Fig. 6.1
As highlighted in a 2007 review by Hanke et al., the data currently available on the relationship between occlusion and posture are discordant: some studies claim that these two aspects are closely related, while others deny the existence of such a relationship. Many factors contribute to the uncertainty in this field: the quality of the studies (often poor), the assessment instruments used (different in different studies), and the definition of posture itself.
To avoid misunderstandings, it must be clarified that there are two generally accepted definitions of posture: the position of the body in space, and the position of the different body segments in relation to each other. The two are actually closely linked and are governed by common neurological control mechanisms.
Postural maintenance is based on feedback and feedforward reponses that, indeed, depend on information originating from the visual apparatus and from the proprioceptive and vestibular systems.
Proprioception is particularly refined and developed at mandibular level: mandibular system proprioception, arising from the masticatory muscle system receptors (neuromuscular spindles, Golgi tendon organs), the periodontal ligament receptors (encapsulated nerve endings) and the temporomandibular joint receptors (Ruffini and Pacini corpuscles, Golgi tendon organs in periarticular tissue and free nerve endings in the subsynovial space), is transmitted to the central nervous system via the trigeminal nerve. It is thus clear that jaw position influences head position, as was demonstrated in a study by Gangloff and Perrin, who showed that pharmacologically induced unilateral truncular anaesthesia of the mandibular nerve leads to a significant, worsening of balance control, documented through stabilometry. This effect seems to be related to the connections that exist between the trigeminal and vestibular nuclei, and to the important role of the vestibulospinal bundle in balance maintenance reactions (Fig. 6.2).

Fig. 6.2
Tardieu et al., studying the influence of different occlusal conditions on postural control, discovered a significant relationship between these two aspects. However, the correlation was found to be short-lasting, in the sense that the subjects investigated showed an adaptation to the different conditions, posture being found to be progressively less affected as they were submitted to repeated recording sessions.
The influence of the jaw on general posture also appears to have a more strictly anatomical basis: D’Attilio et al. found different degrees of cervical lordosis in different skeletal classes: apparently less marked in the subjects in class III compared with those in the other classes, and more marked in the class II subjects. These differences seem to be due to the need for antero-posterior equilibrium of the craniocervical complex, a very important aspect given the distance that separates this complex from the ankle, considered to be the fulcrum around which we oscillate in order to maintain our balance. Tecco showed that a palatal expansion treatment was able to modify the cervical lordosis angle, even though it is not clear whether this effect has any clinical significance.
Whereas the relationship between posture and occlusion appears to be well documented, the same cannot be said of the relationship between posture and vertebral disorders or deformities. Indeed, the posture-occlusion relationship is clinically important inasmuch as it has a structural effect and can thus determine the appearance of vertebral abnormalities. At this point, two classical definitions, now probably obsolete, appear particularly helpful: those of paramorphism and dysmorphism. A paramorphism is the attitude, posture, or manner in which a body segment or the body as a whole is held in space, particularly in relation to the spine and trunk. A dysmorphism, on the other hand, is an out-and-out vertebral deformity. Posture, per se, is never pathological: it may not be “correct”, or optimal, but this does not constitute a disorder. The presence of vertebral deformities, on the other hand, does take us into the realm of pathological conditions. One example for all: the literature reports, in school age, a high frequency of paramorphisms in more sedentary compared with sportier youngsters, whereas the frequency of dysmorphisms in the two populations is the same. Put simply, sport, leading to better physical development and muscle training, favours the adoption of more correct postures, whereas it offers no benefits in terms of reducing pathological conditions, i.e. vertebral deformities. The reciprocal interaction between different districts of the body that determines posture, and thus paramorphisms, has no equivalent in the sphere of pathological conditions: occlusion can modify the position of the head in space, and perhaps of the trunk too, but it cannot give rise to a vertebral deformity; at least it has not, to date, been demonstrated that occlusion can cause a vertebral deformity. Furthermore, whenever correlations have emerged between occlusal abnormalities and the presence of vertebral deformities, no cause-effect relationship showing that one of the two alterations preceded the other has ever been demonstrated. Liselotte Sonnesen’s many studies have shown an association between occlusal class and some cervical vertebral malformations, such as posterior fusions. The occlusal dysfunctions most linked to vertebral disorders seemed to be open bites, class II malocclusion, class III malocclusion with mandibular overjet, and deep bite. However, these studies contain a major flaw: the healthy reference population used was always the same and was also rather small, numbering just 21 subjects; moreover, the study populations, too, were always rather small. It is thus possible that the results obtained are not representative of the healthy general and odontostomatologically dysfunctional populations. Korbmacher et al. found a correlation between frequency of scoliosis and unilateral crossbite, moreover without observing any side prevalence. This study also highlighted a correlation between clinically observable trunk asymmetries and crossbite. The selection of this population constitutes a main limitation of this study, given that the children considered were all drawn from an orthopaedic cohort: probably selected on the basis of specific characteristics, they may not be representative of the general population. Furthermore, the subjects with a positive clinical assessment were never submitted to confirmatory radiographic studies, which means some of the cases may have been false positives. The authors’ conclusions nevertheless reinforce a fundamental point: on the basis of the study data, it was not possible to establish a cause-effect relationship between occlusal alterations and spinal disorders. The association between scoliosis and crossbite is actually not all that surprising, given that it is known from the literature that scoliosis is correlated with numerous asymmetries, not only of the trunk but also of the face.
In view of the functional relationship that exists between the stomatognathic apparatus and the spine, in particular the cervical spine, it is possible that there could exist a cause-effect relationship as regards pain. Sipilä et al. found a clear correlation between facial pain and neck pain, even though the facial pain, in most cases, arose from the temporomandibular joint (TMJ), while the neck pain seemed to be mainly of myofascial origin. Komiyama et al. found a cognitive-behavioural intervention associated with posture correction to be more effective than cognitive-behavioural intervention alone for treating myofacial pain and limited mouth opening. However, the differences did not reach statistical significance. Apart from the odd case report, there exist no methodologically sound studies that have demonstrated a link with low back pain.
In general, evidence of the effectiveness of dental/orthodontic treatments in spinal pain is lacking, just as there is no real evidence that interventions at spinal level can effectively improve occlusion. It is possible that some patients may resolve their spinal pain through treatments of the oral cavity, but they are certainly the exception rather than the rule.
CONCLUSIONS
The mouth is an organ rich in proprioceptive receptors, which is why occlusal relations play a role in posture, of the head primarily but also of the cervical spine and the entire trunk. Accordingly, modifications induced at oral level can have repercussions on the rest of the body. However, an induced perturbation seems to be short-lasting and the organism tends return rapidly to its initial state. Some occlusal abnormalities seem to be related to anatomical abnormalities of the cervical vertebrae and to scoliosis, but the evidence supporting these relationships must be considered very weak, given the weakness of the studies the describe them.
A cause-effect relationship between occlusal and spinal alterations has never been demonstrated; similarly, it has not been demonstrated that dental or orthodontic treatments can influence, positively or negatively, vertebral anatomy or function, or that the treatment of scoliosis through exercise or orthopaedic braces can cause or improve a malocclusion. Therefore, the spinal expert and the dentist can work in parallel on the single patient without needing to worry about possible influences of their work on the work of the other, and vice versa.
There exists no indication for postural assessment using baropodometric platforms, stabilometric platforms, or instrumental surface topography in evaluation of the trunk before, during or after dental treatment, given the documented ability of postural control to adapt to perturbations and the poor or even absent specificity of these examinations in the dental setting (one need only think of how stabilometric results can vary according to a patient’s mood!). Today, the results of such examinations have no direct application in clinical practice and, unless they serve research purposes, are merely a source of business for commercial enterprises and specialists. In the clinical research setting, the question of the possible relations between posture and occlusion is currently attracting more and more resources; more than anything, there is a need for longitudinal studies that might finally establish whether or not there exists a cause-effect relationship between these two aspects, which at present do not appear particularly closely linked.
Recommended reading
Bolmont B, Gangloff P, Vouriot A, Perrin PP.: “Mood states and anxiety influence abilities to maintain balance control in healthy human subjects”. Neurosci Lett 2002;329(1):96-100.
Bucci Sabattini V, Bucci Sabattini F, Zaina F.: “La cartella clinica, gli esami preliminari e la modulistica fondamentale per il trattamento chirurgico impiantare. In: Vincenzo BS, editor. Tecniche ricostruttive e rigenerative dei mascellari atrofici I biomateriali: scelta, indicazioni e metodi di uso”. Torino: TU.E.OR.; 2007.
Chinappi AS, Jr., Getzoff H.: “Chiropractic/dental cotreatment of lumbosacral pain with temporomandibular joint involvement”. J Manipulative Physiol Ther 1996;19(9):607-612.
D’Attilio M, Caputi S, Epifania E, Festa F, Tecco S.: “Evaluation of cervical posture of children in skeletal class I, II, and III”. Cranio 2005;23(3):219-228.
Ferrario VF, Sforza C, Dellavia C, Tartaglia GM.: “Evidence of an influence of asymmetrical occlusal interferences on the activity of the sternocleidomastoid muscle”. J Oral Rehabil 2003;30(1):34-40.
Gangloff P, Perrin PP.: “Unilateral trigeminal anaesthesia modifies postural control in human subjects”. Neurosci Lett 2002;330(2):179-182.
Hanke BA, Motschall E, Turp JC.: “Association between orthopedic and dental findings: what level of evidence is available?” J Orofac Orthop 2007;68(2):91-107.
Kandel E, Schwartz J, Jessel T.: “Principi di neuroscienze”. Milano: C.E.A.; 1994.
Komiyama O, Kawara M, Arai M, Asano T, Kobayashi K.: “Posture correction as part of behavioural therapy in treatment of myofascial pain with limited opening”. J Oral Rehabil 1999;26(5):428-435.
Korbmacher H, Koch L, Eggers-Stroeder G, Kahl-Nieke B.: “Associations between orthopaedic disturbances and unilateral crossbite in children with asymmetry of the upper cervical spine”. Eur J Orthod 2007;29(1):100-104.
Sipila K, Zitting P, Siira P, Laukkanen P, Jarvelin MR, Oikarinen KS, Raustia AM.: “Temporomandibular disorders, occlusion, and neck pain in subjects with facial pain: a case-control study”. Cranio 2002;20(3):158-164.
Sonnesen L, Pedersen CE, Kjaer I.: “Cervical column morphology related to head posture, cranial base angle, and condylar malformation”. Eur J Orthod 2007;29(4):398-403.
Sonnesen L, Kjaer I.: “Anomalies of the cervical vertebrae in patients with skeletal Class II malocclusion and horizontal maxillary overjet”. Am J Orthod Dentofacial Orthop 2008;133(2):188 e115-120.
Sonnesen L, Kjaer I.: “Cervical column morphology in patients with skeletal open bite”. Orthod Craniofac Res 2008;11(1):17-23.
Sonnesen L, Kjaer I.: “Cervical column morphology in patients with skeletal Class III malocclusion and mandibular overjet”. Am J Orthod Dentofacial Orthop 2007;132(4):427 e427-412.
Sonnesen L, Kjaer I.: “Cervical vertebral body fusions in patients with skeletal deep bite”. Eur J Orthod 2007;29(5):464-470.
Tardieu C, Dumitrescu M, Giraudeau A, Blanc JL, Cheynet F, Borel L.: “Dental occlusion and postural control in adults”. Neurosci Lett 2009;450(2):221-224.
Tecco S, Caputi S, Festa F.: “Evaluation of cervical posture following palatal expansion: a 12-month follow-up controlled study”. Eur J Orthod 2007;29(1):45-51.
CHAPTER 7
IMPLANT PROSTHETICS AND TEMPOROMANDIBULAR DISORDERS
Daniele Manfredini
Visiting professor, Department of Maxillofacial Surgery, University of Padua
Temporomandibular disorders (TMDs) are a heterogeneous group of clinical problems involving the temporomandibular joint (TMJ) and/or masticatory muscles and associated structures (1).
These disorders are the most frequent cause of orofacial pain of non-dental origin, and even though their prevalence is still debated (2), there is evidence that signs and symptoms of TMDs are frequent in the general population (3).
The classical triad of clinical signs characterising TMDs is: pain in temporomandibular joints or masticatory muscles; TMJ sounds; limited or abnormal mandibular movements (4).
Recent studies in the literature suggest that, while the prevalence of TMDs is commonly believed to peak at around 35-45 years of age (5), in clinical populations there exist at least two different, age-related patterns of distribution of TMDs: a predominance of disc displacements associated with TMJ pain in 28- to 35-year-olds, and of symptoms of inflammatory-degenerative joint disorders in subjects aged 50 to 60 years (6).
As regards gender differences, the literature unequivocally demonstrates a strong female preponderance, with female:male ratios found to range from 2.5:1 to 7:1 in various studies involving different ethnicities (7,8).
Over the years, many diagnostic-classification systems have been proposed for TMDs (9).
At present, different standards of reference for framing TMDs are adopted in the clinical and research settings: whereas the guidelines and recommendations of the American Academy of Orofacial Pain (AAOP) (10) and of the European Academy of Craniomandibular Disorders (EACD) (11) are widely used in clinical practice, in research, extensive use is made of the Research Diagnostic Criteria for TMD (RDC/TMD)(12).
The RDC/TMD system, now translated into more than fifty languages in order to encourage its diffusion and multicentre and cross-cultural application, comprises coded criteria allowing a two-axis diagnostic assessment that covers both physical/organic (axis I) and psychosocial (axis II) aspects (Box 7.1).

Box 7.1 • Research Diagnostic Criteria for Temporomandibular Disorders (Dworkin and Leresche, 1992) (12).
A fundamental aspect of TMD diagnosis, acknowledged by all the most accredited systems, is the crucial role played by clinical assessment, which can achieve higher levels of diagnostic accuracy and inter-operator calibration than the various currently available electronic instruments (e.g. electromyography, mandibular kinesiography, posturometry) (13-16).
The aetiology of TMDs is recognised as multifactorial.
Numerous factors (psychological, socio-cognitive, parafunctional, skeletal, hormonal, occlusal) can, in ways that are often difficult to decipher, contribute to causing the symptoms in the single individual (17).
Given this lack of understanding of the aetiology of TMDs at single patient level, and also in view of encouraging success rates obtained with a variety of conservative therapeutic approaches, careful management of the clinical symptoms using reversible methods with low biological cost is now the standard of care in clinical treatment settings (18,19).
Over the years, therefore, there has been, both in the literature and in clinical practice, a gradual movement away from the old occlusal conception, according to which all TMD symptoms were due to presumed dental occlusion abnormalities, whose correction through extensive rehabilitation was the sole therapeutic objective.
Nowadays, a TMD is no longer seen as a dental pathology to be approached using dental-based treatments, but rather as a muscular-skeletal disorder that demands a broader, medical-based approach (20).
Notwithstanding this, the translation of correct scientific information into daily clinical practice remains incomplete, with many remnants of old theories still seeming to influence the way occlusion is routinely regarded.
Indeed, the role of occlusion is often exaggerated in many dental specialties, in spite of the absence of evidence demonstrating either the superiority or the iatrogenicity of given occlusal schemes (21,22).
The fact that TMD issues have, for years, been dealt with in association with prosthetic issues, even in the context of university training courses, explains the need to include TMDs in a book on implant prosthetics.
In the light of these opening remarks, it is clear that a discussion of the relationship between implant prosthetics and TMDs cannot fail to include a detailed analysis of the relationship between these disorders and dental occlusion.
OCCLUSION AND TMDs:
EVIDENCE-BASED DATA
The role of occlusion as an aetiological factor in TMDs has, for years, been fiercely debated, even giving rise to what has been called a “clash of cultures”, that is a diatribe between those who do not see occlusion as the aetiological primum movens and those who, instead, consider TMD signs and symptoms to be due to some occlusal abnormality.
Naturally, the most credible scientific position can only be that which is based on the currently available evidence, and actually this evidence throws into question the existence of an association, and certainly of a cause-effect relationship, between the presence of certain occlusal abnormalities and TMDs (20).
In particular, numerous systematic reviews of the literature, some of whose conclusions are summarised in Table 7.1, have confirmed that the available scientific evidence does not support the use of occlusal treatments as a therapeutic approach to TMDs.

Table 7.1 • Main systematic reviews of the literature on the role of occlusion as an aetiological factor in TMDs and on the efficacy of occlusal treatment in TMDs.
Historically, theories on the role of occlusion in the aetiology of TMDs date back to the 1930s and the studies of Costen, an otorhinolaryngologist who described a set of ear symptoms which he claimed were caused by a lack of posterior occlusal support (27). Even though the earliest published reports of TMJ disorders appeared in the century before that (28), meaning that Costen was not actually the “discover” of TMDs, the syndrome he described nevertheless took his name and today, seventy-five years on, it is not uncommon to come across the term Costner’s syndrome, even in specialist and medical-legal contexts. Actually, from a historical perspective, the most significant thing these founders of modern gnathology did was to identify dentists as the professionals who should treat patients with TMDs. Over the years, even though some authors had already realised that occlusion could not be the only aetiological factor in TMDs, and that an important role is played by psychoscocial factors (29), research into the relationship between TMDs advanced along two main lines: 1. study of the statistical association between certain occlusal variables and the presence of signs and symptoms of TMDs; 2. attempts to simulate, experimentally, situations of occlusal stress and overloading in order to verify their potential to damage the TMJ and masticatory muscles. A number of occlusal variables have been suggested to have a causal relationship with TMDs, for example, unilateral crossbite, anterior open bite, deep bite, and lack of posterior occlusal support (Fig.s 7.1-7.3), although studies using adequate statistical methods to depict biological models, reappraising the association of these variables with TMJ problems (30) and with muscular disorders (31), have instead suggested that they may simply be the means through which muscle forces developed during parafunctional activities are transmitted to the different structures of the stomatognathic system (32) (Table 7.2).

Table. 7.2 • Studies using multivariate statistical analysis to explore the “weight” of the occlusal variables in the identification of patients with TMDs.

Fig. 7.1 • Unilateral crossbite. The odds ratio for the presence of TMJ pain in subjects with this occlusal characteristic is 1.79 (95% confidence interval: 0.63-5.02) 32.

Fig. 7.2 • Anterior open bite. The odds ratio for the presence of TMJ pain in subjects with this occlusal characteristic is 0.91 (95% confidence interval: 0.19-4.40) 32.

Fig. 7.3 • Lack of posterior occlusal support. On application of multivariate statistical models, even this occlusal characteristic was found not to be predictive of TMDs 30.
The most accepted line of evidence is that the presence of occlusal abnormalities in patients with TMDs is actually due to joint degeneration and/or remodelling resulting in an occlusal shift (33) (Fig. 7.4).

Fig. 7.4 • In some cases, dental-skeletal abnormalities can be secondary to joint remodelling (e.g. “hot condyle”).
As regards experiments exploring the potential of occlusal interferences to provoke TMD signs and symptoms, the evidence produced by the numerous studies conducted on human and on animal models shows that possible iatrogenic interferences (e.g. high occlusal restorations) can, in the worst hypotheses, cause local trauma (26). In short, these interferences demand postural and functional adaptation of masticatory patterns which rarely gives rise to symptoms such as dental and/or masticatory muscle pain. Furthermore, when they do occur, these effects seem to be mainly transitory and easily reversed through removal of the iatrogenic interference. Data from more recent randomised controlled studies suggest that in healthy subjects, the application of an occlusal interference leads to a reduction in the usual electromyographic activity of the masseter muscles (34) and does not significantly affect pressure pain thresholds (35).
Interesting reflections are also prompted by a study which found that subjects with a history of TMDs tended to respond differently to iatrogenic occlusal interferences compared with subjects who had no history of TMDs (36). It seemed that the former have an increased risk of developing muscular pressure pain in response to occlusion abnormalities provoked by dental procedures. These observations should be borne in mind when carrying out occlusal treatments such as prosthetic or orthodontic rehabilitations (based, respectively, on prosthetic and orthodontic indications), which may involve periods of occlusal instability (e.g. temporary restorations, increases in vertical dimension, shifting teeth). Even though the publication of this study gave fresh impetus to those believing occlusion to be the main factor in the aetiology of TMDs (37), it is clearly important to avoid overestimating the importance of these results. First of all, the responses to the placement of the artificial interferences cannot be equated with the presence of TMDs; similarly, an acute occlusal alteration, like the simulated occlusal alterations in the various experimental studies, cannot really be compared to a clinical situation characterised by the presence of “non-ideal” dentition to which the patient, thanks to the exceptional adaptive capacity of the neuromuscular system, has gradually adapted over a period of years (20).
In the light of these remarks, it is legitimate to ask ourselves how much importance should be attached to occlusal considerations in the planning of extensive rehabilitations using prosthetic implants. In short: 1. Does carrying out rehabilitations using prosthetic implants increase a patient’s risk of developing signs and symptoms of TMDs? 2. Are there any particular indications that should be followed when performing such rehabilitations (for prosthetic, aesthetic, or other reasons) in patients affected by TMDs?
IMPLANT PROSTHETICS AND TEMPOROMANDIBULAR DISORDERS
In medicine, the best method for broaching any given medical topic is to search the available relevant literature in PubMed, a database of all the main publications appearing in journals submitted to editorial quality control.
The results of a PubMed search for publications identified by combining the key words “dental implants” and “temporomandibular joint disorders” were rather discouraging (Table 7.3).

Table 7.3 • Example of PubMed search conducted to identify studies on prosthetic implants and temporomandibular disorders (25/06/2009).
In particular, of the 35 publications identified, only two set out, as their main aim, to assess the prevalence of signs and symptoms of TMDs in patients previously submitted to oral implant surgery.
A study conducted to analyse and quantify the prevalence of sensory and affective aspects of orofacial pain in patients undergoing dental implant treatment found no significant associations between dental implantology procedures and pain in the TMJ and masticatory muscles (38). Another study, more detailed and better designed, was conducted to establish the prevalence of the appearance of TMJ osteoarthritis in patients wearing implant dentures; similarly, this study found no association between degenerative TMJ disease and dental implants after a mean follow up of over five years (39).
Obviously the evidence that can be drawn from such studies is extremely limited and a discussion of the relationship between implant prosthetics and TMDs must thus be based on several considerations of a biological nature that are actually applicable to all types of prosthetic rehabilitation, both those based on natural teeth and those using dental implants.
Theoretically, the results of the experiments on occlusal interferences, considered in a wider context, seem to suggest that any perturbation of a stomatognathic system that works, and works in the absence of TMD symptoms, should be avoided.
The stomatognathic system has extraordinary powers of adaptation, both to “natural” dental-skeletal abnormalities (40) and to iatrogenic modifications of the system (41).
In the different stages of the planning and execution of extensive rehabilitations using prosthetic implants, the primary objective, in order to protect the stomatognathic system against the appearance of symptoms of TMDs, is not to provoke modifications that demand an adaptation that exceeds the system’s own capacity for accommodation. In short, it is never advisable to perform rehabilitations according to predetermined occlusal schemes (Fig. 7.5) and according to preconceived ideas of interarch relations that fail to take into account the muscle engrams that the neuromuscular system of a patient with no TMD symptoms have developed naturally. In this regard, it is worth highlighting a few aspects and clinical considerations relating to the concepts of centric relation and vertical dimension.

Fig. 7.5 • Artificial or “forced” models and occlusal schemes, like the one shown here, have failed to stand the test of time, and new, simplified models are now emerging that are more valid and for which there are more evidence-based clinical indications.
CENTRIC RELATION IN THE PHYSIOPATHOLOGY OF MASTICATION
Centric relation (CR) has, for decades, been a controversial and much debated concept. Indeed, its definition has been changed on many occasions, even in the prestigious Glossary of Prosthodontic Terms, evolving from a retruded, postero-superior condyle position to an anterior-superior position of the condyle, which lies against the wall of the articular eminence (42) (Box 7.2).
|
Over the years, the definitions of centric relation (CR) and centric occlusion (CO) have changed. CR is currently taken to refer to an anterior-superior position of the condyles, which lie against the wall of the articular eminence. The term CO refers to the occlusion of the antagonist teeth when the jaw is in centric relation. This position, and that of maximum intercuspation, do not necessarily coincide.. |
Box 7.2 • The definitions of “centric relation” and “centric occlusion” (The Glossary of Prosthodontic Terms, 2005) (43).
Over the years, there has been a considerable reduction in the importance given to centric relation as a key reference position and, to some extent, as a “therapeutic” position; in the same way, it has been found that there is no real evidence to support the affirmation that in ideal conditions centric relation should coincide with the position of maximum intercuspation (MI) (44).
Furthermore, even though some studies have described a weak association between CR-MI slides and TMJ disorders, it is probable that such occlusal slides are actually effects, rather than a cause, of joint remodelling (45).
In view of these considerations, the need to play down the importance of having perfect harmony between the position of maximum intercuspation and some, too often arbitrary and “philosophical”, centric relation has been underlined repeatedly in both orthodontic(46) and gnathological settings (22) (Fig.s 7.6a-b).

Fig.s 7.6a-b • Example of discrepancy between CR and MI. a) “Hypothetical” centric relation position obtained by means of a manual relaxation technique. b) Position of maximum intercuspation. The odds ratio for the presence of TMJ pain in subjects with this slide between the CR and the MI is 0.92 (95% confidence interval: 0.37-2.23) 32.
Indeed, the available large-scale scientific evidence suggests that there actually exists a range of possible centric relations, and not a single position of reference common to all individuals.
Furthermore, no particular spatial position of the condyles in the glenoid fossa has been found to be predictive of TMDs; asymptomatic joints show various combinations of non-extreme glenoid fossa shapes and sizes and condyle positions (47) (Fig. 7.7).

Fig. 7.7 • There exist no relations between articular surfaces and condyle positions in the glenoid fossa that constitute certain indicators of the presence/absence of joint disorder. Asymptomatic TMJs appear to be simply those that are characterised by non-extreme glenoid fossa shapes and sizes and condyle positions.
In the light of these observations, therefore, there are no grounds for considering any single condyle position superior to the others.
Similarly, there is absolutely no need for obsessive pursuit of a particular condyle position, often termed myocentric, established in conditions of artificial muscle relaxation (48).
This position is one of the cornerstones of so-called neuromuscular dentistry, one of the many pseudosciences that lie at the border between dentistry and the other medical professions, and whose validity has never been demonstrated (49).
In particular, without dwelling too much on the lack of usefulness of this approach in the management of TMDs – its limits have been widely described in extensive literature reviews and are due above all to the absence of a relationship between the levels of masticatory muscle electromyographic activity and the presence of clinical symptoms (50,51) – the myocentric position can prove to be a misleading reference even in the simple planning stage of an extensive prosthetic rehabilitation.
This affirmation is based on several demonstrations that the muscles are active even in the postural position and that a mandibular rest position, namely the myocentric position, identified by the absence of EMG activity, is associated with an increase in vertical dimension, sometimes in the order of as much as 10 mm (52).
Even though it has also been underlined that the postural position and the EMG rest position are to be considered two distinct entities (53), it is a common clinical observation that rehabilitations based on electromyographic identification of the “therapeutic” position are characterised by an increase in the occlusal vertical dimension compared with the usual one.
The physiopathological significance of this increase, which can nevertheless sometimes be exploited to useful ends, deserves to be discussed in the light of the most recent considerations on the clinical importance of the occlusal vertical dimension.
IS THERE AN IDEAL VERTICAL DIMENSION AND IDEAL OCCLUSION?
For decades, the occlusal vertical dimension (OVD), which may be defined as the distance measured between two points on the face when the teeth are in occlusion (43), has been considered a fundamental parameter in the planning of extensive dental restorations.
Initially a static relation, the OVD is determined by the interaction of an individual’s genetic potential for craniofacial tissue growth with environmental factors and with the dynamic development of neuromuscular function during skeletal growth. Its maintenance is linked, more than anything, to the interaction of the latter two factors during ageing. Interindividual variability of the OVD is attributable mainly to facial morphology, growth and dental morphology (54).
In particular, contrary to what is commonly believed, even the most severe cases of vertical dental wear do not seem to be associated with considerable loss of vertical dimension, thanks to the process of continuous eruption which compensates, albeit sometimes only in part, for the loss of tooth substance (55).
Over the years, many different methods for accurately determining vertical dimension have been proposed, using mathematical calculations involving proportions between measurements of the face (56), phonetic techniques (57), mechanical or electronic instruments (58), and presumed physiological approaches (59), but none has emerged as unequivocally superior to the others; indeed, during pre-prosthetic planning, the established practice is to take the pre-existing vertical dimension as the reference (60).
These considerations notwithstanding, there is little doubt that, in daily clinical practice, increasing the OVD slightly in patients with extremely worn dentition (61) or with masticatory muscle or TMJ pain, in the latter case by means of occlusal splints (62), can sometimes be indicated.
When teeth are very badly worn, as in patients affected by severe forms of nocturnal tooth grinding (Fig. 7.8a), it is very often necessary to increase the OVD for prosthetic reasons, mainly related to the patient’s insufficient interarch space or special aesthetic needs.
This approach can be adopted without hesitation, given that in asymptomatic subjects, who are often the ones that present with bruxism-related tooth wear (63), problems adapting to the new OVD are very unlikely to occur (Fig. 7.8b).

Fig.s 7.8a-b • Prosthetic rehabilitation in a patient affected by a severe form of nocturnal tooth grinding. a) Effects of nocturnal tooth grinding. b) Definitive restoration where a slight increase in the occlusal vertical dimension was needed for prosthetic reasons. The patient adapted to the new OVD without developing any clinical symptoms.
In patients who, instead, do have TMD symptoms, temporarily increasing the OVD by means of occlusal splints worn 24 hours a day can have real benefits, bringing relief of both muscle and joint pain (Fig. 7.9).

Fig. 7.9 • Relaxation occlusal splint. The beneficial effects seem to be due largely to the occlusal lifting and the consequent shift in the joint loading area, as well as to the lengthening of the contracted muscle fibres.
Indeed, contrary to what was believed until a few years ago, i.e., that occlusal splints do not offer biological advantages but are merely a sort of “crutch” and a source of psychological support for patients with TMDs (64), it has recently been hypothesised that the improvement in symptoms obtained using occlusal splints may be due to objective and not just subjective benefits (20).
Indeed, there is some experimental evidence supporting the hypothesis that occlusal splints, by increasing OVDs and modifying the relations between the joint components, can favour a reorganisation of muscle fibre recruitment patterns (65) and a shift in the area of highest loading of the joint (66). These effects, obtained largely simply through occlusal lifting (Fig.s 7.10a-b), can explain a series of frequent clinical observations, for example:
- Moderate increases in the OVD are well tolerated by the healthy masticatory system and are often even perceived as functional improvements (67);
- Different types of occlusal splint, often fabricated to achieve different objectives, seem to show comparable success rates in the management of TMD (68);
- The clinical efficacy of occlusal splints created according to theories with no scientific basis (e.g. neuromuscular orthotic appliances) is similar to that of other types of splint (69).

Fig. 7.10 • The fabrication of an occlusal splint. a) Plaster models mounted on a basic average-value articulator. b) Vertical dimension increase planned on the articulator. Courtesy of Massimo Labori, Dental Technician, Massa, Italy.
In view of these considerations, it seems clear that, even in these times of evidence-based medicine, numerous aspects of topics “central” to many branches of dentistry still remain to be clarified, such as, indeed, centric relation and the occlusal vertical dimension.
However, for the moment, the best approach to occlusion seems to be, also in the prosthetic field, that of low-tech, conservative methods that are as simple as possible, as has already been underlined in relation to the management of TMDs (18).
GUIDELINES FOR “TMD-SAFE” PROSTHETIC IMPLANT REHABILITATIONS
Obviously, the earlier remarks about the need to minimise the importance of assessing occlusion in the context of TMD and orofacial pain do not give us licence to ignore or avoid assessing this aspect in the planning and execution of prosthetic or prosthetic implant work (70).
Indeed, even though the stomatognathic system has remarkable capacities of adaptation and accommodation (71), the potential risk of iatrogenically provoked TMD symptoms arising in the wake of extensive rehabilitations nevertheless remains.
The rare cases in which this happens are those in which the position of the condyle and the usual OVD of a healthy individual are not respected and, also, are modified beyond the system’s tolerance threshold.
Any repositioning planned on the basis of some preconceived idea of “normal” relations between the components of the stomatognathic system can, potentially, backfire, especially considering that there exists no way of predicting how the system might react (72).
Therefore, as a general rule, modifications should be carried out only for valid prosthetic reasons, as occurs in orthodontics and orthognathic surgery (73); furthermore, the extent of these modifications should be kept to the minimum and they should be performed over a longer period of time (Fig. 7.11).

Fig. 7.11 • Occlusal repositioning and modifications of the OVD should be carried out in maximum safety, exploiting the stomatognathic system’s remarkable capacity for accommodation. It is necessary to consider the timing of interventions and the entity of modifications as, interventions that are carried out in too short a period of time and modifications that are too great compared with the system’s usual position are the main risk factors for the appearance of iatrogenic TMD symptoms following orthodontic treatments.
On the basis of these considerations, there seem to emerge at least two potential risk situations: 1. Rehabilitations that hinge on a rest position identified by the absence of EMG activity, given the considerable increase in OVD that can sometimes be required in these cases; 2. Full-arch restorations with immediate loading, on account of the fact that these solutions allow the system very little time to adapt to the possible new position, or even no time at all in the case of some protocols in which the definitive prosthesis is delivered directly, without a provisional one. In the first case, there can emerge, for example, paradoxical situations of anterior open bite following rehabilitation of the four quadrants (Fig. 7.12), while in the second, OVD or centric relation errors may not be clearly apparent on a first clinical inspection.

Fig. 7.12 • Patient with anterior open bite following prosthetic rehabilitation of the four quadrants in which the OVD was identified using the myocentric method. This technique is associated with the risk of excessive increases in the OVD and the appearance of clinical symptoms, usually excessive muscle tension and pressure pain.
The clinical symptom that characterises these situations is the onset, sometimes immediate and sometimes more delayed, of pain in the temporal area; this may be described by the patient as a feeling of oppressive tension, or it may be detectable by muscle palpation (Fig. 7.13). In such cases, an initial, conservative approach could be to submit the patient to a course of treatment with myorelaxant drugs (74), although in many cases it is necessary to remake the prosthesis.

Fig. 7.13 • Palpation of the medial portion of the temporal muscle.
As far as the occlusal design is concerned, there are very few grounds for believing that one particular design is superior to others in preventing the onset of TMD symptoms (22). There are, of course, certain characteristics, such as, for example, the need for bilateral distribution of the posterior contacts, whose importance to dental stability and the patient’s subjective comfort are supported by evidence derived from clinical research. In the absence of such evidence, the most accepted and best rules of clinical practice currently available indicate simply that the dental prosthesis should satisfy the patient’s priorities and expectations as regards his/her dental and facial anatomy (72). The clinical view that certain occlusal schemes (primarily, maximum intercuspation in retruded condyle position; canine guide) are indicated in order to optimise function and prevent the onset of clinical symptoms have never been scientifically demonstrated (75). The literature data, as a whole, seem to suggest that somatosensory information plays an important role in the control of all mandibular movements, and that this information is used by the mandibular motor system to learn new tasks and adapt to occlusal modifications (76). Therefore, thanks to the neuroplasticity of the motor control system, relatively small occlusal modifications are well accomodated without resulting in clinical symptoms or imbalances (77). It is interesting to note that this neuroplasticity provides a basis for explaining the fact that different occlusal schemes (different protrusive guides; different types of antagonistic contacts; different characteristics of contacts in centric occlusion; different CR-MI position relations) are all enthusiastically extolled by their proponents and, in fact, are similarly able to optimise function (Fig. 7.14) (78,79).
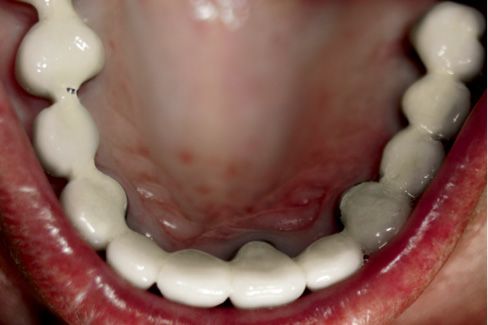
Fig. 7.14 • Occlusal view of a full-arch, fixed prosthetic restoration supported by both natural teeth and implants. The occlusal design must ensure, as its main features, freedom of movement and punctiform contacts bilaterally.
In conclusion, a prosthetic implant rehabilitation that optimises function and is “TMD-safe” must reflect a few simple principles, summarised in Box 7.3.

Box 7.3 • Technical principles of “TMD-safe” prosthetic implant rehabilitations.
PROSTHETIC IMPLANTS IN PATIENTS WITH TMD
In routine clinical practice, it is not uncommon to be asked to perform prosthetic or prosthetic implant rehabilitations in patients with signs and/or symptoms of TMD.
Since, as explained in the section on occlusion and TMDs, neither the literature nor clinical observations support the hypothesis that an irreversible occlusal treatment can be considered the standard approach to the treatment of TMDs, and given that there exists a high risk of planning errors when defining the usual occlusion of a patient with musculoskeletal pain, it is fundamental to manage the TMD-related symptoms before performing any definitive occlusal intervention (80).
In particular, the presence of TMJ and/or masticatory muscle pain and a limited range of joint movement must be considered absolute contraindications to prosthetic implant treatments and to the definitive modification of a patient’s occlusal schemes.
Instead, the presence of clinical signs, such as “click” sounds that indicate anterior displacement of the articular disc from its normal position in relation to the condyle (Fig. 7.15), do not per se constitute contraindications to occlusal rehabilitations, even though such conditions should be considered to warrant specialist investigation.

Fig. 7.15 • Magnetic resonance image of the TMJ showing anterior displacement of the disc. According to the most recent literature guidelines, this is not necessarily to be considered a pathological finding and it constitutes neither an indication for nor a contraindication to occlusal rehabilitations.
Parafunctional subjects, too often generically referred to as “bruxers”, should also be carefully assessed in an attempt to distinguish between grinding and clenching activities.
Indeed these two activities, quite apart from having different aetiopathogenetic substrates (81,82), undoubtedly have different effects on the components of the stomatognathic system (83).
In the case of prosthetic implant rehabilitations, the complications relating to grinding activities concern only technical-mechanical aspects (prosthetic implant materials and components) (84), whereas clenching activities can be associated with the appearance of masticatory muscle and/or TMJ fatigue and pain (85), which can be aggravated in cases in which the rehabilitation considerably modifies the occlusal pattern (86). All these aspects considered, there exist numerous approaches to the management of TMD symptoms.
The key concept to underline is the fact that TMDs must be considered from a medical and not just a purely dental perspective.
In other words, TMDs are not occlusal disorders, but rather musculo-skeletal disorders whose clinical signs and symptoms need to be managed in line with the standards followed for similar disorders in other specialist settings (e.g. in rheumatological, physical medicine and orthopaedic settings) and, in the most severe cases, with support from other specialists for the management of chronic pain (e.g. neurologists, psychiatrists, psychologists) (87).
The treatment of TMDs is based on the use of reversible and conservative therapeutic methods, the most frequently used being physiotherapy, pharmacotherapy, cognitive-behavioural therapy and occlusal splints (17).
Even though only a very small minority of patients require surgical correction, there now also exist temporomandibular joint replacement prostheses, which have given encouraging results (88).
As regards treatments designed to relieve pain in patients with inflammatory-degenerative disorders of the TMJ, or to increase the range of joint motion in patients with acute closed lock of the TMJ, the recent literature supports the efficacy, in the medium- to long-term, of arthrocentesis (or wash out) of the joint, followed by injections of medications such as cortisone or hyaluronic acid (89-91), also performed using an innovative single-needle technique (92) (Fig. 7.16).

Fig. 7.16 • Arthrocentesis of the TMJ using a single-needle technique according to Guarda-Nardini et al.92 The arthrocentesis of the joint, performed under pressure, is usually followed by the injection of medications such as cortisone or hyaluronic acid, and the literature supports the use of this treatment both in patients with acute closed lock of the TMJ and in those with inflammatory-degenerative disorders.
Individuals affected by TMDs display hypervigilance to many events and stressors, both local and systemic (93).
This hypervigilance plays a very important role both in the onset and in the maintenance of the symptoms, and it manifests itself as increased sensitivity to stress (94) and as an increased tendency to respond abnormally even to non-nociceptive stimuli (95).
This characteristic explains the results of the study mentioned earlier in which the placement of acute occlusal interferences resulted in the appearance of clinical symptoms in individuals with a history of TMDs, but not in a sample of healthy controls (36).
Thus, even though occlusion is not a key factor in the aetiopathogenesis of TMDs, clinical experience nevertheless shows that patients with a history of TMDs have an extremely delicate stomatognathic equilibrium and can be hypersensitive to occlusal scheme modifications.
This observation provides further justification for minimising the importance of occlusion as a therapeutic target and for advocating simple rehabilitation schemes that do not grossly alter the previous occlusal status, in order to prevent patients from developing occlusal hypervigilance (i.e. from focusing excessively on their occlusion) (96,97).
CONCLUSIONS
The relationship, if one exists, between TMDs and rehabilitations using prosthetic implants does not seem to differ, in any aspect, from the relationship between TMDs and any procedure that intervenes on the dental occlusion. In short, the literature evidence is sufficient to allow a series of conclusions to be drawn:
- TMDs are not occlusal disorders and therefore should not be treated with extensive occlusal rehabilitation interventions.
- Because the stomatognathic system has such a remarkable capacity for accommodation, its adaptability, in most clinical situations, is guaranteed.
- There is no such thing as the ideal articular position or ideal occlusal design, superior to all the others and valid for every single individual; for this reason, it is best to follow the rule “simple is best”, avoiding the use of preconceived and “forced” or artificial rehabilitation protocols.
- Prosthetic implant procedures give rise to clinical symptoms but only if their execution repeatedly and considerably modifies the patient’s usual occlusal scheme.
- The clinical symptoms of individuals with TMDs who require or request rehabilitation with prosthetic implant procedures for prosthetic, aesthetic or functional reasons must be carefully managed using internationally accepted approaches (e.g. occlusal splints, physiotherapy, pharmacotherapy, injections); moreover, definitive rehabilitation can only be undertaken after a symptom-free period.
BIBLIOGRAPHY
1. McNeill C.: “Management of temporomandibular disorders: concepts and controversies”. J Prosthet Dent 1997; 77: 510-522.
2. Manfredini D, Guarda-Nardini L.: “TMD classification and epidemiology. In: Manfredini D (Ed). Current concepts on temporomandibular disorders. Berlin, Quintessence Publishing, 2009.
3. Rutkiewicz T, Kononen M, Suominen-Taipale L, Nordblad A, Alanen P.: “Occurrence of clinical signs of temporomandibular disorders in adult Finns”. J Orofac Pain 2006; 20: 208-217.
4. Laskin DM.: “Etiology of the pain-dysfunction syndrome”. J Am Dent Assoc 1969; 79: 147-153.
5. Leresche L, Drangsholt M.: “Epidemiology of orofacial pain: prevalence, incidence, and risk factors”. In: Sessle BJ, Lavigne GJ, Lund JP, Dubner R (Eds). Orofacial Pain. From basic science to clinical management. 2nd ed. Chicago, Quintessence Publishing, 2008. pp13-18.
6. Manfredini D, Guarda-Nardini L, Piccotti F, Ferronato G.: “Prevalence and age distribution patterns of RDC/TMD diagnoses in an Italian patients population”. J Dent 2010; 38: 392-399.
7. Yap AU, Dworkin SF, Chua EK, List T, Tan KB, Tan HH.: “Prevalence of temporomandibular disorder subtypes, psychologic distress, and psychosocial dysfunction in Asian patients”. J Orofac Pain 2003; 17: 21-8.
8. Manfredini D, Chiappe G, Bosco M.: “Research Diagnostic Criteria for Temporomandibular Disorders (RDC/TMD) axis I diagnosis in an italian patients population”. J Oral Rehabil 2006; 33: 551-8.
9. Okeson JP.: “The classification of orofacial pains”. Oral Maxillofac Surg Clin N Am 2008; 20: 133-144.
10. De Leeuw R.: “The American Academy of Orofacial Pain. Orofacial pain: guidelines for assessment, diagnosis, and management”. Quintessence Publishing, Chicago, USA, 2008.
11. De Boever JA, Nilner M, Orthlieb JD, Steenks MH.: “Recommendations by the EACD for examination, diagnosis and management of patients with temporomandibular disorders and orofacial pain by the general dental practitioner”. J Orofac Pain 2008; 22: 268-278.
12. Dworkin SF, LeResche L.: “Research diagnostic criteria for temporomandibular disorders: Review, criteria examinations and specifications, critique”. J Craniomandib Disord Fac Oral Pain 1992; 6: 301-355.
13. Schmitter M, Ohlmann B, John MT, Hirsch C, Rammelsberg P.: “Research Diagnostic Criteria for Temporomandibular Disorders: a calibration and reliability study”. J Craniomandib Pract 2005; 23: 212-218.
14. Gonzalez YM, Greene CS, Mohl ND.: “Technological devices in the diagnosis of temporomandibular disorders”. Oral Maxillofac Surg Clin North Am 2008; 20: 211-220.
15. Manfredini D, Guarda-Nardini L.: “Agreement between Research Diagnostic Criteria for Temporomandibular Disorders and Magnetic Resonance diagnoses of temporomandibular disc displacement in a patient population”. Int J Oral Maxillofac Surg 2008; 37: 612-616.
16. Schmitter M, Kress B, Leckel M, Henschel V, Ohlmann B, Rammelsberg P.: “Validity of temporomandibular disorder examination procedures for assessment of temporomandibular joint status”. Am J Orthod Dentofac Orthop 2008; 133: 796-803.
17. Greene CS.: “Concepts of TMD etiology: effects on diagnosis and treatment”. In: Laskin DM, Greene CS, Hylander WL (eds). TMDs. An evidence-based approach to diagnosis and treatment. Quintessence Publishing, Chicago, USA, 2006. pp 219-228.
18. Stohler CS, Zarb GA.: “On the management of temporomandibular disorders: A plea for a low-tech, high-prudence therapeutic approach”. J Orofac Pain 1999; 13: 255-261.
19. Stohler CS.: “Temporomandibular joint disorders – The view widens while therapies are constrained”. J Orofac Pain 2007;21: 261.
20. Turp JC, Schindler HJ.: “Occlusal therapy of temporomandibular pain”. In: Manfredini D (Ed). Current concepts on temporomandibular disorders. Berlin, Quintessence Publishing, 2010. pp 359-382.
21. Greene CS.: “Science transfer in orofacial pain”. In: Lund JP, Lavigne GJ, Dubner R, Sessle BJ (eds). Orofacial pain. From basic science to clinical management. Quintessence Publishing, Chicago, USA, 2008. pp 281-286.
22. Turp JC, Greene CS, Strub JR.: “Dental occlusion: a critical reflection on past, present and future concepts”. J Oral Rehabil 2008; 35: 446-453.
23. Forssell H, Kalso E.: “Application of principles of evidence-based medicine to occlusal treatment for temporomandibular disorders: are there lessons to be learned?” J Orofac Pain 2004; 18: 9-22.
24. Koh H, Robinson PG.: “Occlusal adjustment for treating and preventing temporomandibular joint disorders”. J Oral Rehabil 2004; 287-292.
25. Forssell H, Kalso E, Koskela P, Vehmanen R, Puukka P, Alanen P.: “Occlusal treatments in temporomandibular disorders: a qualitative systematic review of randomized controlled trials”. Pain 1999; 83: 549-560.
26. Clark GT, Tsukiyama Y, Baba K, Watanabe T.: “Sixty-eight years of experimental occlusal interference studies: what have we learned?” J Prosthet Dent 1999; 82: 704-713.
27. Costen JB.: “A syndrome of ear and sinus symptoms dependent upon disturbed function of the temporomandibular joint”. Ann Otol Rhinol Laryngol 1934;43:1-15.
28. Annandale T.: “On displacement of the interarticular cartilage of the lower jaw and its treatment by operation”. Lancet 1887; 1: 411-4.
29. Molin C.: “From bite to mind: TMD – a personal and literature review”. Int J Prosthodont 1999; 12: 279-88.
30. Pullinger AG, Seligman DA.: “Quantification and validation of predictive values of occlusal variables in temporomandibular disorders using a multifactorial analysis”. J Prosthet Dent 2000; 83: 66-75.
31. Landi N, Manfredini D, Tognini F, Romagnoli M, Bosco M.: “Quantification of the relative risk of multiple occlusal variables for muscle disorders of the stomatognathic system”. J Prosthet Dent 2004; 92: 190-195.
32. Manfredini D, Peretta R, Guarda-Nardini L, Ferronato G.: “Role of clinically diagnosed bruxism and occlusion in TMJ pain: a logistic regression model”. J Craniomandib Pract, in press.
33. De Boever JA, Carlsson GE, Klineberg IJ.: “Need for occlusal therapy and prosthodontic treatment in the management of temporomandibular disorders. Part II: Tooth loss and prosthodontic treatment”. J Oral Rehabil 2000;27:647-659.
34. Michelotti A, Farella M, Gallo LM, Veltri A, Palla S, Martina R.: “Effect of occlusal interference on habitual activity of human masseter”. J Dent Res 2005; 84: 644-648.
35. Michelotti A, Farella M, Steenks MH, Gallo LM, Palla S.: “No effect of experimental occlusal interferences on pressare pain thresholds of the masseter muscles in healthy women”. Eur J Oral Sci 2006; 114: 167-170.
36. Le Bell Y, Jämsä T, Korri S, Niemi PM, Alanen P.: “Effect of artificial occlusal interferences depends on previous experience of temporomandibular disorders”. Acta Odontol Scand 2002;60:219-222.
37. Alanen P. “Occlusion and Temporomandibular Disorders (TMD): still unsolved question?” J Dent Res 2002; 81: 518-9.
38. Muller E, Rios Calvo MP.: “Pain and dental implantology: sensory quantification and affective aspects. Part I: at the private dental office”. Implant Dent 2001; 10: 14-22.
39. Engel E, Lachmann S, Axmann-Krcmar D.: “The prevalence of radiologic TMJ findings and self-reported orofacial pain in a patients group wearing implant dentures”. Int J Prosthod 2001; 14: 120-126.
40. De Boever JA, De Laat A.: “Prosthetic rehabilitation in TMD patients”. In: Manfredini D (Ed). Current concepts on temporomandibular disorders. Berlin, Quintessence Publishing, 2009.
41. Türp JC, Strub JR.: “Prosthetic rehabilitation in patients with temporomandibular disorders”. J Prosthet Dent 1996;76:418-423.
42. Rinchuse DJ, Kandasamy S.: “Centric relation: A historical and contemporary orthodontic perspective“. J Am Dent Assoc 2006;137:494-501.
43. The Academy of Prosthodontics: “The Glossary of Prosthodontic Terms, Eighth Edition (GPT-8)”. J Prosthet Dent 2005;94:10-92.
44. Rinchuse DJ, Kandasamy S.: “Orthodontics and TMD management”. In: Manfredini D (Ed). Current concepts on temporomandibular disorders. Berlin, Quintessence Publishing, 2010. pp 429-445.
45. McNamara JA, Seligman DA, Okeson JA.: “Occlusion, orthodontic treatment, temporomandibular disorders: a review”. J Orofac Pain 1995;9:73-90.
46. Rinchuse DJ, Kandasamy S.: “Articulators in orthodontics: an evidence-based perspective”. Am J Orthod Dentofac Orthop 2006;129:299-308.
47. Pullinger AG, Seligman DA, John MT, Harkins S.: “Multifactorial modeling of temporomandibular anatomic and orthopedic relationships in normal versus undifferentiated disk displacement joints”. J Prosthet Dent 2002; 87: 289-297.
48. Jankelson B, Hoffman GM, Hendron JA Jr.: “The physiology of the stomatognathic system”. J Am Dent Assoc 1952; 46: 375-86.
49. Greene CS.: “The role of biotechnology in TMD diagnosis”. In: Laskin DM, Greene CS, Hylander WL (eds). TMDs. An evidence-based approach to diagnosis and treatment. Quintessence Publishing, 2006. pp 193-202.
50. Klasser GD, Okeson JP.: “The clinical usefulness of surface electromyography in the diagnosis and treatment of temporomandibular disorders”. J Am Dent Assoc 2006; 137: 763-71.
51. Suvinen TI, Kemppainen P.: “Review of clinical EMG studies related to muscle and occlusal factors in healthy and TMD subjects”. J Oral Rehabil 2007; 34: 631-44.
52. Suvinen T, Reade P, Kononen M, Kemppainen P.: “Vertical jaw separation and masseter muscle electromyographic activity: a comparative study between asymptomatic controls and patients with temporomandibular pain and dysfunction”. J Oral Rehabil 2003;30:765–772.
53. Michelotti A, Farella M, Vollaro S, Martina R.: “Mandibular rest position and electrical activity of the masticatory muscles”. J Prosthet Dent 1997;78:48–53.
54. Moyers RE, Wainright RL.: “Skeletal contributions to occlusal development”. In: McNamara JA Jr (ed). The Biology of Occlusal Development, monograph 7, Craniofacial Growth Series. Ann Arbor, University of Michigan Press, 1977.
55. Ainamo A.: “The continuous eruption in adult man and its influence on the width of the anatomical attached gingival”. Doctoral Thesis, University of Helsinki, 1977.
56. McGee GF.: “Use of facial measurements in determining vertical dimension. J Am Dent Assoc 1947;35:342-350.
57. Pound E.: “Controlling anomalies of vertical dimension and speech. J Prosthet Dent 1976;36:124.
58. Jankelson B, Pulley ML.: “Electromyography in clinical dentistry”. Seattle: Myotronics Research, 1984.
59. Shanahan TEJ.: “Physiologic vertical dimension and centric relation”. J Prosthet Dent 2004; 91: 206-209.
60. Fayz F, Eslami A.: “Determination of occlusal vertical dimension: a literature review”. J Prosthet Dent 1988;59:321-3.
61. Johansson A, Johansson AK, Omar R, Carlsson GE.: “Rehabilitation of the worn dentition”. J Oral Rehabil 2008; 35: 548-566.
62. Klasser GD, Greene CS.: “Oral appliances in the management of temporomandibular disorders”. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2009;107:212-223.
63. John MT, Frank H, Lobbezoo F, Drangsholt M, Dette KE.: “No association between incisal tooth wear and temporomandibular disorders”. J Prosthet Dent 2002; 87: 197-203.
64. Dao TT, Lavigne GJ.: “Oral splints: The crutches for temporomandibular disorders and bruxism?” Crit Rev Oral Biol Med 1998; 9: 345-361.
65. Van Eijden TMGV, Blanksma NG, Brugman P.: “Amplitude and timing of EMG activity in the human masseter muscle during selected motor tasks”. J Dent Res 1993;72:599-606.
66. Ettlin DA, Mang H, Colombo V, Palla S, Gallo LM.: “Stereometric assessment of TMJ space variation by occlusal splints”. J Dent Res 2008;87:877-881.
67. Rivera-Morales WC, Mohl ND.: “Relationship of occlusal vertical dimension to the health of the masticatory system”. J Prosthet Dent 1991;65:547-553.
68. Türp JC, Komine F, Hugger A.: “Efficacy of stabilization splints for the management of patients with masticatory muscle pain. A qualitative systematic review”. Clin Oral Invest 2004;8:179-194.
69. Cooper BC, Kleinberg I.: “Establishment of temporomandibular physiological state with neuromuscular orthosis treatment affects reduction of TMD symptoms in 313 patients”. Cranio 2008; 26: 104-117.
70. Rinchuse DJ, Rinchuse DJ, Kandasamy S.: “Evidence-based versus experience-based views on occlusion and TMD. Am J Orthod Dentofac Orthop 2005; 127: 249-254.
71. Sessle BJ.: “Biological adaptation and normative values”. Int J Prosthodont 2003; 16 (Suppl): 72-73.
72. Klineberg I, Stohler CS.: “Interface of occlusion”. Int J Prosthodont 2003; 16 (Suppl): 89-90.
73. Sforza C, Peretta R, Grandi G, Ferronato G, Ferrario VF.: “Soft tissue facial planes and masticatory muscle function in skeletal class III patients before and after orthognathic surgery treatment”. J Oral Maxillofac Surg 2008; 66:691-698.
74. Manfredini D, Romagnoli M, Cantini E, Bosco M.: “Efficacy of tizanidine hydrochloride in the treatment of myofascial face pain. Minerva Medica 2004; 95: 165-71.
75. Klineberg I, Kingston D, Murray GM.: “The bases for using a particular occlusal design in tooth and implant-borne reconstructions and complete dentures”. Clin Oral Impl Res 2007; 18 (Suppl. 3): 151–167.
76. Sessle BJ.: “Mechanisms of oral somatosensory and motor functions and their clinical correlates”. J Oral Rehabil 2006; 33: 243–261.
77. Ren K, Dubner R.: “Central nervous system plasticity and persistent pain”. J Orofac Pain 1999; 13: 155-163.
78. Beyron H.: “Occlusion: point of significance in planning restorative procedures. J Prosthet Dent 1973; 30: 641-652.
79. Carlsson GE.: “Dental occlusion: modern concepts and their application in implant prosthodontics”. Odontology 2009; 97: 8-17.
80. Stohler CS.: “Management of dental occlusion”. In Laskin DM, Greene CS, Hylander WL (eds). TMDs. An Evidence-Based Approach to Diagnosis and Treatment. Chicago: Quintessence, 2006:403-411.
81. Macaluso GM, Guerra P, Di Giovanni G, Boselli M, Parrino L, Terzano MG.: “Sleep bruxism is a disorder related to periodic arousal during sleep”. J Dent Res 1998; 77: 565-73.
82. Manfredini D, Lobbezoo F.: “Role of psychosocial factors in the etiology of bruxism”. J Orofac Pain 2009; 23: 153-166.
83. Manfredini D, Lobbezoo F.: “Bruxism and TMD”. In: Manfredini D (Ed). Current concepts on temporomandibular disorders. Berlin, Quintessence Publishing, 2010. pp 135-152.
84. Lobbezoo F, Brouwers JEIG, Cune MS, Naeije M.: “Dental implants in patients with bruxing habits”. J Oral Rehabil 2006; 33: 152-159.
85. Svensson P, Jadidi F, Arima T, Baad-Hansen L, Sessle BJ.: “Relationships between craniofacial pain and bruxism”. J Oral Rehabil 2008; 35: 524-47.
86. Racich MJ.: “Orofacial pain and occlusion: Is there a link? An overview of current concepts and the clinical implications”. J Prosthet Dent 2005; 93: 189-196.
87. Manfredini D.: “Fundamentals of TMD management”. In: Manfredini D (Ed). Current concepts on temporomandibular disorders. Berlin, Quintessence Publishing, 2010. pp 305-317.
88. Guarda-Nardini L, Manfredini D, Ferronato G.: “Temporomandibular joint total replacement prosthesis: current knowledge and considerations for the future”. Int J Oral Maxillofac Surg 2008; 37: 103-110.
89. Al-Belasy FA, Dolwick MF.: “Arthrocentesis for the treatment of temporomandibular joint closed lock: a review article”. Int J Oral Maxillofac Surg 2007; 36: 773-82.
90. Guarda-Nardini L, Stifano M, Brombin C, Salmaso L, Manfredini D.: “A one-year case series of arthrocentesis with hyaluronic acid injections for temporomandibular joint osteoarthritis”. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2007; 103: 14-22.
91. Manfredini D, Bonnini S, Arboretti R, Guarda-Nardini L.: “Temporomandibular joint osteoarthritis: an open label trial of 76 patients treated with arthrocentesis plus hyaluronic acid injections”. Int J Oral Maxillofac Surg 2009; 38: 827-834.
92. Guarda-Nardini L, Manfredini D, Ferronato G.: “Arthrocentesis of the temporomandibular joint: a proposal for a single-needle technique”. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2008; 106: 483-6.
93. Sarlani E, Grace EG, Reynolds MA, Greenspan JD.: “Evidence for up-regulated central nociceptive processing in patients with masticatory myofascial pain”. J Orofac Pain 2004;18:41–55.
94. Manfredini D, Bandettini di Poggio A, Cantini E, Dell’Osso L, Bosco M.: “Mood and anxiety psychopathology and temporomandibular disorder: a spectrum approach”. J Oral Rehabil 2004; 31: 933-940.
95. Maixner W, Fillingim R, Sigurdsson A, Kincaid S, Silva S.: “Sensitivity of patients with painful temporomandibular disorders to experimentally evoked pain: evidence for altered temporal summation of pain”. Pain 1998; 76: 71-81.
96. Palla S.: “Mioartropatie del sistema masticatorio e dolori orofacciali”. Ed. RC Libri, 2001.
97. Dao TTT.: “Musculoskeletal disorders and the occlusal interface”. Int J Prosthodont 2003; 16 (suppl): 83-84.
CHAPTER 8
BRUXISM AS A RISK FACTOR IN IMPLANT PROSTHETICS
Daniele Manfredini
Visiting professor, Department of Maxillofacial Surgery, University of Padua
The American Academy of Sleep Medicine defines bruxism as a stereotyped oral motor disorder characterised by sleep-related grinding and/or clenching of the teeth (1), while the American Academy of Orofacial Pain has extended the definition to the presence of the same characteristics during wakefulness (2). Of all the parafunctional activities of the stomatognathic system, bruxism is considered the most detrimental, causing tooth wear and representing a potential risk factor for the appearance of signs and symptoms of temporomandibular disorders (TMD) (3). Even though bruxism has major clinical consequences, a discussion on its role in the different branches of dentistry must first seek to clarify certain complex aspects of its aetiology.
Indeed, the aetiology of bruxism is undoubtedly an extremely complex and controversial topic. Most authors agree that it is a disorder of central origin, as shown by many literature reviews which reveal a tendency to attach less importance to the role of peripheral (e.g. occlusal) factors, and more to central ones (e.g. arousal responses, stress, anxiety, temperamental traits) (4-9). Only a small proportion of the most recent studies (around 10%) explore the role of morphological factors in the aetiology of bruxism, while a slightly higher proportion (around 20%) focus on the role of psychosocial factors. The vast majority of studies (around 70%), however, consider the role of potential aetiological factors classifiable as pathophysiological (10). These percentages confirm the trends commonly observed in the research setting, where attention is turning away from the role of occlusion and increasing importance is being attached to biomedical and biopsychosocial models. Psychosocial factors can play a role in the aetiopathogenesis of bruxism, to varying degrees in different forms of grinding or clenching occurring in the different stages of the sleep-wake cycle (11-13) (Box 8.1), while occlusal interferences have not been found to show a significant association with the presence of bruxism (14) (Table 8.1); at most, the placement of acute occlusal interferences can constitute a stimulus capable of triggering parafunctional activity in subjects with occlusal hypervigilance (15) (Fig. 8.1).

Box 8.1 • Considerations on the role of psychosocial factors in the aetiopathogenesis of bruxism (Manfredini and Lobbezoo, 2009) (12).

Table 8.1 • Significance (P), accuracy, specificity, sensitivity, PPV and NPV of single occlusal characteristics in predicting the presence of bruxism, and percentage of total variance (R2) explained by the multifactorial model including the variables with the greatest predictive power (indicated by *). The very low percentage of variance explained by the occlusal variables is a clear demonstration of their marginal role in the aetiopathogenesis of bruxism (modified from Manfredini et al., 2004) (14).

Fig. 8.1 • Introduction of acute occlusal interferences on teeth 44, 45, 46. According to the patient, the aim of the treating dentist had been to “reposition the jaw” for reasons (not better explained) linked to the need to resolve her headache symptoms. It subsequently emerged that these symptoms were part of a complex clinical picture of recurrent migraine without aura, for the resolution of which there are no justifiable grounds for using occlusal treatment. Furthermore, the patient (who has occlusal hypervigilance) reported worsening of a parafunctional habit, both during wakefulness and during sleep, following the introduction of the occlusal modifications.
Over the years, in efforts to synthesise knowledge of this topic, various aetiological theories have been proposed.
At present, the most plausible explanation of the aetiopathogenesis of bruxism hypothesises the existence of a multifactorial model in which psychosocial and pathophysiological factors interact with peripheral morphological stimuli (16-18).
In short, one or more factors (stress, anxiety, genetics, neurochemical factors, arousal response, deficits in dopaminergic transmission, factors linked to the quality of sleep, oral and dental anatomy) may, in different ways and to different degrees, contribute to increasing the frequency, duration and intensity of normal masticatory muscle activity during sleep, in accordance with the “bruxism generator model” (19).
While this hypothesis fits perfectly into a context of a multifactorial aetiology of bruxism, there nevertheless remain many aspects to be clarified as regards the weight of the various factors in the different forms.
Indeed, study of the aetiology of bruxism is complicated by the existence of diagnostic framing difficulties that have always constituted an obstacle to research in this field.
At the current state of knowledge, it seems fairly apparent that the pathogenesis of sleep bruxism is different from that of bruxism during wakefulness (12), but it is difficult to distinguish between the two conditions clinically (20).
Similarly, there is a need for a clearer distinction between objectively diagnosed bruxism and subjective, patient-reported bruxism, as the latter is liable to be over-diagnosed as a result of preconceived ideas, on the part of either the patient or the examining physician (21).
Currently, polysomnographic recording (PSG) is considered the gold standard for the diagnosis of sleep bruxism, but the use of this technique is limited by the high costs involved and the shortage of adequately equipped sleep laboratories (22).
For this reason, a number of diagnostic hypotheses based on electromyographic recording of masticatory muscle activity during sleep have recently been advanced (23,24), even though it will not be possible to collect data of real clinical value until we have portable EMG devices able to record the nocturnal activity of various muscle groups, and not just that of the masseter muscles (25).
Therefore, at present, the clinical approach to the diagnosis of bruxism, albeit incomplete and lacking the ability to distinguish between the different forms of the disorder, continues to be the simplest assessment and data collection method available, and the most widespread.
THE CLINICAL DIAGNOSIS OF BRUXISM
The literature proposes various methods of diagnosing bruxism (Box 8.2).

Box 8.2 • Proposed methods of diagnosing bruxism.
However, given that, for the reasons already given, both prolonged (hours-long) electromyography of the masseter muscles and PSG are unsuitable for routine use, clinical assessment, combined with the use of questionnaires, remains the best of the diagnostic options currently available to the clinician for identifying bruxers.
Because questionnaires, even though many different types have been proposed, generally do not provide sufficient diagnostic accuracy, their use without integrated objective approaches is not recommended (20).
The many different questionnaires constructed in order to detect bruxist activity are all based on a series of common questions (e.g. Does your partner say that you tend to grind your teeth loudly at night? Do you have a sensation of pain and/or tenderness in your jaw muscles on awakening? Do you ever wake up at night aware that you are clenching your teeth? Do you ever have tooth or gum pain on awakening? During the day, do you tend to contract your jaw muscles, clenching your teeth? Do you ever grind your teeth during the daytime?); the limitation of such instruments, however, is the subjective nature of the responses, and thus of the “diagnosis”.
Furthermore, the few studies that have attempted to quantify the diagnostic validity of questionnaires as a means of diagnosing bruxism, while producing generally encouraging results, lack methodological rigour, failing to include “control” assessments conducted using reference standards (e.g. PSG, EMG) (27,28).
These questions, also commonly used during clinical history-taking, are based on a few preconceptions that become sources of bias in the clinical and research settings.
For example, many of the questions are based on the assumptions that bruxist activity produces muscle pain/tenderness and/or manifests itself through sounds caused by tooth-grinding.
The literature has shown that neither of these assumptions is necessarily true and thus that their use as the basis for the formulation of diagnostic criteria will result in a low positive predictive value.
Indeed, the association between bruxism and muscle, tooth or gum pain is much more complex than was believed in the past, given that the proportion of bruxers who develop pain is smaller than that of bruxers who do not (3,29,30).
Similarly, individuals whose nocturnal tooth-grinding is so loud that they wake themselves or their bed partner usually account for only a small proportion of the complex category of subjects with “bruxism behaviour”, given that at least 80% of episodes of bruxism do not produce noise, i.e. are clenching activities (19).
For years, the use of self-report diagnostic instruments based on the above questions fed the belief that bruxism is associated with muscle pain (31).
In actual fact, the results of many literature studies that describe a strong association between bruxism and myofascial pain were probably influenced by the fact that the presence of muscle pain was considered a potential diagnostic criterion for bruxism (32).
Indeed, experimental attempts to reproduce bruxism-induced muscle pain have failed to confirm the existence of an unequivocal cause-effect relationship between prolonged bruxist activity and myofascial pain (33,34), to the point that the time is now ripe for a careful review of the literature on this topic (3).
Clinicians’ convictions constitute a further potential source of bias in the clinical-anamnestic assessment of bruxism: when a clinician believes a patient to be a bruxer, the likelihood of that same patient replying affirmatively to the above questions has been found to be increased, regardless of how the questions are presented: in the form of a self-report questionnaire or during history-taking (35).
In view of all these considerations, it is clearly fundamental that any information furnished by patients be supplemented with the results of objective examinations able to detect the presence of bruxist activities.
Tooth wear has always been considered the clinical hallmark of bruxism (36).
In truth, tooth wear, which is linked to functional activities (e.g. mastication) and parafunctional activities (e.g. bruxism, nail-biting, chewing of objects), is due to a combination of phenomena – abrasion, erosion and friction – which are conditioned by jaw movement patterns (e.g. laterotrusion and protrusion guides) and external factors, such as diet (e.g. consumption of soft or acid drinks) and concomitant disorders (e.g. gastric reflux, eating behaviour disorders) (37).
The literature proposes numerous systems for quantifying and objectively evaluating tooth wear, as well as assessment schemes aiming to identify the wear facets associated with different jaw movements (Fig. 8.2).

Fig. 8.2 • Example of areas of contact between posterior teeth during lateral grinding movements and of the potential tooth wear caused by such movements. The blue dots indicate the areas of laterotrusive contact and the yellow dot the area of mediotrusive contact.
The most widely used classification systems, apart from the seminal one by Murphy (38), which, being shown to be reliable, has stood the test of time (39), include the ones proposed by Johansson et al., who, in a series of studies, produced a scale of tooth wear severity, separately considering the different surfaces of the anterior teeth and the occlusal/incisal surface (40,41), by Ekfeldt et al., who attempted to introduce an individual tooth wear index, independent of the number of missing teeth (42), and by Seligman and Pullinger, who described different patterns of tooth wear associated with different jaw movements (43) (Box 8.3).

Box 8.3 • Grading of progressive occlusal/incisal wear (Johansson et al., 1993)(40).
Despite the existence of criteria for evaluating tooth wear objectively, in daily clinical practice it is very difficult to discriminate between the different possible causes and, above all, to differentiate between functional and parafunctional wear (Fig.s 8.3-8.5).

Fig. 8.3 • Marked tooth wear in a 43-year-old woman. Even though the patient claims to be a bruxer, some functional wear is likely, given that the upper dental prosthesis, applied around 25 years previously, could have contributed to the excessive wear of the lower teeth. Lack posterior occlusal support may have been a contributing factor.

Fig. 8.4 • Evidence of reciprocal wear on the cusps of the canines and the incisal surface of the lateral incisors, probably resulting from a previous parafunctional habit involving eccentric jaw movements. The parafunctional habit was presumed not to be ongoing, given the patient’s negative answers on relevant questionnaires and the absence of clinical signs and symptoms. The clinical history of this patient, a 19-year-old male, suggested that the eccentric bruxism had begun in adolescence and regressed with age, in accordance with what is often reported in the literature.

Fig. 8.5 • Flattened occlusal surfaces and generalised reciprocal wear, probably the result of periods of grinding activity, as reported by the patient herself.
Moreover, another criticism often levelled at tooth wear-based bruxism assessment systems is the impossibility of determining whether the bruxism itself is still present, in other words, of establishing whether the signs of wear are due to previous or ongoing parafunctional activity.
In some cases, other clinical signs indicating bruxist activities may be present; in particular, there may be evidence of tongue scalloping (Fig. 8.6) or of marked masseter muscle hypertrophy (Fig. 8.7).

Fig. 8.6 • Tongue scalloping caused by parafunctional activity.

Fig. 8.7 • Patient with bilateral masseter hypertrophy, more marked on the left. This patient has a positive history of tooth-clenching, both during wakefulness and during sleep.
In the first case, the presence, usually bilateral, of the characteristic indentations on the tongue may be due to a “conscious” parafunctional habit on the part of the patient, who may report a tendency to chew his tongue, rather in the way some people tend to chew objects, or bite their lips or nails. Tongue scalloping is certainly a sign to be noted in a patient’s clinical records and its aetiopathogenesis should be determined through patient interview (44). As regards masseter muscle hypertrophy, the literature contains descriptions of interesting cases in which the increased muscle volume, sometimes also seen in the temporal muscles, was considered to be related to bruxism (45,46).
From a strictly pathogenetic point of view, masseter muscle hypertrophy can be considered a consequence of static clenching, which is characterised by isometric muscle activity that, if protracted for long periods of time (e.g. for many hours a day, in some cases for years), can have a training-type effect and thus result in increased muscle volume. It is also worth underlining that the effects of clenching may potentially differ according to the individual’s facial morphology, since it has been found that the subjects who develop muscle hypertrophy tend to have a brachycephalic facial type (25).
In accordance with the typical clinical observations of those involved in the management of bruxers (observations supported by an ever-increasing body of scientific evidence), occlusal splints are currently the best assessment instrument clinicians have for diagnosing the presence of parafunctional habits.
The assessment itself, which in research settings can also involve measuring the intensity and direction of forces transmitted to a splint during a parafunctional activity (47,48), consists of observation of the wear facets that appear on the splint itself in the weeks following its application (49). In most cases, splint wear, when present, is aspecific and does not make it possible to distinguish between clenching and grinding activities, even though the wear pattern can sometimes be so peculiar as to leave little doubt as to the type of mandibular movement involved (Fig. 8.8).

Fig. 8.8 • Wear facet, typical of bruxism, on an occlusal splint: the path made by the cuspid of the lower canine during lateral excursions is suggestive of grinding.
In short, diagnosing the presence and severity of bruxism continues to be a highly complex clinical problem, and the clinical classifications proposed to date inevitably reflect the limitations of what is still an exclusively clinical-anamnestic approach. At the same time, the paucity of data so far gathered in research settings suggests that electronic methods for achieving standardised measurement of bruxism will not be available on a large scale in the near future. This is despite the existence of a practical need in many dental specialties, primarily implant prosthetics, to quantify the intensity and direction of parafunctional activities.
BRUXISM AND IMPLANTOLOGY
In many specialist texts, bruxism is referred to as a risk factor capable of jeopardising the successful outcome of a rehabilitation based on prosthetic implants (50).
Few studies in the literature seek to clarify the relationship between bruxism and implants: indeed, the only experimental study conducted concludes that a prosthetic implant rehabilitation does not increase the severity of bruxism (51); indeed, all the suggestions advanced in this field seem to be based on expert opinion rather than on scientific evidence (52).
The caution that is urged when using prosthetic implants in bruxers is due to the common fear that bruxism can cause overloading and may lead to implant failure.
This aspect is, therefore, worth considering in more depth, also in the light of the ways in which the bone-dental implant interface differs from the bone-natural tooth one.
Since the periodontal ligament has a certain amount of give, it allows the tooth a degree of mobility within the tooth socket, quantified as 25-100 μm in the apical-coronal and 56-108 μm in the vestibular-lingual direction (50)
In normally-supported dentition, occlusal overloading due to the introduction of a premature occlusal contact leads to compressive trauma in the area subjected to greatest pressure.
This causes thickening of the periodontal ligament and increased tooth mobility, without loss of attachment in the apical direction.
Whereas in normal conditions this process is completely reversible, in the presence of periodontal disease it can lead to irreversible loss of bone support around the teeth (53).
Compared with natural teeth, implants are fixed much more rigidly to bone tissue, having a maximum mobility of 3-5 μm in the apical-coronal and 10-50 μm in the vestibular-lingual direction; for this reason, unlike the tooth-periodontium interface, they lack the ability to adapt reversibly to different loading conditions (54).
During normal functional loading, bone homeostasis is maintained through a continuous process of bone resorption and apposition of new bone.
Literature descriptions of peri-implant tissue responses to occlusal overloading are not unequivocal, and the most plausible hypothesis so far advanced to explain the possible progressive loss of bone at cervical level in response to excessive overloading is that of fatigue micro-damage resulting in bone resorption (55).
It is suggested that when microtissue damage exceeds the capacity for repair there is an irreversible loss of bone support.
Most of these data derive from studies on animal models of occlusal interference, which also allow us to formulate several considerations relating to bruxism-induced overloading (56).
Animal studies suggest that dynamic overloading, as occurs during tooth-grinding for example, leads to angular bone loss (56).
The eccentric horizontal forces that characterise occlusal loading during tooth-grinding could thus be considered a risk factor for biological implant failure (57).
This affirmation is based on the best evidence currently available, provided by studies of masticatory load distribution in different types of prosthetic implant rehabilitation, which suggest that the maximum forces applied to implants during functional loading can reach bending moments of 90Nmm in the vestibular-lingual direction and even 170Nmm in the opposite direction (58).
Consequently, the forces developed during unconscious activities, like sleep bruxism, and moreover in individuals, such as those with implants, who have reduced proprioception (59), reach levels of intensity so great as to constitute a real risk factor, also – perhaps mainly – because of biomechanical complications (52).
However, these suppositions notwithstanding, the current evidence for a cause-effect relationship between bruxism and implant failure is not high-level evidence.
There exist few prospective studies in which bruxism has been evaluated as a risk factor in implanted patients monitored over long periods of time, and the majority of studies examined in the most reliable literature review currently available were retrospective studies with various sources of bias and confounders (57).
In what is, to date, the longest prospective study to include bruxism among the factors evaluated – it lasted 15 years –, clenching was not found to be related either to loss of bone support or to problems with dental implant superstructures (60).
Instead, other studies, including one by the authors of the previous trial, have produced contrasting results, reporting for example, a greater frequency of biomechanical complications (60% vs 20% at five years) and implant failures (41% vs 12% at one year) in bruxers compared with non-bruxers (61-63).
Therefore, the literature provides conflicting data on the relationship between bruxism and implant failures, a situation that is not improved even by attempts to apply the criteria deemed necessary to demonstrate a cause-effect relationship (Box 8.4).




