity (see Fig. 13-5). If the defect occupies more than 50% of the surface area of a convex-shaped nasal aesthetic unit (tip and alae), the remaining skin of the unit is marked for removal (Fig. 13-8). When a unilateral cutaneous defect encompasses half or less of the surface area of the nasal tip, the defect is usually enlarged only to the degree that the enlargement creates a hemi-tip defect (Fig. 13-9). This will limit the size of the forehead flap necessary for reconstruction, which in turn minimizes the deformity of the forehead. The aesthetic result of resurfacing a heminasal defect with a paramedian forehead flap is as pleasing as resurfacing the entire nasal tip, provided the remaining nasal skin is of thin or medium thickness.
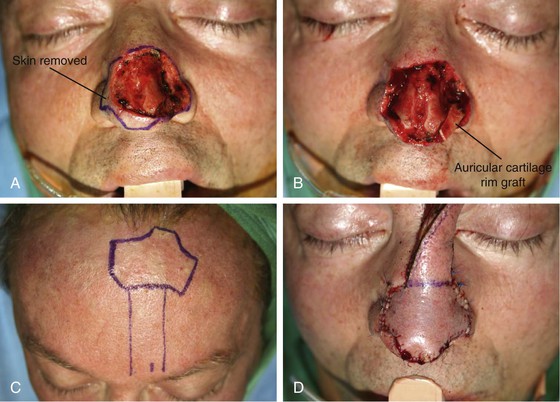
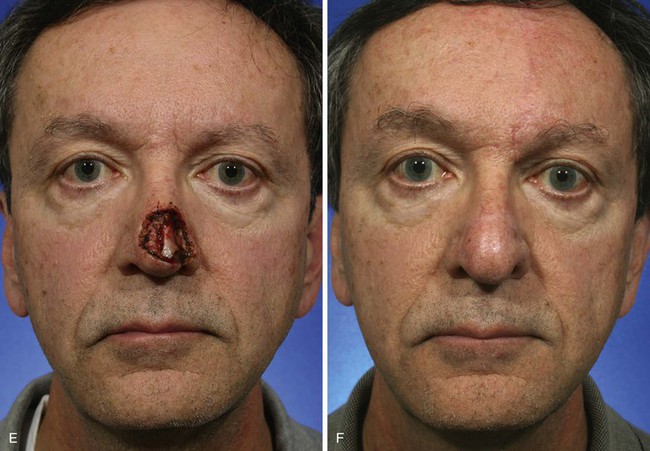
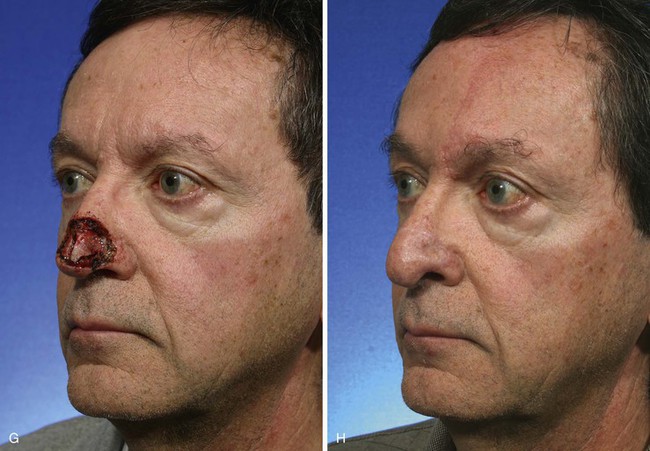
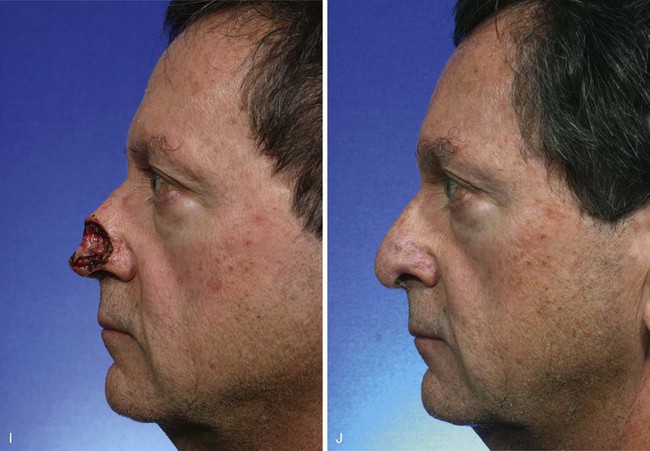
FIGURE 13-8 A, A 3 × 2.5-cm cutaneous defect of nasal tip and small portion of left ala. Defect involves left nostril margin. Remaining skin of tip marked for excision. B, Skin excised. Auricular cartilage rim graft in place to prevent nostril retraction. C, Paramedian forehead flap designed for repair. D, Flap in place. E-J, Preoperative and 9-month postoperative views. Patient had contouring procedure and forehead scar revision 7 months postoperatively.
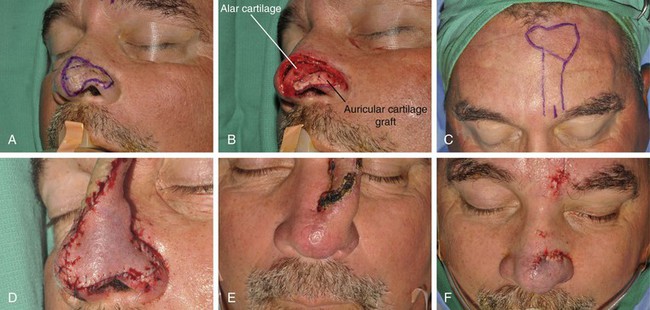
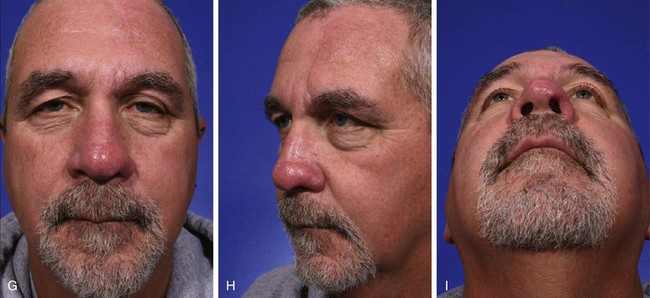
FIGURE 13-9 A, A 2.8 × 3.5-cm melanoma of nasal hemi-tip marked for excision. B, When a unilateral skin defect encompasses half or less of surface area of nasal tip, defect is usually enlarged only to create hemi-tip defect. This will limit size of forehead flap necessary for reconstruction, which in turn minimizes deformity of forehead. Ear cartilage graft in place for structural support. C, Paramedian forehead flap designed for repair. D, Flap in place. E, F, Flap inset 3 weeks after flap transfer. G-I, Postoperative views at 5 months. No revision surgery performed.
An exception to the rule of resurfacing the entire nasal unit when the defect occupies half or more of the surface area of the unit is the dorsum. When cutaneous defects occur on the dorsum, the thin skin of the rhinion is never removed unless it is required for tumor ablation. In most patients, the intrinsic thickness of forehead skin is greater than that of the native skin covering the rhinion. Even with removal of all the subcutaneous tissue from a
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


