Chapter 10 Immunity and infection
Bacterial, viral, parasitic and fungal infections are major causes of morbidity and mortality worldwide, especially in poorer societies with less access to medicines and vaccines, greater exposure to infectious agents and poorer nutrition. Infectious and parasitic diseases were responsible for 29.6% of the world’s disease burden in 1999, according to the World Health Organization (Table 10.1).
| Infectious disease | Cause | Annual deaths |
|---|---|---|
| Acute respiratory infections (mostly pneumonia) | Bacterial or viral | 4 300 000 |
| Diarrhoeal diseases | Bacterial or viral | 3 200 000 |
| Tuberculosis | Bacterial | 3 000 000 |
| Hepatitis B | Viral | 1 000 000–2 000 000 |
| Malaria | Protozoan | 1 000 000 |
| AIDS | Viral | 1 000 000 |
| Measles | Viral | 900 000 |
| Neonatal tetanus | Bacterial | 600 000 |
| Pertussis (whooping cough) | Bacterial | 360 000 |
AIDS, acquired immune deficiency syndrome.
From The World Health Report (1999). WHO, Geneva.
Immunity to bacteria
Summary of defence mechanisms
Damage caused by immune responses to bacteria
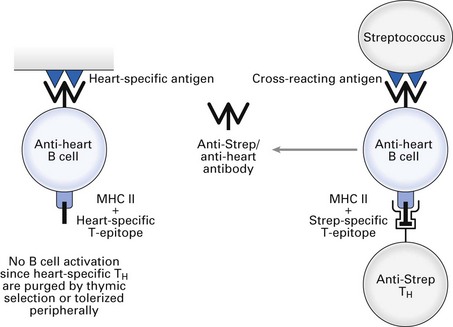
Group A β-haemolytic streptococci cause sore throat and scarlet fever, which resolve on induction of specific antibody. Certain components of some strains of streptococci contain epitopes that are cross-reactive with epitopes present on heart tissue. Antibodies that eliminate the infecting bacteria can bind to heart tissue and cause complement-mediated lysis and antibody-dependent cellular cytotoxicity (rheumatic heart disease). Furthermore, circulating immune complexes can deposit in synovia and glomeruli, causing complement-mediated joint pain and glomerulonephritis, respectively. Induction of cross-reacting anti-heart antibody by group A streptococci is illustrated in Fig. 10.1 (see also Fig. 23.2).
Immunity to viruses
Summary of defence mechanisms
HIV and AIDS
At the end of the year 2008, approximately 40 million people worldwide had become infected with HIV and approximately 25 million had died of the acquired immune deficiency syndrome (AIDS) (see also Chapter 30). The virus causes depletion of CD4+ T-helper lymphocytes over many years. Patients eventually succumb to opportunistic infections (Pneumocystis carinii, M. tuberculosis, atypical mycobacteria, Histoplasma, Coccidioides, Cryptococcus, Cryptosporidium and Toxoplasma spp., herpes simplex, cytomegalovirus) and may develop Kaposi’s sarcoma, B cell lymphomas and other malignancies. Infection of the brain by HIV can cause dementia and encephalitis.
The life cycle of HIV is shown in Figure 10.2. The virus gains entry into target cells by binding its surface gp120 molecule (glycoprotein of 120 kDa) to CD4 on T-helper cells and a subset of macrophages. The latter can also take up opsonized HIV via Fc or complement receptors. A coreceptor is also required for infection of target cells: CXCR4, also known as fusin or LESTR, is the receptor for the chemokine SDF-1 and is the coreceptor for infection of T cells by HIV; CCR5, the receptor for chemokines RANTES, MIP-1α and MIP-1p, is the coreceptor for infection of macrophages. Viral gp41 cau/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses