Prevention of Disease Transmission in Oral Health Care
Disease Transmission
See the section on “Microbial Virulence and Disease Transfer” in Chapter 9.
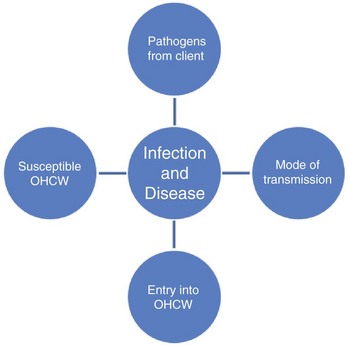
A Development of an infectious disease
1. Source of microorganism or pathogen (microorganism capable of causing infectious disease)
a. Primarily originating from a person’s mouth
b. May originate from a dental team member (this mode less likely than from a client’s mouth)
c. Microbes may be present in saliva, blood, respiratory secretions, and other bodily fluids
2. Escape of microorganism from the source
a. Coughing, sneezing, and talking
b. Contaminated dental equipment, instruments, and supplies used in a client’s mouth
c. Spatter droplets and aerosol particles generated by using power instrumentation, low-speed and high-speed handpieces, and air/water syringe
3. Spread of microorganisms to another person
a. Direct contact with microorganisms in a client’s mouth that penetrate through nonintact skin
b. Indirect contact with items contaminated by a client’s microorganisms, including sharps (all items that can puncture skin), instruments, surfaces, hands, and equipment
c. Droplet infection from spatter that contacts nonintact skin or mucous membranes (oral, nasal, ocular)
d. Airborne infection from inhaling aerosols
e. Waterborne infection from contaminated dental unit waterlines (DUWL)
4. Entry of a microorganism into the person
a. Inhalation—breathing aerosol particles
b. Ingestion—swallowing droplets of saliva or blood spattered into the mouth
c. Through mucous membranes—droplets of saliva or blood spattered into the eyes, nose, or mouth
d. Through breaks in the skin—by touching contaminated objects, spattering of microorganisms onto nonintact skin, or punctures with contaminated sharps
6. Damage to the body—the disease occurs when microorganisms multiply to a harmful level
7. Pathogenic agents and diseases associated with oral health care (Table 10-1)
TABLE 10-1
Pathogenic Agents Important in Oral Health Care
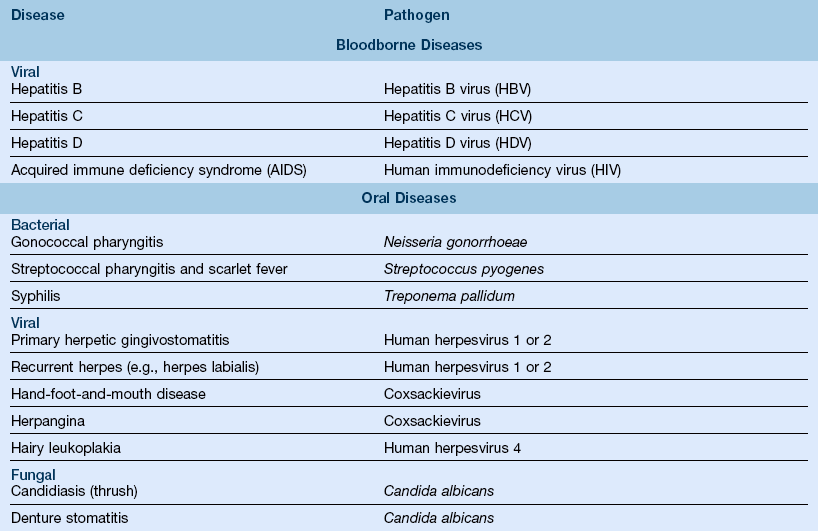
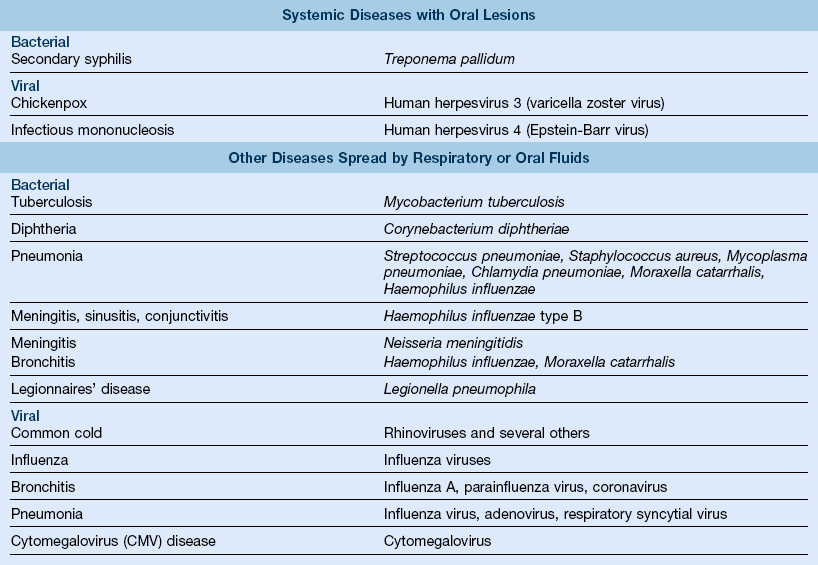

Modified from Miller CH, Palenik CJ: Infection control and management of hazardous materials for the dental team, ed 4, St Louis, 2010, Mosby.
1. Reducing the numbers of microorganisms that may be transmitted from individual to individual or from individuals to contaminated surfaces, and vice versa
2. Infection control protocol to reduce or eliminate the likelihood of disease transmission by eliminating one or more factors required for disease transfer (Figure 10-1)
a. Asepsis—absence of infectious materials; achieved by removing or killing microorganisms
b. Disinfection—reducing the number of pathogenic organisms in or on an object, thereby minimizing the potential for disease transmission
c. Engineering controls—devices that reduce the risk of exposure to potentially infectious materials (e.g., self-sheathing needles)
d. Personal protective equipment (PPE)—items designed to protect oral health care workers (OHCWs) from exposure to bloodborne and other pathogens (e.g., gloves, masks, safety glasses, face shields, and barrier gowns)
e. Standard precautions—treating all blood and bodily fluids (including secretions and excretions, except sweat), nonintact skin, and mucous membranes as potentially infectious in all clients; in the oral health care setting, saliva is an important source of contamination, as it is invisible but capable of containing infectious agents that can survive on surfaces for a long period
f. Sterilization—the destruction or removal of all microorganisms in or on an object
g. Work practice controls—procedures that reduce the chance of exposure to potentially infectious materials (e.g., using a one-handed “scoop” technique to recap needles)
(1) Maintaining a healthy lifestyle and immune status
(2) Receiving appropriate vaccinations
(3) Abiding by the recommended U.S. Public Health Service work restrictions for OHCWs in the case of certain infections and after exposure to some diseases
(1) Prophylactic antibiotic premedication to reduce the possibility of infection in immunocompromised individuals and others with various conditions
(2) Pre-procedural rinse to decrease the microbial load in spatter and aerosols and to help protect against autogenous (self-derived) infection
(3) Avoiding treatment of clients with known communicable illnesses (i.e., influenza) or contagious lesions (i.e., herpes)
3. Designed to reduce or prevent the spread of pathogens from:
4. Table 10-2 outlines the mechanisms of the spread and the prevention of diseases
TABLE 10-2
Mechanisms of Disease Spread and Prevention
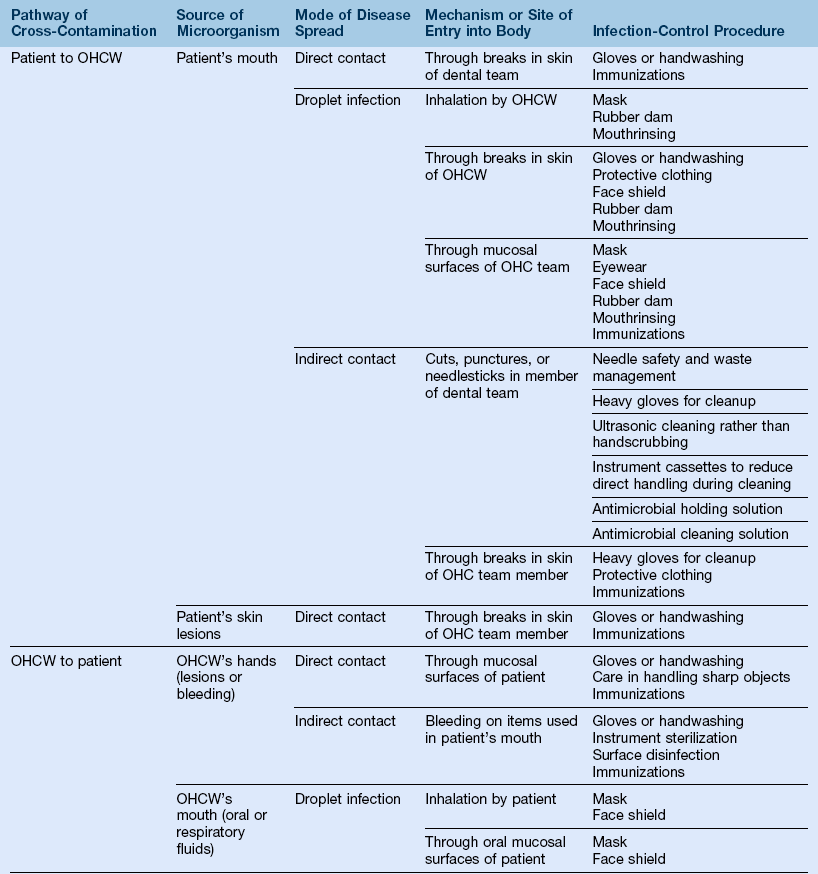
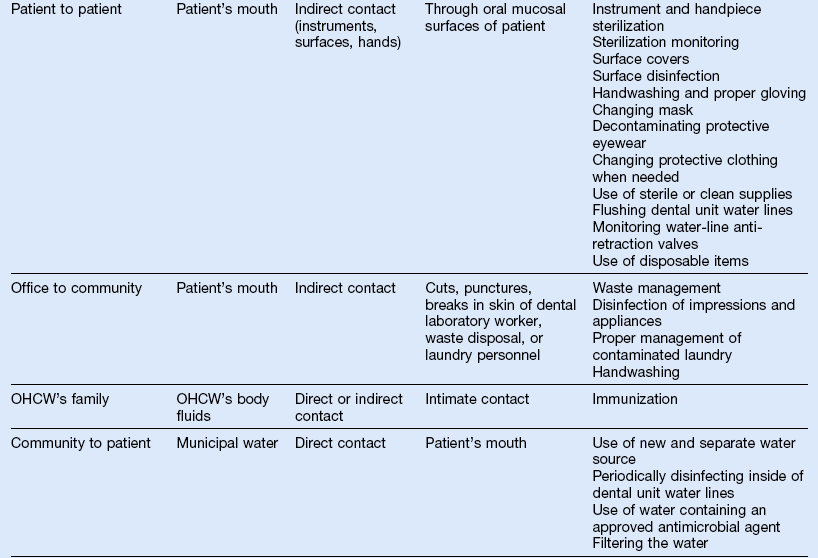
OHCW, Oral healthcare worker; OHC, oral healthcare.
Data from US Department of Labor, Occupational Safety and Health Administration: Controlling occupational exposure to bloodborne pathogens, Washington, D.C., 1996, 2001, OSHA 3127 (revised).
Infection Control Procedures for Oral Health Care Workers
1. Obtain, review, and update clients’ health histories at all visits
2. Address specific questions related to present health status, physician care, hospitalizations, surgery, diseases, medications, allergies, current or chronic illness, review of major organ systems, and recent overseas travel
3. Be aware that obtaining health histories will not identify all infectious clients; clients may suppress information purposely or unknowingly (many persons with hepatitis B virus [HBV], hepatitis C virus [HCV], and human immunodeficiency virus [HIV] are asymptomatic)
4. Health history may identify conditions requiring:
a. Diagnostic evaluation and laboratory testing
c. Prophylactic antibiotic premedication to reduce the incidence of autogenous infections, that is, conditions caused by introducing the microflora of clients into injured tissues (e.g., bacteremia, abscess)
5. The Centers for Disease Control and Prevention (CDC) recommends conducting a tuberculosis risk assessment for an oral health care setting and then formulating a prevention program appropriate for its designated risk category; most dental settings will be low or very low risk
B Immunizations are recommended for OHCWs
1. Essential component of an infection control program
2. Most effective method to prevent contracting a vaccine-preventable disease
3. Immunizations are recommended for the following diseases, unless evidence of past infection and immunity: hepatitis B, varicella zoster, influenza, measles, mumps, polio, rubella, tetanus, influenza A (H1N1); consult with a primary care physician before vaccination, as some vaccines are contraindicated for immunocompromised and pregnant persons
4. The Occupational Safety and Health Administration (OSHA) policies regarding hepatitis B vaccination
a. The OSHA requires employers to make hepatitis B vaccine available at no cost to employees who are or may be exposed by occupation to bloodborne pathogens and within 10 working days
b. Employers must provide information about the vaccine and ensure that all medical records concerning the vaccination are kept confidential
c. Employees have the right to refuse vaccination but must read and sign an OSHA declination statement
d. Employers cannot demand that employees be serologically prescreened before vaccination
a. Two single antigen vaccines available
(1) Serologic screening for antibodies to hepatitis B surface antigen before vaccination is not recommended, unless infection is suspected
(2) Pregnancy is not a contraindication for vaccination
(3) A series of three injections in the deltoid muscle given at 0, 1, and 6 months
(4) Seroconversion rates are 95% to 97% in healthy younger adults; lower rates (approximately 70%) in persons over 40 years old, smokers, overweight persons, and those receiving injections in the buttocks
(5) Genetic factors may influence seroconversion rates
(6) Testing for antibody (anti-HBsAg) is recommended after vaccination to ensure protection against HBV; testing should be conducted 1 to 2 months after the final injection
(7) With successful seroconversion, protective antibodies have been sustained for at least 20 years
(8) Failure to seroconvert requires a second series of three injections
(9) Continued failure to seroconvert could signal chronic HBV infection in a person who is unable to produce antibodies; nonresponders should be evaluated by a medical provider
(10) The CDC currently does not recommend a booster injection until further research is conducted on past recipients of vaccine
(11) Minimal side effects of vaccine include injection site soreness, headache, and fever
(12) Individuals with allergies to yeast or iodine (vaccine preservatives) must consult a physician before the vaccination
1. Extremely important disease prevention practice in oral health care
2. Hands are a primary source of microorganisms capable of disease transmission
a. Handwashing reduces both resident and transient flora on skin
(1) Resident flora are permanent residents that colonize several skin layers and cannot be completely removed. This type is less important and less likely to transmit disease than are transient flora
(2) Transient flora contaminate hands when hands touch contaminated surfaces. This type colonizes the outer layers of skin, only survive for a limited time, and can be easily removed by routine handwashing
b. Protects clients and OHCWs by reducing the spread of microorganisms and subsequent infections via the hands
c. Unwashed hands can contaminate sterile instruments, dental equipment, and environmental surfaces
4. Use of gloves is not a substitute for routine handwashing
5. Hands must be washed before gloves are put on to minimize organisms that can multiply rapidly when enclosed in a moist, warm environment; bacteria and yeast growth can cause skin irritation
6. Hands must be washed after removal of gloves because defects, tears, and punctures may occur in gloves, permitting microorganisms to be transferred to hands; this also helps remove glove powder, which contains latex protein and other glove chemicals that can elicit irritant contact dermatitis or an allergic reaction in sensitized individuals
7. Watches, bracelets, and rings must be removed to prevent harboring of microorganisms; also, rings may perforate glove materials
8. Nails must be kept short and clean and the cuticles well maintained; artificial nails and nail jewelry are not recommended, since current research has implicated them in disease transmission in hospitals
9. Intact skin is the best protection against infection; this can be achieved by:
a. Minimizing trauma to hands (e.g., cuts and scrapes) while outside the dental setting
b. Protecting hands from drying and chapping during cold weather
10. OHCWs who have open or weeping lesions or dermatitis on hands should not provide client care until the condition resolves, since dermatitis reduces the effectiveness of handwashing and nonintact skin provides a portal of entry for microorganisms
11. Hand hygiene procedures for routine dental hygiene care include:
a. Vigorous lathering of hands using an interlacing finger motion with either an antimicrobial or plain soap for 15 seconds
(1) Disposable soap containers should not be refilled, and when empty should be discarded and replaced
(2) Soap dishes accumulate microorganisms and are not recommended
(3) Sinks and soap dispensers should be electronically operated or have foot controls; if not, paper towels should be used to turn off sink faucet
b. Rinsing with cool to lukewarm water while rubbing hands together for 10 seconds
c. Drying hands with single-use paper towels
d. Using alcohol-based hand rubs as an alternative to handwashing
(1) Used when no visible soil appears on the hands
(2) Have been shown to be effective, sometimes reducing bacterial counts more effectively than soap and water
(3) May increase the frequency of hand hygiene because of ease of use
(4) According to manufacturer’s directions, place an adequate amount of alcohol-based hand rub in hand and then vigorously rub hands until dry
(5) Alcohol-based hand sanitizers may cause dry skin (choose ones that contain emollients)
e. Washing hands between clients, before and after lunch, before and after restroom visits, or any time hands become contaminated
f. Maintaining asepsis by touching only sterile instruments or disinfected surfaces
12. For surgical procedures, an antimicrobial soap with substantivity (prolonged anti-microbial effect) is recommended; chlorhexidine digluconate or triclosan
D Personal protective equipment (PPE)
1. Protective barriers used to reduce exposure of mucous membranes, hands, and body of OHCWs to microorganisms and also to prevent injury from chemicals and particles of debris
2. Used during client care, laboratory, disinfection, and sterilization procedures
3. PPE includes gloves, masks, protective eyewear, and protective clothing
4. Sequence for donning PPE: protective clothing, then mask and eyewear, and finally, after handwashing, gloves
5. The employer is responsible for providing and maintaining appropriate PPE for employees
a. The use of gloves provides a high level of protection for both OHCWs and clients
(1) Prevents direct contact with microorganisms in the client’s mouth and on contaminated surfaces (bare hands often will have areas of nonintact skin providing portals of entry for pathogenic microorganisms)
(2) Prevents saliva and blood from being retained under fingernails; saliva and blood have been shown to persist for several days even with handwashing
(3) Protects against contact with disinfecting and cleaning chemicals and x-ray solutions
(4) Protects clients from the microorganisms on the hands of the OHCW
(5) Examination gloves provide little protection against sharps injuries; utility gloves provide more protection; however, injuries still can occur
b. Risks associated with not routinely wearing gloves
(1) OHCW exposure to potentially infectious client tissues and contaminated surfaces—most likely route by which OHCWs have acquired HBV from clients
(2) Client exposure to infectious agents originating from the OHCW
(a) Documented source of transmission of HBV from ungloved dentist to clients
(b) Documented case in which an ungloved hygienist transmitted herpes to 20 of her clients
(3) Skin irritation from contact with disinfecting chemicals
(4) Burns resulting from contact with hot items from sterilizer
(1) Wear during intraoral procedures and when in contact with contaminated items or surfaces (e.g., contaminated laundry or waste)
(2) If it is necessary to leave the chairside during client care, remove gloves, and after hand hygiene, don a new pair on returning (prevents contamination of additional surfaces one may touch and also prevents contamination of the client with microorganisms that already may be present on those surfaces)
(3) Ensure that gloves cover the cuff of a long-sleeved gown
(4) Change gloves between clients and during long appointments because defects in gloves increase with use beyond 60 minutes
(5) Do not wash or disinfect gloves; may cause “wicking” or enhanced penetration of liquids through undetected defects in gloves
(6) Remove torn or punctured gloves as soon as possible. Wash hands and don new gloves
(7) Do not apply petroleum-based hand lotion prior to wearing gloves because it degrades latex gloves; for the same reason, do not apply petroleum-based lubricants to client’s lips
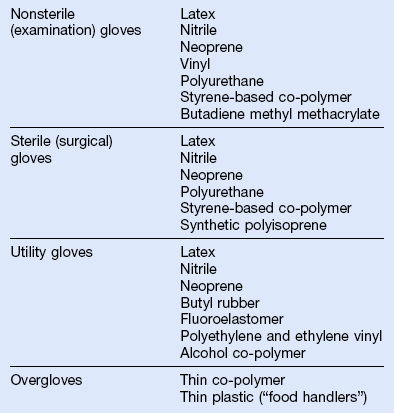
d. Types of gloves—See Table 10-3 for glove materials
(1) Nonsterile, ambidextrous gloves in sizes extra-small, small, medium, and large are adequate for most procedures; proper glove fit is important to ensure efficient instrumentation and to prevent hand fatigue and possibly carpal tunnel syndrome
(2) Sterile gloves are recommended for surgical procedures
(3) Use puncture-resistant and chemical-resistant utility gloves to prepare chemicals, handle contaminated instruments, and clean and disinfect surfaces
(a) Utility gloves are reusable and can be disinfected or sterilized in an autoclave
(b) Replace utility gloves when they show any signs of wear (e.g., cracks, punctures, discoloration)
(4) Overgloves are worn over treatment gloves to prevent cross-contamination of items and surfaces such as pens, charts, and drawers
(5) Heat-resistant gloves are worn when handling hot items (e.g., unloading sterilizers)
e. Dermatitis and latex allergy
(1) Irritant contact dermatitis
(a) Nonimmunologic (nonallergic) reaction of skin to chemicals used in glove manufacturing
(b) Skin on hands becomes dry, red, itchy, and cracked
(c) The condition is aggravated by soaps, not rinsing or drying hands completely, perspiration, or cornstarch powder
(d) Most skin reactions from wearing gloves are caused by irritant contact dermatitis and are not true allergic reactions
(2) Allergic contact dermatitis
(a) Type IV or delayed hypersensitivity occurring within hours or days because of allergy to glove chemicals
(b) Limited to area of contact, causing itching, redness, and vesicles to appear within 24 to 48 hours followed by dry skin, fissures, and sores
(a) Type I or immediate hypersensitivity within minutes or hours
(b) Allergy to naturally occurring latex proteins
(c) Symptoms: skin (hives, swelling, burning, tightness, itching, redness, tingling), lungs (asthma, wheezing, constriction, coughing, sneezing, rhinitis, angioedema), and other (nausea, vomiting, diarrhea, cramps, hypertension, tachycardia, shock)
(d) Anaphylactic shock and death can occur with subsequent exposures to latex
(e) High-risk individuals for latex allergy include: persons who have had multiple surgeries and persons with spina bifida, urogenital anomalies, spinal cord injuries, and allergies to bananas, kiwis, chestnuts, or avocados
(f) Reductions in exposure to latex proteins are known to decrease sensitivity (important for OHC team to reduce their daily exposure to airborne latex proteins by wearing powder-free, reduced-protein latex or latex-free gloves)
(g) The CDC indicates that OHCWs need to be educated about skin problems that can occur with frequent hand hygiene and the use of gloves
(h) Latex-free environment should be provided to clients and OHCWs with a latex allergy
(4) Procedures for management of persons with latex allergy
(a) Include questions in health history appropriate for identifying possible latex allergy
(b) Document latex allergy in health history record in a way that will ensure observation by OHCWs
(c) Schedule allergic clients for the first appointment of the day when airborne latex proteins are at their lowest levels (still risky)
(d) Ensure that OHC team uses latex-free gloves for treatment area preparation and for touching all items that will come in contact with the client
(e) Use latex-free gloves and dental materials during client care
(1) Worn to protect the mucous membranes of the nose and mouth from spatter of oral fluids
(2) Provide a lesser degree of protection from inhalation of aerosol particles
(a) Surgical masks will not provide protection from airborne infections (e.g., SARS)
(b) The N-95 respirator is needed to protect against airborne infections
(3) May provide some protection to client from nasal or oral secretions of OHCWs
b. Composed of synthetic material that should filter at least 95% of small particles
d. Should have a seal against the face to minimize leakage around the margins
e. Maximizing effectiveness and minimizing cross-contamination
(1) Use a new mask for each client
(2) Change the mask if it becomes moist. Moistness compromises its effectiveness by increasing the passage of unfiltered air around the edges of the mask and may wick contaminants through the mask
(3) Don a new mask every 20 to 30 minutes to maintain high filterability
(4) Adjust the mask so that it fits snugly against the face
(5) Avoid touching the mask during the appointment
(6) Keep the mask on after completing the procedure to reduce inhalation of aerosols
(7) When removing the mask, handle it by its strings or elastic
(8) Do not leave the mask on the head, dangling around neck, or in your pocket
a. Purpose—protect the mucous membranes of the eye from microbial invasion, chemicals, and physical projectiles
(1) Regular glasses offer limited side or top protection and are not recommended
(2) Safety glasses, goggles, or loupes
(a) Cover entire eye orbit (providing protection on all sides)
(b) Some may be worn over prescription glasses
(c) Are more shatter resistant than regular glasses
(d) Provide minimal visual distortion
(e) Are able to withstand disinfection (should be cleaned and disinfected between clients)
(f) Should meet American National Standards Institute (ANSI) guidelines
(a) An alternative to safety glasses
(b) Should be chin-length and provide top and side protection
(c) Masks should still be worn to reduce inhalation of aerosols
(d) Provide maximum coverage of face for high-spatter procedures (e.g., air polishing or power scaling)
(e) Face shields made of thin plastic may have limited impact resistance
9. Protective (barrier) clothing
(1) Protects nonintact skin from contamination by microorganisms
(2) Reduces the risk of bringing contaminants beyond dental setting on unprotected clothing
(3) May protect client from microorganisms on street clothing or may provide protection against disease transmission when soiled protective clothing is changed between clients (fomites—clothing and paper that can absorb and transmit infectious agents)
(4) Microorganisms adhere to clothing; however, lack of evidence exists to support the extent to which barrier clothing protects against disease transmission
b. Characteristics of protective clothing
(2) Gowns, aprons, laboratory coats, or uniforms used as a covering for street clothing or scrub uniforms
(3) High collar that fits closely around the neck
(4) Long-sleeved garments with fitted cuffs allow gloves to extend over them for complete coverage
c. To maximize effectiveness and minimize cross-contamination
(1) Use fabric made of synthetic material, which is more fluid-resistant
(2) Do not wear protective clothing outside treatment area (remove before going out [e.g., to lunch] or leaving the dental setting)
(3) Change protective clothing daily or when visibly soiled
(4) Avoid touching clothing during client care and throughout the day
(5) Roll protective clothing inside out to minimize contact with exposed surface
10. Additional barrier protection used when cleaning or performing surgery
Oral Health Care Environment and Promotion of Infection Control
A Design and equipment selection emphasizes
1. Smooth construction—eliminates knobs, hooks, and crevices
2. Design of client chairs and operator stools that:
3. Avoidance of fabric-covered, coiled, or mechanically retracted tubings
4. Sink faucets and soap dispensers with foot or electronic controls
5. Paper-towel dispenser that is designed to avoid touching hardware or is electronically controlled
6. Plastic-lined waste containers recessed under cabinet, with opening on countertop
7. Surfaces that are compatible with disinfectants and detergents
8. Plastic laminate instead of wood for cabinets and countertops
9. Vinyl flooring and walls that are smooth and seamless
10. Carpeting or wallpaper not recommended
11. Dental unit water lines (DUWLs) that provide:
12. Reduction of airborne microbes
a. Air circulation exchange system or single-room filtration units
b. Ventilation systems to control noxious sterilization and laboratory vapors
c. Prevention of recirculation of contaminated air or transport of microbes
d. Cooling and heating system filters that prevent transfer of microbes
13. Housekeeping surfaces (e.g., walls, cabinets, and floors) should be cleaned and disinfected routinely with detergent and water or with detergents or low-level hospital disinfectants
a. Clean mops and cloths after use and allow to dry before reuse, or use single-use items
b. Clean walls, blinds, and window treatments in client-care areas when they are visibly soiled
c. Perform cleaning with a wet cloth or mop to prevent distribution of microorganism-laden dust particles
14. Keep treatment area free of unnecessary or seldom-used equipment and items
Maintaining Asepsis in the Oral Health Care Environment
A Items associated with oral health care are classified as:
1. Critical—instruments that penetrate oral soft tissue or bone, enter the bloodstream, or enter other sterile tissues of the mouth (e.g., curet); must be heat sterilized or be single-use (disposable) devices (SUDs)
2. Semi-critical—items that come in contact with mucous membranes (used in the mouth) but will not penetrate soft tissue, contact bone, enter the bloodstream, or enter other sterile tissues of the mouth (e.g., radiographic film holders)
a. Should be heat sterilized or be SUDs (disposable)
b. If heat sensitive, decontaminate using a chemical sterilant or a high-level disinfectant, or cover the device with a barrier to prevent contamination (e.g., digital radiography sensors)
3. Noncritical—items that contact intact skin (e.g., blood pressure cuff)
B Maintaining asepsis with the use of chemicals and surface covers
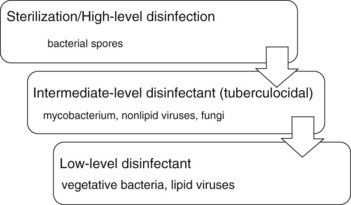
1. Categories of disinfecting or sterilizing chemicals (Table 10-4)
TABLE 10-4
Categories of Disinfecting or Sterilizing Chemicals
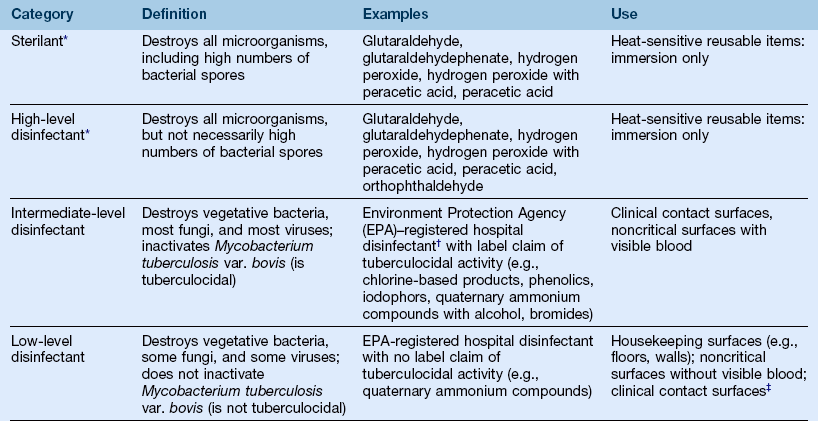
*Some, but not all, of these products can serve as high-level disinfectants and sterilants, depending on the immersion time used.
†A hospital disinfectant is one that has been shown to kill Staphylococcus aureus, Pseudomonas aeruginosa, and Salmonella choleraesuis.
‡The Centers for Disease Control and Prevention (CDC) indicates that low-level disinfectants can be used on clinical contact surfaces if the product has a label claim of killing human immunodeficiency virus (HIV) and hepatitis B virus (HBV) in addition to being an EPA-registered hospital disinfectant.
Modified from Centers for Disease Control and Prevention: Guidelines for infection control in dental health-care settings, Morbidity and Mortality Weekly Report 52(RR-17):1–66, 2003.
a. Sterilant—destroys all microorganisms, including high numbers of bacterial spores
b. High-level disinfectant—destroys all microorganisms but may not destroy bacterial spores (depending on contact time, can be either a disinfectant or sterilant)
c. Intermediate-level—destroys vegetative bacteria, most fungi, and most viruses; inactivates Mycobacterium tuberculosis var. bovis
d. Low-level—destroys vegetative bacteria, some fungi and viruses; does not inactivate M. tuberculosis var. bovis (Figure 10-2)
C Surface disinfection of clinical contact surfaces
1. Any surface that is touched by contaminated hands, instruments, devices, or other items during the provision of oral health care (e.g., light handles, dental equipment)
2. Identify and list surfaces that will be contaminated during treatment
3. Post the list in the treatment area to increase effectiveness and efficiency of decontamination procedures
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses