Periodontology
Chapter Contents
1.5 Microbiology and pathogenesis of periodontal diseases
1.6 Risk factors and predisposing factors
1.7 Furcation and periodontal–endodontic lesions
1.9 Trauma and the periodontium
1.10 Syndromes and medical conditions associated with aggressive periodontitis
Overview
A healthy or a stable periodontium is an important prerequisite both for the maintenance of a functional dentition and to ensure a long-term, successful outcome of restorative dental treatment. In view of the high prevalence of gingivitis and chronic periodontitis in the population, all dental patients should undergo periodontal screening, although more thorough clinical and radiographic examinations are essential before a definitive periodontal diagnosis is confirmed and a treatment plan formulated. These examinations, together with medical, dental and social histories, may also reveal predisposing and risk factors that increase an individual’s susceptibility to, and the subsequent rate of progression of, periodontal disease.
The intensive oral hygiene phase of treatment and the patient’s compliance with a personalised plaque-control regimen are of major importance in stabilising the disease and improving the long-term prognosis for an affected dentition. Scaling and root surface instrumentation (RSI) are frequently indicated to disrupt the subgingival biofilm and remove calculus. Additional adjunctive treatments that may be indicated are periodontal surgery, guided tissue regeneration, systemic or locally delivered antimicrobials and the management of localised problems such as furcation defects, mucogingival problems, periodontal–endodontic lesions and loss of attachment that has been exacerbated by a traumatic occlusion.
1.1 Healthy periodontium
The diagnostic skills required to identify periodontal diseases, particularly in the early stages, are based upon a sound knowledge of the clinical appearance of healthy tissues.
Clinical features
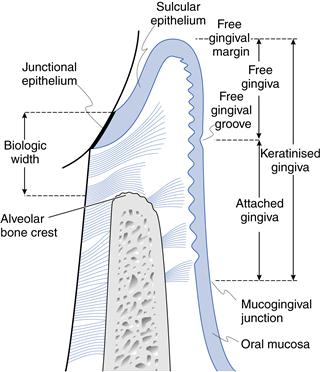
The gingiva is pink, firm in texture and extends from the free gingival margin to the mucogingival line. The interdental papillae are pyramidal in shape and occupy the interdental spaces beneath the contact points of the teeth. Gingiva is keratinised and stippling is frequently present. The gingiva comprises the free and the attached portions.
The free gingiva is the most coronal band of unattached tissue demarcated by the free gingival groove, which can sometimes be detected clinically. The depth of the gingival sulcus ranges from 0.5 to 3.0 mm.
The attached gingiva is firmly bound to underlying cementum and alveolar bone and extends apically from the free gingival groove to the mucogingival junction. The width of attached gingiva varies considerably throughout the mouth. It is usually narrower on the lingual aspect of the mandibular incisors and labially, adjacent to the canines and first premolars. In the absence of inflammation, the width of the attached gingiva increases with age.
The mucogingival line is often indistinct. It defines the junction between the keratinised, attached gingiva and the oral mucosa. Oral mucosa is non-keratinised and, therefore, appears redder than the adjacent gingiva. The tissues can be distinguished by staining with Schiller iodine solution; keratinised gingiva stains orange and non-keratinised mucosa stains purple–blue. This can be used to determine clinically the width of keratinised tissue that remains (e.g. in areas of gingival recession).
Radiographic features
The crest of the interdental alveolar bone is well defined and lies approximately 0.5–1.5 mm apical to the cementoenamel junction (Fig. 1.1). The periodontal membrane space, often identifiable on intraoral radiographs taken using a paralleling technique, is approximately 0.1–0.2 mm wide. This accounts for the slight tooth mobility that is sometimes observed when lateral pressure is applied to a tooth with a healthy periodontium.
Histology
Epithelial components include:
• sulcular epithelium: non-keratinised and lines the gingival crevice
• oral epithelium: keratinised and extends from the free gingival margin to the mucogingival line.
Gingival connective tissue core contains ground substance, blood vessels and lymphatics, nerves, fibroblasts and bundles of gingival collagen fibres (dentogingival, alveologingival, circular and trans-septal). The combined epithelial and gingival fibre attachment to the tooth surface is the biologic width, which is typically 2 mm, not including the sulcus depth (see Fig. 1.1).
Periodontal connective tissues comprise alveolar bone, periodontal ligament, principal and oxytalan fibres, cells, ground substance, nerves, blood vessels and lymphatics, and cementum.
Periodontal tissues in children
The gingiva in children may appear red and inflamed. Compared with mature tissue, there is a thinner epithelium that is less keratinised, greater vascularity of connective tissues and less variation in the width of the attached gingiva.
During tooth eruption, the gingival sulcus depths may reach 5 mm and gingival margins will be at different levels on adjacent teeth. Following tooth eruption, a persistent hyperaemia can lead to swollen and rounded interproximal papillae, thus giving an appearance of gingivitis.
Radiographic features
In the primary dentition, the radiographic distance between the cementoenamel junction (CEJ) and the alveolar crest is 0–2 mm. Greater variation (0–4 mm) is observed at sites adjacent to erupting permanent teeth and exfoliating primary teeth. The periodontal membrane space is wider in children because of the thinner cementum, immature alveolar bone and a more vascular periodontal ligament.
Gingival crevicular fluid
Gingival crevicular fluid (GCF) is a serum exudate that is derived from the microvasculature of the gingiva and periodontal ligament. The ‘preinflammatory’ flow of GCF may be mediated by bacterial products from subgingival plaque that diffuse intercellularly and accumulate adjacent to the basement membrane of the junctional epithelium. This creates an osmotic gradient; consequently, GCF flow can be regarded as a transudate rather than an inflammatory exudate.
GCF is, in some ways, similar to serum but also contains components from microbial sources, interstitial fluid and locally produced inflammatory and immune products of host origin. The proportions of these components are dependent upon:
• the presence and composition of subgingival plaque
• the rate of turnover of gingival connective tissue
Several techniques have been developed for collecting GCF from the gingival sulcus:
The fluid can then be analysed for specific mediators of the immunoinflammatory response (e.g. cytokines) and breakdown products of connective tissues, both of which have been associated with ongoing periodontal destruction.
1.2 History and examination
From the periodontal viewpoint, the aims of history taking and the clinical examination are to establish the extent of periodontal destruction and to evaluate the effects of disease on the remaining dentition. It is also important to evaluate the individual patient’s susceptibility to periodontal disease and, as far as possible, identify the sites that appear to be associated with active or ongoing destruction and need to be considered a priority for treatment.
Presenting complaint
One of the principal features of periodontal diseases is that their onset and progression occur often in the absence of pain. This means that the onus for detection rests firmly with the clinician, and the importance of regular examinations must be impressed upon the patient, with emphasis placed on prevention rather than cure. The well-informed patient who is a regular dental attender should be able to detect some of the signs or symptoms that are associated with the early stages of plaque accumulation. Unfortunately, many patients are irregular dental attenders and only present with complaints that are the consequence of oral neglect. When gingivitis and periodontal inflammation do cause symptoms, the chief complaints are usually ‘bleeding gums’, ‘bad taste or breath’, ‘localised pain’ and teeth that have ‘changed position’ or ‘become loose’. Details of when such problems started, the frequency of pain or discomfort and any associated symptoms should be recorded. The expectations of the patient with regard to the outcome of treatment should also be discussed at this stage.
Gingival bleeding
Bleeding gums is perhaps the most common complaint of patients with periodontal disease. The bleeding is usually noticed during, or following, toothbrushing or eating. When bleeding occurs spontaneously, a patient may complain of tasting blood on awakening in the morning. The severity of the haemorrhage does not necessarily relate to the severity of disease, as a marginal gingivitis can be associated with quite profuse bleeding. Gingival bleeding is exacerbated by the use of certain drugs (anticoagulants, antithrombotics and fibrinolytic agents). Symptoms of relatively recent and sudden onset should be investigated thoroughly when taking the medical history.
Drifting of teeth
Drifting of anterior teeth and the appearance of spaces between teeth are often the first signs of an underlying periodontal problem. When teeth begin to drift, it is because their periodontal support has been compromised to such an extent that the teeth are no longer in equilibrium with forces from occlusion and the adjacent soft tissues. In some instances, this position of equilibrium is so finely balanced that the destruction of only crestal bone and the coronal periodontal fibre groups will precipitate changes in tooth position. Furthermore, the pressures exerted on the teeth by gingiva that are swollen through oedematous or fibrous change can also induce tooth movement. Drifting of anterior teeth may also be a consequence of an occlusal interference in the posterior segments, which leads to a forward slide of the mandible during its arc of movement from the retruded contact position to the intercuspal position.
Loose teeth
When periodontal disease remains untreated, attachment loss is progressive and teeth become increasingly mobile. The degree of mobility that some patients accept before attending for treatment is remarkable and many patients still believe that increasing tooth mobility and, ultimately, tooth loss is a natural consequence of the ageing process. An increase in mobility also occurs when a tooth is subject to increased or abnormal occlusal forces, particularly those of a ‘jiggling’ nature. Mobility may be the first signs of an advanced stage of periodontitis or perhaps a rapidly progressive, or aggressive, type of disease.
Bad taste and halitosis
Altered sensation of taste can accompany the halitosis that is associated with:
Pain
Acute and often quite severe pain is a feature of NUG and herpetic gingivostomatitis. Pain, particularly on eating, is also a symptom of an acute periodontal abscess and/or a periodontal–endodontic lesion. Gingival recession with exposure of root surfaces can also precipitate pain if dentine is exposed as a result of toothbrush abrasion. This pain is characterised as sharp and transient, with a sudden onset that is precipitated by extremes of temperature. Pain is not typically a feature of chronic periodontitis, however.
Dental history
The dental history provides an indication of the patient’s overall attitude to dental care. Lengthy intervals between appointments and attendance for only symptomatic treatment suggest a low priority on dental health and a patient who is unlikely to appreciate and comply with comprehensive periodontal care.
The reasons for previous loss of teeth should be established and a record made of previous and recent dental treatment. Information (including radiographs) relating to previous dental treatment should, whenever possible, be sought by written request from a previous dentist, and the written response incorporated in the patient’s notes. Another criterion sometimes used to assess dental behaviour is the frequency with which a patient brushes (or claims to brush!) his or her teeth. It is more important to assess the efficiency of the method of toothbrushing rather than to place too much emphasis on frequency. An individual who brushes once a day for 4–5 minutes is often able to maintain a superior standard of oral hygiene than a patient who brushes several times a day, but ineffectively and for only short periods of time.
In young patients in particular, a note should be made of previous orthodontic treatment. Extended periods of fixed appliance therapy can cause loss of crestal alveolar bone partly from tooth movements and partly from the periodontal inflammation that is a consequence of limited access to cleaning interproximally and subgingivally. More importantly from the diagnostic viewpoint, teeth that have been tipped rather than moved bodily through bone often have an angular alveolar crest on the mesial and distal surfaces. Such topography gives the appearance of the lesions often seen in localised aggressive periodontitis.
Social history
Details of the patient’s occupation, diet and consumption of alcohol and tobacco should be noted. When an occupation involves considerable social contact, there may be a greater awareness of small changes of tooth position and appearance.
Stress induced by examinations, divorce or change of employment should also be noted as they may promote bruxism and aggravate existing tooth mobility from periodontal disease. Stress has been shown to be associated with delayed wound healing of connective tissue and bone, NUG and, maybe, chronic periodontitis. As most patients have some element of stress in their lives, the potential influence of this on periodontal disease should be appreciated. A lack of ability to cope with stress, and particularly financial strain, has been implicated as a specific risk factor in periodontal disease.
Smoking is a known risk factor for periodontal disease and is considered in Section 1.6. The frequency and duration of smoking should be established and the detrimental effects of smoking on periodontal health must be conveyed to the patient before any treatment is started.
It should now be apparent that much of the information that can be derived from a thorough personal and dental history has a bearing on establishing the susceptibility of an individual to periodontal disease. When potential risk factors are established, it is often not possible to determine their individual effects on the disease process because many of the factors are inter-related. For example, an individual who has job insecurity and is under financial strain may be also a smoker and a poor dental attender.
Medical history
A thorough medical history must be recorded and updated at each visit. The patient’s perception of their present health status is also a valuable indicator of their psychological make-up and potential compliance with treatment.
A patient with a history of rheumatic fever, congenital cardiac defects or prosthetic heart valves does not require antibiotic prophylaxis before periodontal probing and treatment. Similarly, patients who have received prosthetic joint implants do not require antibiotic prophylaxis. Ultrasonic scalers can be used in patients with cardiac pacemakers in accordance with the manufacturers’ guidance, which normally recommends that the ultrasonic handpiece and cables should be kept at least 15 cm away from the pacemaker device.
Diabetic patients are at particular risk of periodontal breakdown, especially when poorly controlled. A positive family history should be noted and vigilant periodontal monitoring undertaken. The HIV-positive patient is also at risk from very extensive and aggressive periodontal breakdown.
Patients with particular food fads or unusual diets should be questioned as part of an overall dietary analysis to evaluate their vitamin and protein intake. Nutritional deficiencies may modify the severity and extent of periodontal disease by altering the host resistance and potential for repair, although such deficiencies are rare in Westernised societies.
Gastric hyperacidity and reflux from hiatus hernia and gastric ulceration predispose to erosion and root caries if there is existing gingival recession. Pregnant patients should be monitored carefully during the second and third trimesters as endocrine changes may lead to marked gingival inflammation and the development of epulides. Radiographic assessment of periodontal disease should be avoided during pregnancy.
Current medications must be noted, especially dosage and types of medication. When a patient is receiving anticoagulant therapy, the general medical practitioner or patient’s physician must be consulted with a view to modifying the anticoagulant dosage to coincide with invasive periodontal treatment, thus reducing the risk of postoperative haemorrhage. Some drugs such as phenytoin, ciclosporin and nifedipine can cause gingival overgrowth, which may compromise good oral hygiene, leading to aesthetic problems. For patients taking bisphosphonates, extractions and osseous surgery should be avoided due to the risk of osteonecrosis. Antimicrobials often used in the treatment of periodontal diseases are contraindicated for certain patients for whom the unwanted effects of the drugs may be enhanced, or because of a potential interaction with drugs that the patient is already taking.
Examination
Extraoral examination
A careful extraoral examination may reveal important signs that are associated with periodontal problems. A severe periodontal abscess can lead to facial swelling and a regional lymphadenopathy. Prominent maxillary incisors make a lip seal difficult to achieve and this may aggravate an existing gingivitis. The drying effect on exposed gingiva produced by mouth breathing leads to enlarged and erythematous gingiva, particularly in the maxillary anterior region. Mouth breathing does not inevitably lead to increased plaque accumulation and gingivitis but should be regarded as a predisposing factor in a susceptible patient.
Intraoral examination
A record should be made of local factors that predispose to the accumulation of plaque (e.g. restorations with overhanging margins, poorly contoured and deficient restorations and partial dentures).
A quick and simple method of assessing the level of oral hygiene is to score, after disclosing, the number of plaque-covered smooth tooth surfaces as a percentage of all smooth surfaces. On each surface, plaque is recorded as being either present or absent (a dichotomous scoring method). Patients are informed of their scores and realistic targets can be set for the patient to achieve at future visits. This method gives a useful overall assessment of plaque control as well as identifying tooth surfaces that are difficult to clean. These occur typically at interproximal sites and on the lingual smooth surfaces of mandibular molars.
In epidemiological studies, it is easier and quicker to select six teeth per subject to be representative of the entire dentition. These so-called Ramfjord teeth are: 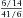
A number of indices have been used for scoring plaque, oral debris and calculus on a quantitative basis (Table 1.1). Periodontal diseases and gingivitis occur in all patients regardless of age. Chronic periodontitis is prevalent in adults and gingivitis is extremely common in children. Furthermore, children and young adults are also at risk from the more aggressive, early-onset diseases. It is, therefore, imperative that all dental patients undergo a screening examination to provide a rapid, basic assessment of periodontal status. The Basic Periodontal Examination (BPE) has evolved from the Community Periodontal Index of Treatment Needs (CPITN) and is a quick method for assessing a patient’s periodontal status. The examination involves the use of a specially designed periodontal probe with a 0.5-mm diameter ball end and a coloured band extending 3.5–5.5 mm from the tip (Fig. 1.2). The dentition is divided into sextants; each tooth is probed circumferentially and only the highest score in each sextant is recorded. The score codes are used as a guide to determine the need for periodontal treatment (Table 1.2). A BPE score of 3 means that full probing depths (six sites per tooth) around all the teeth in that sextant should be recorded. A score of 4 in any sextant means that probing depths should be recorded throughout the entire dentition.
Table 1.1
Indices for scoring oral debris, plaque and calculus
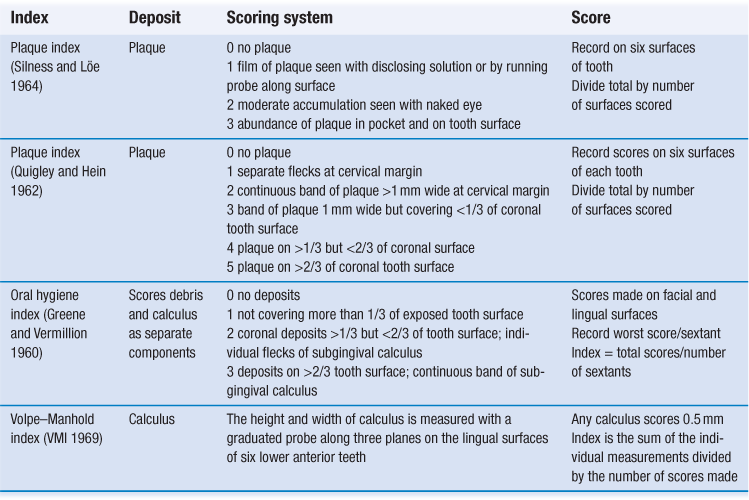
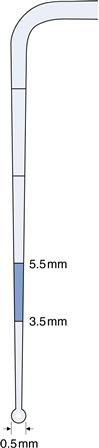
Fig. 1.2 Colour-coded probe for the basic periodontal examination. (World Health Organization (WHO) probe.)
Table 1.2
The Basic Periodontal Examination (BPE)
| Code∗ | Probing | Treatment needs |
| 0 | Coloured area of the probe is completely visible; no calculus detected; no gingival bleeding on probing | No need for periodontal treatment |
| 1 | Coloured area is completely visible; no calculus detected; bleeding on probing | Oral hygiene instruction (OHI) |
| 2 | Coloured area is completely visible; supra- or subgingival calculus detected, or overhanging restorations | OHI; elimination of plaque-retentive areas; scaling and RSI |
| 3 | Coloured area is partly visible, indicating probing depth of greater than 3.5 mm but less than 5.5 mm | OHI; elimination of plaque-retentive areas; RSI |
| 4 | Coloured area completely disappears, indicating probing depth of greater than 5.5 mm | Assess the need for more complex treatment in addition to OHI and RSI; referral to a specialist may be necessary |
The symbol (∗) should be added to score where furcation involvement is evident
Reproduced by kind permission of the British Society of Periodontology
Gingiva
Visual examination of the gingiva may reveal colour changes of the tissues, gingival swelling (generalised or localised), ulceration, suppuration and gingival recession. Where there is gingival enlargement, the tissues should be probed gently to assess consistency and texture. Oedematous tissues are soft and may have a tendency to bleed spontaneously or following pressure and gentle manipulation. Conversely, fibrous tissue is usually quite firm and resistant to pressure.
The width of attached gingiva should be assessed and measured as the distance from the free gingival margin to the mucogingival line minus the depth of the gingival crevice (in health) or periodontal pocket (when disease is present). Sites with minimal or no apparent attached gingiva should be noted together with the inflammatory condition of the associated marginal tissues. At such sites, the attached gingiva can be dyed with Schiller iodine solution so that the border between the keratinised (orange) and non-keratinised (dark blue) epithelium (mucogingival junction) is seen and the actual width of keratinised tissue becomes more readily apparent. Sites of gingival recession are recorded by measuring from the CEJ to the free gingival margin of the affected site. Sensitivity of associated exposed root surfaces should also be recorded.
The presence of a prominent labial frenum may effectively reduce the width of attached gingiva, although the precise role of a frenal attachment as a predisposing factor to gingival recession is disputed. A prominent frenum can, however, reduce sulcus depth and restrict access for tooth brushing; it can thus lead to the development of local periodontal problems.
Periodontal probing
Periodontal probing should be undertaken systematically on each tooth to determine the probing depth, the presence of bleeding after probing and the extent of attachment loss. The probe should be moved gently around the sulcus to avoid trauma. A force of approximately 0.25 N is recommended, but this is difficult to achieve consistently without the use of a pressure-sensitive probe. An attempt should be made to probe along the contour of the root surface although, interproximally, it is necessary to angle the probe slightly to reach the site directly beneath the contact area. This site should be probed from the buccal and the lingual aspects since deep pockets frequently develop here.
A number of factors may lead to errors in measuring probing depths:
The extent of inflammation is also important. A probe will more easily penetrate the pocket epithelium and the adjacent connective tissues when the tissues are inflamed. A probing depth measurement is influenced by the position of the gingival margin and the integrity of the tissues at the base of the pocket, and these factors are dependent upon the extent of inflammation in the tissues. Attempts have been made to reduce probing errors by using constant pressure probes and, more recently, electronic probes. In addition, computer-assisted probes have been developed for automatic recording of probe measurements or to allow voice-activated data entry. These probes have a high degree of resolution, measuring with a precision of 0.1–0.2 mm, but their accuracy and repeatability still depends upon angulation and positioning of the probe.
A more precise assessment of the degree of periodontal destruction is made by measuring from the CEJ to the base of the pocket. This gives an approximation of the loss of connective tissue attachment to the root surface. The loss of attachment is easier to measure when there has been gingival recession and the CEJ is visible. When patients are being monitored longitudinally before and after treatment, sequential attachment level measurements can be made relative to a fixed point (e.g. an incisal edge or cusp tip). The differences between successive measurements then give an estimate of the change in attachment level, which is often used to assess the success or failure of a particular treatment.
About 20–30 seconds after probing, each site is re-evaluated to determine the presence or absence of bleeding from the base of the pocket. Bleeding is simply a consequence of the trauma caused by probing the epithelial pocket lining and connective tissue. Bleeding on probing has been implicated as an indicator of active disease. Longitudinal clinical trials, however, suggest that bleeding has a low sensitivity for disease progression. Conversely, absence of bleeding is a good indicator of periodontal health or inactivity. Any site with a probing depth of less than 4 mm that does not exhibit bleeding on probing is not likely to require treatment beyond supragingival scaling and polishing.
Furcation involvement
A curved explorer is used to determine the topography of the furcation lesion in multirooted teeth, allowing accurate classification into three groups.
Tooth mobility
Mobility is assessed by applying a labiolingual, horizontal force to each tooth in turn using the handles of dental mirrors. Movement is scored according to a simple index, such as:
Radiographic evaluation
Radiographic selection criteria for periodontal disease should take into account the diagnosis made from the clinical examination and the overall state of the patient’s dentition. The panoramic radiograph, particularly if obtained using modern machines, is an alternative to full mouth periapical radiography on the basis of diagnostic yield of clinically unsuspected patterns of bone loss. In the posterior segments, vertical bitewings are often a useful supplement if a panoramic view suggests bone loss localised to this region.
Radiographic features that can be identified include:
• pattern of bone loss: horizontal/vertical, localised/generalised
• widening of the periodontal membrane space
A decreased alveolar bone height on a radiograph is only a historical record of previous periodontal involvement and gives little, if any, information on recent or current activity.
1.3 Gingivitis
In this section, we describe the clinical features of the chronic and acute forms of gingivitis. The treatment of gingivitis and periodontitis is discussed in Section 1.12 although specific aspects of treatment are also noted in this section (and Section 1.4). The microbiology and pathogenesis of gingivitis are discussed in Section 1.5.
Chronic gingivitis
Chronic gingivitis is a plaque-induced, inflammatory lesion of the gingiva. Accumulation of dental plaque in the gingival sulcus initiates the development of an inflammatory lesion (subclinical) that, after 10–20 days, is detected clinically as an established chronic gingivitis.
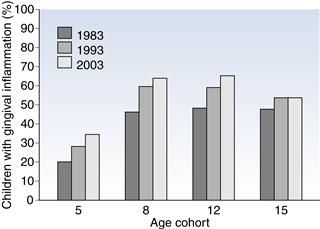
Gingivitis occurs in 32% of 5-year-olds, 63% of 9-year-olds and 52% of 15-year-olds (O’Brian 2003). There has been no reduction in the prevalence of gingival inflammation in children over the 20-year period between 1983 and 2003 (Fig. 1.3). Indeed, in the 5-, 8- and 12-year-old age groups, there were more children with gingival inflammation in successive decades (1983–2003) although, in 15-year-olds, the proportion of children with gingival inflammation appears to be stable (48–52%).
Clinical features
The gingiva become red, shiny, swollen and soft or spongy in texture. Sulcus depths increase (false pockets) as a result of the tissue swelling from inflammatory oedema. Bleeding occurs after gentle probing. The interdental papillae and marginal gingiva are initially involved before inflammation spreads to the attached gingiva.
Pregnancy gingivitis
An increase in circulating levels of oestrogen, progesterone and their metabolites may aggravate a pre-existing gingivitis. The hormones and their metabolites effect an increase in gingival vasculature and the permeability of the capillary network. A similar increase in the severity of gingivitis may also be seen at, or around, puberty.
Clinical features
Pregnancy gingivitis is a generalised, marginal, oedematous inflammation. The extent of gingival enlargement is variable but an increase in gingival bleeding is a common complaint. The severity of the gingivitis tends to increase from the second to the eighth month of pregnancy. There is often some resolution during the final trimester and after parturition. A local gingival overgrowth (i.e. pregnancy epulis) may result from chronic irritation or mild trauma to the soft tissues.
Treatment
A preventive regimen is preferred whenever possible. Otherwise a conventional treatment approach including oral hygiene instruction (OHI) and scaling should be undertaken.
Plasma cell gingivitis
Plasma cell gingivitis is a contact hypersensitivity reaction most frequently attributed to cinnamon flavoured chewing gum. Cinnamon, mint and herbal flavoured toothpastes are also implicated. Microscopically, the epithelium is atrophic and there is a massive infiltrate of plasma cells in the connective tissues.
Clinical features
In plasma cell gingivitis, the gingiva are fiery-red in appearance with varying degrees of swelling. The lesion extends to involve the entire width of attached gingiva. The reaction may affect other areas such as the tongue, palate and cheeks. Lips can be dry and desquamative with an angular cheilitis. The principal symptom is extreme soreness of the affected areas.
Treatment
Treatment involves dentification and withdrawal of the causative allergen. If toothbrushing is painful during the acute stage, chlorhexidine mouthrinse can be given for chemical plaque control.
Desquamative gingivitis
Desquamative gingivitis is not a discrete clinical entity but rather a term used to describe a gingival manifestation common to plasma cell gingivitis and mucocutaneous disorders including:
Clinical features
The fiery-red, desquamative lesions affect the entire width of keratinised gingiva and may be localised or generalised throughout the mouth.
Treatment
A joint management approach by specialists in oral medicine and periodontics is warranted. A biopsy of the affected region may be indicated to confirm the specific diagnosis. Oral hygiene instruction and scaling to remove hard deposits which may act as irritants should be undertaken. See Volume 1 for the pharmacological management of mucocutaneous lesions.
Necrotising ulcerative gingivitis
NUG is an acute condition that has characteristic signs and symptoms and a tendency to recur. In Europe and the USA, the incidence is highest in the 16–30-year-old age group. In African countries, a more severe, aggressive form/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses