Chapter 12 Fiber Reinforcement
Section A Direct Fiber-Reinforced Restorations
Clinical Considerations
Beam Theory
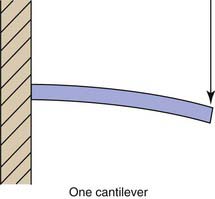
Any understanding of FRC bridges must begin with beam theory. A beam is defined as a structural member that is subjected to loads applied transverse to the long axis. The simplest type of beam is known as a cantilever (Figure 12-1). This concept, familiar to most dentists, refers to a beam supported and retained at one end, called encastré by engineers. Near the middle of any beam is a line (or a plane in three dimensions) known as the neutral axis. This axis coincides with the centroid or geometric center of the beam. The longitudinal stresses along this line are zero. The stresses in a cantilever beam are essentially tensile above this line and compressive below it. The farther from the neutral axis, the greater the stress. This explains the success of I-bar beams, as the greatest strength of the beam structure is concentrated farthest from the neutral axis, where there is the greatest stress.
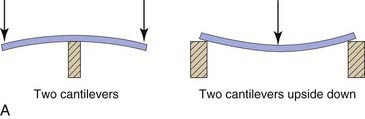
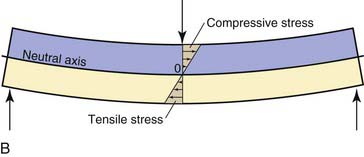
The second type of beam is a simply supported beam. This type corresponds to a board laid across a puddle. A simply supported beam is essentially two cantilevers turned back to back and upside down (Figure 12-2, A). The stresses on it are compressive above the neutral axis and tensile below it (Figure 12-2, B).
Treatment Considerations
Clinical Techniques
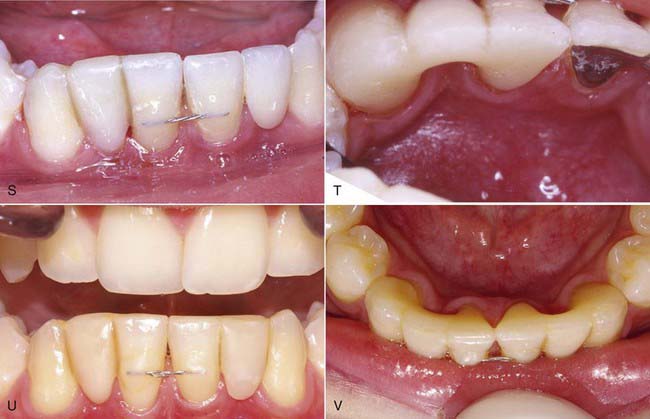
Anterior Bridge Technique
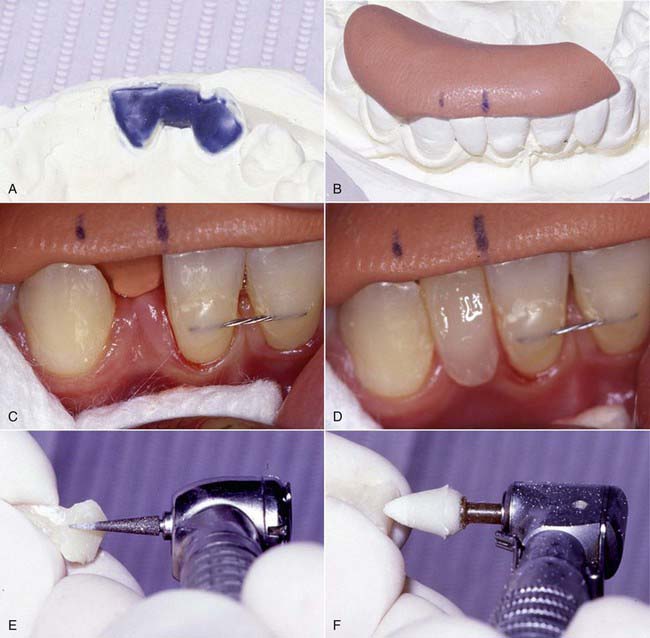
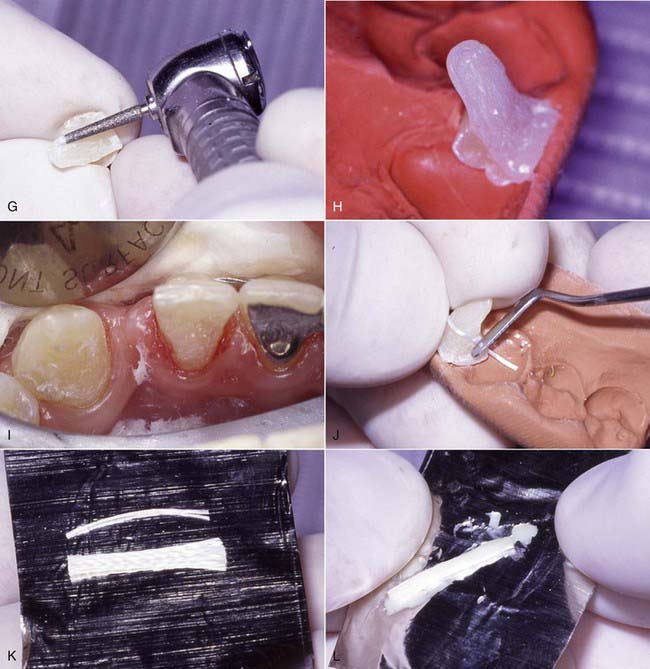
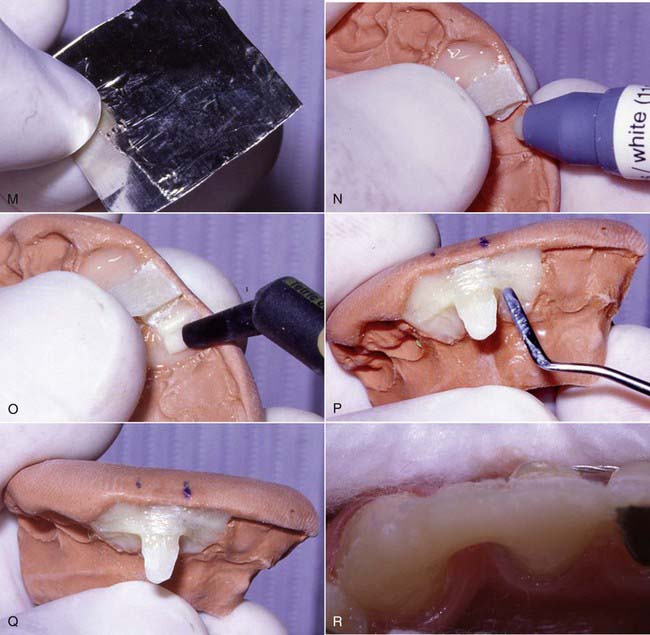
The technique for direct intra-oral fabrication of an anterior FRC bridge begins with a set of study models. The model is modified with wax or composite to create the lingual contours desired of the completed bridge (Figure 12-3, A). The author typically uses composite to mock up the lingual portion of the pontic and then flows inlay wax onto the lingual surface of the abutment teeth on the models to simulate the thickness of the retainer and the fibers on these teeth. The labial surface of the pontic is of no concern at this stage; it will be developed in the mouth when the bridge is fabricated.
A matrix is then fabricated using clear polyvinyl impression or bite registration material. This material is mixed according to the manufacturer’s instructions and applied to the lingual surface of the model to register the contours developed in the mock-up (Figure 12-3, B). Opaque matrix material can be used as shown in Figure 12-3, B, but transparent materials allow for the structure to be polymerized through the matrix after the initial trans-enamel polymerization, which ensures all the composite will fully gel before the matrix is removed.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses