Chapter 10
Skin Rejuvenation
Introduction
The most notable change in the field of cosmetic surgery is the realization that the skin must also be rejuvenated in order to truly achieve a more complete rejuvenation of the patient. This change is reflected in the large increase in the number of cosmetic nonsurgical procedures while there has been a decline in cosmetic surgical procedures. The interest in skin rejuvenation has been the catalyst for the technological advances in the filler, laser, and radiofrequency industry. A well-balanced approach to skin rejuvenation takes into consideration both the skin care regimen that the patient uses at home and the judicious use of skin rejuvenation procedures in the office.
Anatomy of the Skin
Physiologically, the skin serves many important roles including barrier protection against environmental insult (ultraviolet radiation [UVR], heat, and cold), nutrient and electrolyte balance, sensation, and immunological host defense.1 The appearance of the skin may reveal underlying health problems, thus affecting social status. For example, some vitiligo patients in India are incorrectly feared to have leprosy and are ostracized.2 The feel and visual appearance of the skin is also involved in sexual attraction.
The skin is divided into three anatomical layers: the epidermis, the dermis, and the subcutis (Figure 10.1). While each layer has distinct functions, there is constant interaction between the different layers. The following is a brief overview of cutaneous anatomy for the nondermatologist. However, for greater detail, we recommend a good dermatopathology or histopathology text.
Figure 10.1 Anatomy of the skin.
(Reproduced from Obagi S, Bridenstine J. Lifetime skincare. Oral and Maxillofacial Surg Clin North Am 2000;12(4):531–40.)
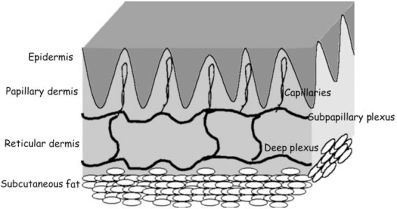
Epidermis
The epidermis is the outermost layer of the skin and consists of terminally differentiated, stratified squamous epithelium (keratinocytes, 80–90% of cells), melanocytes, unmyelinated axons, Langerhans cells, and Merkel cells, which are neuroendocrine in nature.3 The epidermis varies in thickness from less than 0.07 to 0.17 mm based on anatomical location, with the thinnest area on the eyelid and the thickest on the back.4 It is composed of four layers, stratum basale (lowermost layer), stratum spinosum, stratum granulosum, and the stratum corneum (uppermost layer). The cells that regenerate the epidermis reside in the stratum basale (basal layer). The stratum spinosum and stratum granulosum consist of spindled keratinocytes, interconnected by many desmosomes, actively synthesizing keratins, keratohyaline granules (the “glue” for keratin filaments), and Odland bodies that contain neutral sugars linked to lipids and proteins. The Odland bodies’ contents are released into the intercellular space by the granular cells forming a barrier to water loss and mediating stratum corneum cell cohesion.5,6 The stratum corneum consists of anucleate cells that contain macrofibrils of keratin that cross-link to the cell membranes creating a cornified envelope. This cornified envelope protects the epidermis from environmental damage and consists of 8–20 nm of insoluble protein overlaid by a 5-nm lipid envelope containing ceramides, cholesterol, and free fatty acids.7 The epidermal turnover time, defined as the time it takes a cell from the basal layer to reach the stratum corneum and be exfoliated, is 52–75 days depending on anatomical location. It is important to recognize this issue when various skin care products are discussed.
Adnexal structures such as hair follicles, sebaceous glands, apocrine glands, and eccrine glands arise embryologically from the epidermis and grow downward into the dermis. The presence or density of these structures varies with anatomical location, with the face having more adnexal structures than the neck or chest. This must be taken into consideration when resurfacing the skin since the wound reepithelializes from these adnexal structures.
Dermal–Epidermal Junction
The dermal–epidermal junction (DEJ) connects the epidermis and dermis. It consists of the basement membrane, anchoring fibrils, and the hemidesmosome-anchoring filament complex. The DEJ supports the epidermis, allows for cell signaling and protects against external shearing forces by creating an interface between the epidermis and dermis.
Dermis
The dermis is located between the epidermis and subcutaneous fat. It supplies nutrients to the epidermis and appendages while protecting against mechanical injury.8 There are anatomical differences in dermal thickness, with the thinnest at less than 0.5 mm on the eyelids and the thickest at over 4 mm on the back.
Muscular arteries that penetrate through the subcutaneous fat provide the blood supply to the dermis. They form a horizontal plexus in the deep reticular dermis. From this plexus, ascending arterioles extend toward the epidermis where they form a second, superficial plexus, the subpapillary plexus. Capillaries project from the subpapillary plexus into the papillary dermis.
The dermis is divided by the subpapillary plexus of vessels into the papillary dermis (fingerlike projections interspersed between the epidermal rete ridges that project down into the dermis) and reticular dermis. The papillary dermis consists of small-diameter collagen fibrils and elastic fibers, whereas the reticular dermis is made up of large-diameter collagen fibrils organized into thick bundles surrounded by mature elastic fibers.
The connective tissue matrix is the supporting framework of the dermis. It is composed of collagen (mainly Type I), elastin, proteoglycans (PGs), glycosaminoglycans (GAGs), and glycoproteins. Collagen, produced by fibroblasts, provides the dermis with both tensile strength and elasticity. Elastin allows stretched or deformed skin to return back to its normal shape. PGs and GAGs constitute the ground substance of the dermis. They have the ability to bind up to 1000 times their volume in water.
Subcutis
The subcutis is composed of fat lobules, blood vessels, and connective tissue. The subcutaneous fat layer serves as a cushion for the skin, a source of reserved energy, and cosmetically molds the skin over the curves of the body.
Aging of the Skin
The complex process of skin aging is comprised of two distinct components, intrinsic aging (chronological) and extrinsic aging (photoaging).9 Intrinsic aging is the biological result of cellular senescence, while extrinsic aging is caused by environmental damage to the skin. While intrinsic aging is for the most part inevitable, the extent of extrinsic aging is highly affected by a person’s cumulative exposure to UVR and smoking.
Intrinsic Aging
As the skin ages, there is a decrease in normal cell function. Clinically, this appears as dry, wrinkled, fragile, and lax skin. At the cellular level, there appears to be an age-related impairment in signal transduction10 and a decrease in cell doubling exhibited by melanocytes,11 fibroblasts,12 and keratinocytes.13 Additionally, a large portion of senescence is related to the shortening of telomeres (the terminal portion of eukaryotic chromosomes) with each population doubling. Studies have shown a 30% shortening of the telomeres of adult fibroblasts,3 while telomeres of cancer cells (immortalized) do not become progressively shorter.14 In addition, cumulative oxidative damage of both nuclear15 and mitochondrial DNA16 is implicated in cellular senescence.
Within the dermis, production of cytokines and extracellular matrix-modifying enzymes such as elastase, collagenase, stromelysin, and interleukin (IL)-1 are increased.17 These enzymes disrupt normal tissue integrity resulting in the clinical stigmata of aging skin: wrinkling, dermal fragility, and dermal thinning.
These cellular changes can be assessed histologically. There is thinning of the epidermis with flattening of the epidermis at the DEJ and effacement of the dermal papillae and epidermal rete pegs, thus impairing nutrient transfer and making the skin more susceptible to trauma from shearing forces.18 Dermal collagen production declines by 1% per year in adult life and there is an associated decrease in elastin production as well.19 This results in a decrease in dermal thickness that may play a role in the dermal fragility and delayed wound healing seen in elderly skin. While there is no histological correlate for superficial or fine wrinkles, broadening and shortening of subcutaneous fat connective tissue septae may contribute to deep furrows in the skin.20 A deficiency in lipid synthesis, particularly in cholesterologenesis, leading to decreased lipid content in the stratum corneum, contributes to impairment of the skin barrier function seen in aging skin.21
Extrinsic Aging
The majority of skin changes attributed to “aging,” including dyspigmentation, premature wrinkling, coarseness, actinic keratoses, and certain types of skin cancers, are actually a result of photodamage. Other factors, particularly smoking, can intensify these changes.22
While intrinsic aging contributes to a gradual decrease in dermal thickness with age, studies show a decrease in dermal thickness in sun-exposed, versus sun-protected, sites.23,24 Same-donor keratinocytes and fibroblasts exhibit shorter in vitro life spans in sun-exposed skin when compared with sun-protected sites.25,26 Additionally, the propensity of sun-damaged skin to develop cutaneous malignancies invites speculation of a causative role for UVR-induced mutations of tumor suppressor genes27 and a reduced DNA repair capacity.28
Photodamaged skin has specific histological findings distinct from intrinsically aged skin. The epidermis may be atrophied (late stage) or thickened with atypical keratinocytes (actinic keratoses). The most significant findings are seen in the dermis. Collagen and elastin degeneration, due to photodamage, results in deposition of an abnormal elastotic material (solar elastosis).29 In addition to elastin alteration, there is degradation of the collagenous meshwork by matrix metalloproteinases (MMPs).30 Additionally, with increased age, there appears to be an increase in GAGs in the skin, which is counterintuitive given the weathered skin appearance of elderly skin when compared with the well-hydrated skin of a younger individual. This may be accounted for by recent findings that the GAGs in photodamaged skin are deposited on the solar elastotic material rather than between collagen and elastic fibers as in normal skin.31,32 Melanocyte density and activity has been found in some studies to be decreased,33 while other studies have found increased melanocyte density.34 This may be related to inherent differences between the two populations studied with regard to skin color.
Treatment of Aging Skin
The goal of any good skin care regimen is to return the skin to a normal or healthy state and to prevent against future skin pathology. Returning the skin to a healthy state requires identification of the pathological process, recognizing the level of the pathology in the skin, and selecting the correct combination of products to reach this goal. However, choosing the right medicaments alone will not correct the problems without patient compliance. Educating the patient on the correct use of products and any anticipated irritation is paramount to achieving this goal.
A good skin care program can also prevent skin pathology by slowing the development of rhytides, lentigines, laxity, and malignancy. By instituting this regimen in our young patients, the progression of extrinsic aging can be delayed. This regimen should consist of an agent to promote collagen synthesis and maintain even epidermal thickness and pigmentation while preventing further environmental insult from oxidation and UVR.
Retinoids
The topical use of synthetic retinoids has been the most important development in skin rejuvenation (Table 10.1). Natural retinoids are produced by the human body through oxidative metabolism of vitamin A (retinol), and are present in low levels in the blood. The two main metabolic derivatives of vitamin A, retinoic acid (all-trans-retinoic acid and 13-cis-retinoic acid) and retinaldehyde, are important for growth, differentiation, and maintenance of epithelial tissues.35 They promote epidermal normalization and proliferation.
Table 10.1 Topical retinoids
| Topical retinoids | Strength (%) | Indications |
| Retinyl palmitate | 0.5–5.0 | Cosmetic products |
| Retinyl aldehyde | 0.05 | Cosmetic products |
| Tretinoin | 0.025–0.1 | Photodamage, acne, dyspigmentation |
| Adapalene | 0.1–0.3 | Acne |
| Tazarotene | 0.05–0.1 | Photodamage, acne, dyspigmentation |
The majority of retinoids interact with specific cytosolic retinoic acid binding proteins (CRABPs)36 and nuclear receptors to exert their effect. In the skin, the two main nuclear receptors are retinoic acid receptors (RARs) and retinoic X receptors (RXRs).37
Retinoids produce epidermal proliferation, loss of keratinocyte atypia, compaction of the stratum corneum (transient), and even distribution of melanosomes in keratinocytes.38,39Dermal effects of retinoids are seen as an increase in collagen deposition in the subepidermal region, decreased collagen breakdown (inhibition of collagenases such as MMPs), decreased solar elastosis, and increased GAG production.38 Additionally, topical application of all-trans-retinoic acid has been shown to protect the skin against damage from ultraviolet B rays (UVB)40 and ultraviolet A rays (UVA).41 Long-term topical retinoid use has not shown any risk of malignant degeneration.42
Topical retinoids are routinely used in the treatment of acne, epidermal melasma, photoaged skin, pre- and postskin resurfacing regimens, and intrinsic aging (Table 10.2). It is recommended that topical all-trans-retinoic acid (tretinoin) 0.05–0.1% be applied to the entire face on a nightly basis. Once the patient has become satisfied with the clinical results, application frequency can be decreased to three times weekly to maintain the results.43
Table 10.2 Skin care regimens
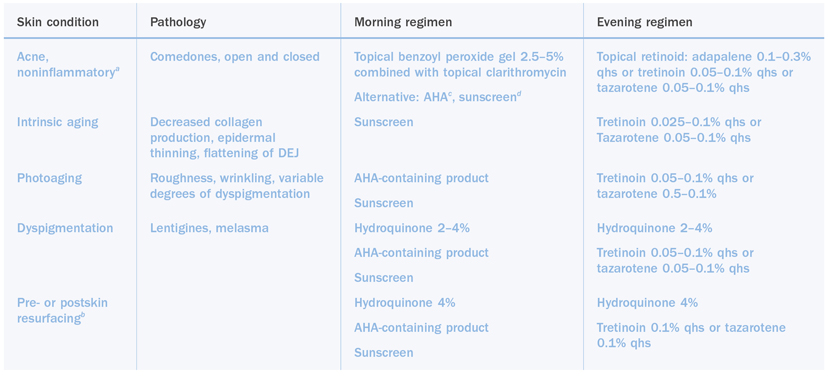
a Inflammatory acne, cystic or scarring, will require systemic agents in addition to the topical medications.
b Optimally, preresurfacing regimens should be initiated at least 6 weeks preoperatively. Postresurfacing regimens are initiated upon full reepithelialization of the skin.
c AHA: alpha-hydroxy acid; should be at least 8%.
d All sunscreens should be at least 30 SPF.
Mild irritation, in the form of erythema, xerosis, mild scaling, and slight stinging, should be anticipated when patients begin a retinoid-containing regimen. This should peak by 2–3 weeks then gradually subside. This is not an allergic reaction as many patients fear, but an indicator that the retinoid is taking effect.
Instructions should stress application of the tretinoin to the face, extending to the hairline, and feathering down along the jawline. The corners of the eyes and mouth should be avoided; however, both the upper and lower eyelids should be treated. The upper eyelids can be treated two to three times per week, while the lower eyelids can be treated almost nightly. Since the effects of retinoids are dose dependent, patients will experience greater improvement with higher concentrations (0.1% vs. 0.05%), daily use, and the application of up to 1 or 1.5 g a day.44
For patients with dyspigmentation, a topical hydroquinone (2–4%) should be applied to the entire face twice daily.45 It is important not to “spot” treat the skin to avoid developing a blotchy complexion. The evening application should be applied prior to tretinoin. The addition of an alpha-hydroxy acid (AHA) cream in the morning will enhance penetration of both the retinoid and the hydroquinone by creating a thinner stratum corneum. Evening application of AHAs is not recommended since they may alter the pH of the skin thus making tretinoin less active.
In recent years, the development of synthetic third-generation retinoids, tazarotene and adapalene, has expanded our treatment options of photodamaged skin. Once daily application of 0.1% tazarotene has shown efficacy comparable with that of 0.05% tretinoin emollient cream in clinical and histological improvement in fine wrinkles and pigmentary mottling of facial skin.46 Adapalene 0.1% gel, developed for the treatment of acne, has demonstrated clinical lightening of solar lentigines with twice daily application. However, a recent study of hairless mice demonstrated greater improvement in dermal thickness with the use of 0.05% tretinoin over that of 0.1% adapalene-treated skin.47 Therefore, at this time, the authors recommend the use of tretinoin or tazarotene for photodamaged skin.
Alpha-Hydroxy Acids
The AHAs are a family of acids used as exfoliating agents due to keratolysis, their ability to disrupt keratinocyte adhesion. They have been used for centuries (washing the face with wine or sour milk to soften the complexion) but have recently experienced resurgence in their popularity. The AHAs are as follows: lactic (milk), glycolic (sugar cane), malic (apple), citric (citrus fruit), and tartaric (grape).1These acids are mainly used for superficial conditions such as roughness, acne, mild dyspigmentation, and fine wrinkles. While widely promoted as antiwrinkle agents, to truly achieve clinical improvement, these products must contain a high enough concentration of AHA (at least over 5%) to be effective.48 It is notable that most over-the-counter products contain less than 5% AHA.
Although AHAs are routinely used in clinical practice for improvement of superficial skin abnormalities, there is a lack of clinical, double-blinded, randomized studies with histopathological correlation. There is some evidence from in vitro studies that glycolic acid increases collagen production by fibroblasts49; however, the clinical significance of this finding is yet to be determined. In fact, an in vivo study comparing glycolic acid 10% with tretinoin 0.05% showed improvement in collagen production only in the tretinoin-treated group.50
Until well-designed long-term clinical studies are performed that evaluate clinical and histopathological changes, it is difficult to scientifically select the correct strength of an AHA-containing cream. Based on the available clinical studies, the authors recommend the application of at least an 8% AHA product on a daily basis, preferably in the morning.51 Furthermore, it seems that concentration of the acid is more important than the type of AHA used. The comedolytic effects of an AHA cream will benefit patients with mild acne, while patients with photodamage (roughness, dyspigmentation, and superficial wrinkles) may notice a gradual improvement with regular use. Solo therapy with AHA creams, however, is probably suboptimal.
Skin Lightening Agents
Hydroquinone
Hydroquinone has been used for over 40 years to treat acquired disorders of hyperpigmentation. Its mechanism of action is through inhibition of tyrosinase, an enzyme exclusive to melanocytes and essential in the melanin synthesis pathway of melanocytes. Contrary to popular opinion, it does not bleach melanin, but is thought to prevent the production of new melanin. At low concentrations this is a reversible effect, while some studies have shown that 3% hydroquinone can be toxic to melanocytes.52 Because of recent safety concerns, over-the-counter sale of hydroquinone has been banned in the United States and Europe. This has been primarily due to the possibility of mutagenicity and carcinogenesis related to hydroquinone as a derivative of benzene.53 However, at this time, there has been insufficient evidence to classify hydroquinone as a carcinogen and it remains an effective option in the treatment of pigmentary disorders.54
Hydroquinone plays a very important role in skin care for several reasons. In strengths of 2–4%, it can be used as a sole agent in the treatment of hyperpigmentation disorders such as melasma,45 solar lentigines, and postinflammatory inflammation. Correctly used prior to and after skin resurfacing modalities, it can help minimize the development of postinflammatory hyperpigmentation. However, it is important, based on its duration of action, that it be applied twice a day.55
Recent case series show enhanced response to the lightening effect of hydroquinone when used in combination with glycolic acid56 or tretinoin.57,58 This may be in part due to the inherent ability of those compounds to improve dyschromias. Another mechanism, however, may be due to enhanced penetration of the hydroquinone since both glycolic acid and tretinoin alter the stratum corneum barrier.
One concern with long-term topical use of hydroquinone is the development of ochronosis. Hydroquinone inhibits homogentisic acid oxidase activity in the skin resulting in the accumulation of homogentisic acid, which polymerizes to form ochronotic pigment in the dermis. This manifests as blue-black pigmentation at sites of hydroquinone application. However, the literature indicates this has been seen mostly in African patients.59,60
Kojic Acid
Kojic acid 1–3% is an effective over-the-counter skin lightening agent. It has been widely used in the Orient prior to coming on the market in America. It is a hydrophilic fungal derivative obtained from Aspergillus and Penicillium species61 and inhibits tyrosinase through binding of copper.62 Studies show it can be just as effective as hydroquinone in the treatment of melasma,56thus making it a useful alternative for hydroquinone-sensitive patients. The biggest limitation to its use, however, is that kojic acid is highly sensitizing and can cause contact dermatitis in many of its users.63
Azelaic Acid
Azelaic acid 20% cream (Azelex™, Allergan Inc., Irvine CA) is a dicarboxylic acid, available by prescription for use in the treatment of rosacea, comedonal, and inflammatory acne. It has been found to improve hyperpigmentation by interfering with tyrosinase activity.64 Several small studies have shown it to be more effective than 2% hydroquinone65 or as effective as 4% hydroquinone if it is used in combination with glycolic acid.66 Some have seen it work well in postlaser hyperpigmentation, while others have not seen any clinical improvement in postinflammatory hyperpigmentation. For patients unable to use to hydroquinone, azelaic acid remains a safe and effective option.
Antioxidants
Photoaging and carcinogenesis are believed to result from oxidative cell damage due to the formation of reactive oxygen species (ROS) or oxygen free radicals. ROS are formed as part of cellular metabolism but their formation is increased by environmental insults such as UVR67 and smoking.22 ROS are damaging because of their harmful affects on DNA, cell membranes, and proteins, including collagen. Interest in preventing ROS damage has lead to the development of an overwhelming number of vitamin-containing products promoting their antioxidative properties. Given the limited scope of this chapter, only the most well-studied antioxidants will be reviewed.
The main vitamins utilized in skin care products are vitamin C (ascorbic acid), vitamin E (alpha-tocopherol), and beta (β)-carotene. However, there are a number of products on the market that are ineffective. To achieve clinical results, it is imperative to use formulations with antioxidants in a bioavailable form. For example, there are over six forms of vitamin C used in cosmetic products, yet the body only uses the L-ascorbate form. In addition, their stability and penetration of the skin are questionable. This is a common problem with studies performed in vitro, as it becomes difficult to extrapolate the findings to clinical relevance since topical bioavailability of many substances is dramatically different than in vitro pharmacokinetics.
Topically applied L-ascorbic acid has been shown to support collagen synthesis and it demonstrates anti-inflammatory and photoprotective effects. In vivo studies have shown increased mRNA levels of collagen Type I and III and tissue inhibitor MMP-I.68 Vitamin C is believed to have anti-inflammatory effects by reducing activation of nuclear factor kappa beta (NF-κB) through inhibition of tumor necrosis factor alpha (TNF-α).69 NF-κB is a transcription factor responsible for production of pro-inflammatory cytokines including TNF-α, IL-1, IL-6, and IL-870. Topical application of L-ascorbate (15%) or alpha-tocopherol (1%) demonstrated a photoprotective effect with a reduction in UVB-induced erythema and sunburn cell formation and protection against UVA-psoralen phototoxicity injury.71
However, timing of application of these products is important as evidenced by the inability of topical vitamins C and E and melatonin to reduce UV-induced erythema if applied to the skin after UV exposure.72 Also promising is the in vitro finding that alpha-tocopherol can indirectly inhibit age-dependent collagenase expression by inhibition of protein kinase C.73
Although vitamin C and vitamin E are widely used in topical cosmetic products, reports of side effects are few. Vitamin E in its topical form has been associated with allergic contact dermatitis, contact urticaria, and erythema multiforme-like eruptions.74 Additionally, in vitro studies have shown that high concentrations of antioxidants can have a paradoxical effect, causing damage to the cells rather than protecting them.75,76 With continued advancements in formulations, the likelihood of these side effects should be minimal; however, caution is still advised.
Photoprotection
As scientific data showing the deleterious effects of UVR exposure grows, the importance of photoprotection cannot be denied. In addition to the causative role UVR plays in photoaging, increased outdoor UV exposure and indoor tanning has led to a rise in cutaneous malignancies such as basal and squamous cell carcinomas and melanoma.77,78Despite awareness of these dangers, many patients feel that the “healthy” appearance of a nice tan is well worth the future risk of skin cancer and photoaging.
There are three main types of UVR, categorized as UVA, UVB, or ultraviolet C (UVC), based on their wavelength. UVC (270–290 nm) is mostly filtered by the ozone layer and therefore does not pose a risk to the skin. UVB and UVA, however, are true sources of cutaneous injury. UVB (290–320 nm), with its shorter wavelength, does not penetrate beyond the epidermis, while UVA (320–400 nm) causes dermal injury.79 To better conceptualize this, UVB can be considered the “burning” rays, while UVA are the “aging” rays. Nucleic acids, proteins, and polyunsaturated lipids seem to be the chromophores for UVB, while UVA alters dermal proteins such as collagen, elastin, and PGs.
UVB exposure is variable, peaking between the hours of 10:00 am and 3:00 pm and is greatest during the summer months. UVA rays are relatively constant throughout the day and year. Since UVA can penetrate glass, it can be a significant source of exposure to people working near windows or driving extended distances on a regular basis.
Protection against UVR should be an integral part of every skin care regimen. Some patients may feel that the damage has already been done. However, it should be stressed to them that studies have shown a reduction in the formation of new precancerous lesions with regular sunscreen use.80
Sunscreens can be thought of as either chemical or physical blocks (Table 10.3). Chemical blocks absorb UVR in either the UVA or UVB range depending on the active ingredient, whereas the physical blockers absorb, scatter, and reflect both UVA and UVB. Therefore, in addition to recommending a high SPF sunscreen (over 15 SPF), one must make sure that the agent used affords protection against both UVA and UVB in order to obtain the maximum benefit.
Table 10.3 Sunscreens
| Sunscreen: active ingredients | UV protection |
| Chemical blocks | |
| Aminobenzoic acid (PABA) | UVB >> UVA |
| Cinnamates | UVB >> UVA |
| Salicylates | UVB >> UVA |
| Benzophenones | UVB > UVA |
| Avenobenzone (Parsol 1789, DSM Nutritional Products Inc., Parsippany, NJ) | UVA |
| Ecamsule (Mexoryl SX, LaRoche-Posay, L’Oreal International, Clichy Cedex, France) | UVA >> UVB |
| Physical blocks | |
| Titanium dioxide | UVA and UVB |
| Zinc oxide | UVA and UVB |
Novel Treatments and Future Trends
There are a growing number of new products on the market that address issues of aging and hyperpigmentation from a slightly different mechanism from traditional agents. Mention of them is made here as their use may hold promise; however, large, well-controlled studies with long-term follow-up are needed before widespread use is advocated.
N-Furfuryladenine
There is a new agent in the treatment of photoaging, N-furfuryladenine (Kinerase™, ICN Pharmaceuticals Inc., Costa Mesa, CA). It is a patented plant growth factor that slows the aging characteristics of human fibroblasts in vitro.81 Ongoing clinical trials are showing promising results with a reduction in fine wrinkles, hyperpigmentation, and tactile roughness. However, it is important to realize that histopathological correlation is needed before it can be determined if this agent is more efficacious than tretinoin.
Hormone Replacement Therapy
Hormone replacement therapy is the latest in the battle against the aging process. As the body ages, there is a gradual and progressive decline in the production of hormones that control various functions (Figure 10.2). Of greatest interest is the increase in dermal thickness (increased collagen I production) created by growth hormone replacement.82 Additionally, improved wound healing noted with topical epidermal growth factor (EGF) is leading to the development of skin creams that will exert their effects on keratinocyte EGF receptors.83
Figure 10.2 Pseudohypopigmentation following chemical skin resurfacing.
Conclusion
Physicians must assess a patient’s skin with respect to intrinsic and extrinsic aging and formulate a regimen with which the patient will comply. Patients who are educated about their skin will be more likely to take an active role in their care. While treating diseased skin states is important, prevention cannot be overemphasized. Sun protection and abstinence from tobacco are as important, if not more so, than skin care products. Every adult patient will benefit from a regimen that keeps his or her skin functioning at a youthful level. Therefore, every adult patient, at the very least, should be using sunscreens and tretinoin.
Advances in our understanding of skin rejuvenation will continue to rapidly impact skin care. While it may feel overwhelming, knowledge of skin function, the aging process, and the mechanisms of action of various products will help the physician make a wise choice. It must be stressed that some of the products mentioned, while promising, must still weather the test of time.
As has been discussed, prevention is certainly the best medicine. Once the skin has been damaged beyond the levels that would be best treated by the methods already discussed, other modalities are available to rejuvenate our patients.
As pointed out in other chapters of this book, botulinum toxin and fillers are very useful and have enjoyed widespread popularity. Therefore, we will focus on refreshing the skin using chemical and laser techniques.
Chemical Peels
Introduction
Chemical peels have been used for centuries to rejuvenate the skin, making them the oldest form of skin resurfacing. While newer technologies such as dermabrasion, fractionated laser resurfacing, and noninvasive lasers continue to evolve, chemical peels remain an important tool in skin rejuvenation. If used correctly, they may be tailored to treat almost any skin defect. In addition, chemical peels can be combined with laser resurfacing or dermabrasion to create a blending effect, thus optimizing outcomes.
Indications for Chemical Peels
Chemical peels can be used to address a variety of skin conditions, including solar lentigines, actinic keratoses, ephelides, dyschromias, rhytides, shallow or saucer-shaped scars, and photodamage.1 To perform a successful peel, it is most important to first assess the patient’s skin, understanding the anatomy, the depth of the cutaneous pathology being addressed, and the mechanisms of action of the various peel solutions. With these factors in mind, the surgeon can select a procedure and present it to the patient in a manner that fosters realistic expectations.
Patient Evaluation
As has been repeatedly stated in this text, proper patient selection is one of the most important aspects of cosmetic surgery. Patients who seek treatment for barely noticeable skin anomalies are likely to be unhappy with postoperative results, even when the physician considers the outcome a success. Thus, it is important that the physician present both what the procedure can and cannot achieve for the patient beforehand.
A thorough history and physical exam should emphasize medications and medical illnesses that could impact wound healing.84 These would include medications such as prednisone, immunosuppressants, or D-penicillamine, along with conditions such as diabetes or scleroderma. Nicotine use or prior radiation treatment to the face and neck can also impair wound healing, as resurfaced skin requires intact and functioning pilosebaceous units and a good blood supply to reepithelialize correctly. The physician should inquire about previous poor wound healing, hypertrophic or keloid scars, or a tendency to develop postinflammatory hyperpigmentation.
The physical exam should take place in a well-lit room and the patient’s skin should be free of all makeup. Any preexisting skin anomalies, including pigmentary abnormalities, or scars should be pointed out to the patient and documented prior. Telangiectasias, in particular, do not resolve with skin resurfacing and may be more noticeable after the procedure, as areas of dyschromia previously masked them. Patients may incorrectly blame the peel, if they were not pointed out prior.
Using before and after photographs is the best way to document this information. In addition, skin conditions that can spread to traumatized skin (vitiligo, psoriasis, lichen planus, verrucae vulgaris, and plana) should be excluded.
Contraindications to Chemical Skin Resurfacing
The advantage of chemical skin resurfacing is that with proper pre- and postprocedure skin conditioning, patients with most skin types can be treated. However, patients with darker skin types (Fitzpatrick Type III or higher) should be treated with caution, as these patients are more at risk for permanent hypopigmentation with procedures that reach the depth of the reticular dermis.85 These same patients are at risk for postinflammatory hyperpigmentation with any procedure. To minimize this risk, the length of the preoperative conditioning phase should be extended to 3 months and resumed immediately upon reepithelialization of the skin.
Patients who have recently taken isotretinoin also pose a special risk for hypertrophic scarring with resurfacing procedures, specifically dermabrasion and certain laser treatments.86–89However, large, controlled studies with skin peels have demonstrated that if the depth of the peel is kept at or above the papillary dermis, peels can be safely performed in patients with past, current, or postoperative use of isotretinoin.90 Exper/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


