Armamentarium
|
History of the Procedure
Reverdin first noted the success of skin grafting in a human patient in 1869, when he removed multiple small pieces of epidermis and autotransplanted them onto a bed of granulation tissue. Due to techniques with “pinch” grafting, Lawson demonstrated success with larger full-thickness grafts in 1870. Ollier thereafter reported improved success with grafts when the donor site was prepared and débrided to healthy fresh tissue. Thiersch evolved the principles of proper débridement and also was the first to demonstrate success using thinner grafts with improved survival and allowing the donor site to heal by reepithelialization without an open wound. Blair and Brown’s paper published in 1929 highlighted the benefits and disadvantages of split-thickness grafts and explained how to achieve reliable success and survival. The current focus is on tissue preparation, innovative dressings to improve and accelerate healing, and tissue engineering techniques that will expand cell populations for vastly reduced donor site size.
History of the Procedure
Reverdin first noted the success of skin grafting in a human patient in 1869, when he removed multiple small pieces of epidermis and autotransplanted them onto a bed of granulation tissue. Due to techniques with “pinch” grafting, Lawson demonstrated success with larger full-thickness grafts in 1870. Ollier thereafter reported improved success with grafts when the donor site was prepared and débrided to healthy fresh tissue. Thiersch evolved the principles of proper débridement and also was the first to demonstrate success using thinner grafts with improved survival and allowing the donor site to heal by reepithelialization without an open wound. Blair and Brown’s paper published in 1929 highlighted the benefits and disadvantages of split-thickness grafts and explained how to achieve reliable success and survival. The current focus is on tissue preparation, innovative dressings to improve and accelerate healing, and tissue engineering techniques that will expand cell populations for vastly reduced donor site size.
Indications for the Use of the Procedure
Within the scope of oral maxillofacial surgery, there are several indications for split-thickness and full-thickness grafting. Grafts are frequently required to repair defects to the head and neck region related to trauma, pathologic/oncologic resection, facial burns, contracture release, and congenital or developmental conditions. In addition, the reconstructive maxillofacial surgeon also utilizes grafts to repair donor site defects following microvascular free flap surgery harvest.
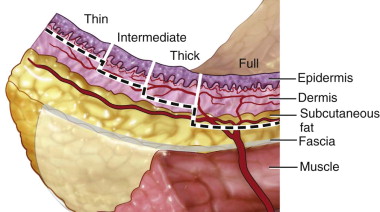
Skin grafts inherently differ based on the amount of dermis harvested. The epidermal layer is quite thin (0.075 to 0.15 mm) compared to the dermis (1 to 4 mm) and is completely harvested in all types of skin grafts. The dermal layer has much more variability in thickness based on location and patient age. Thinner dermis is located on the eyelids; it increases in thickness over the temples, scalp, and is thickest over the back. The dermis thickens as children mature to adulthood and then thins out after the fifth decade.
Full-thickness grafts harvest the entirety of both the epidermis and dermis, thereby requiring the donor site to be closed primarily over top of the underlying subcutaneous tissue to allow for proper healing. Split-thickness grafts split the dermal layer to leave the residual dermis overlying the subcutaneous tissue to heal with secondary reepithelialization. A split-thickness graft can be harvested at a variety of selected thicknesses (0.3 to 0.45 mm/0.012 to 0.018 inch). As thinner grafts are believed to be more reliable for success, often the 0.012-inch setting is used. A handheld purpose-made knife can be used for harvest, but some irregularity and variable thickness can result. Air-driven or electric dermatomes have adjustable thickness controls and interchangeable footplates for the preselected width of the harvest. These dermatomes can allow for uniform harvest at a reliable thickness ( Figure 120-1 ). The donor site often heals within a period of 7 to 21 days, and a variety of dressings are advocated to minimize delays in healing.
Grafting can be considered as a primary treatment strategy or can be delayed if necessary, depending on the wound. When evaluating the recipient site, the surgeon addresses several requirements to optimize graft healing and minimize failures. These include good regional vascularity, the absence of local infection, and a lack of residual tumor. Certain tissue types may afford decreased success rates during healing due to complicating factors. An actively bleeding wound bed will not allow good contact with the graft due to fluid tension and ballooning. Exposed cortical bone, cartilage, and tendons pose difficulties for complete graft take. Maintaining a small layer of periosteum, perichondrium, and paratenon is helpful, but not if it sacrifices marginal clearance during tumor excision. Hypovascular tissues can be problematic due to fibrosis of the adjacent arterial and venous supply caused by prior irradiation or underlying scar tissue from burns or repeated surgeries.
Many wounds demonstrate a bed of granulation tissue, and it is imperative that this tissue is healthy and has minimal bacterial colonization. Sampling tissue for approximate bacterial counts from the recipient bed in delayed procedures can help guide treatment to avoid untoward difficulties with graft healing. It is well accepted that bacterial counts less than 10 5 /g tissue can successfully take a graft, with lower counts affording better results in ideal situations. Local and systemic antibiotics and dressing management can help surgeons to disinfect and decontaminate the wound. In an open wound, frequent dressing changes (two to three times a day) may be necessary to optimize healing and decrease contamination. In addition, the graft must be immobilized during healing to ensure optimal contact between the graft and the recipient site while minimizing shear forces. Immobilization can include the use of bolsters or figure-eight quilting sutures. Although it is ideal to replace tissue defects with identical tissue, skin grafts can be used in multiple applications and offer significant benefits compared to local or regional flaps within the head and neck region.
During the first 24 hours of healing, there is plasma, or more appropriately serum imbibition, which allows nutrients to transfer from the recipient bed to the graft. In addition, fibrinogen release by the capillary system allows for anchorage of the graft. This matures to form vascular inosculation channels primarily at the base and periphery with the adjoining subdermal plexus. Vascular flow is reestablished thereafter, often starting by postoperative day 3 or 4 ( Table 120-1 ).
| Split-Thickness Grafts | Full-Thickness Grafts |
|---|---|
|
|
|
|
|
|
|
|
Split-Thickness Donor Sites
Despite the inherent capacity for the donor site to heal, it is subject to discoloration (hypo- or hyperpigmentation as well as erythema), irregularity, and scarring. Therefore, it is preferable to locate donor sites in inconspicuous areas that clothing can easily hide. Considerations in choosing a site include ease of access during surgery, ease of managing wounds, and postoperative discomfort. Many areas on the proximal extremities and trunk are used, including the upper anterior thigh, lateral thigh, lower abdomen, buttocks, and infrequently the flank of the back. Many other sites are referenced in the literature but are used infrequently for maxillofacial surgical procedures.
Split-Thickness Recipient Site Applications
Although this is by no means a comprehensive listing of all sites, it affords several examples of applications given a multitude of clinical scenarios.
Soft Tissue Defect
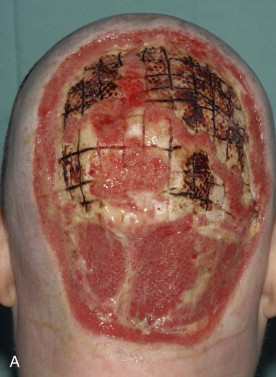
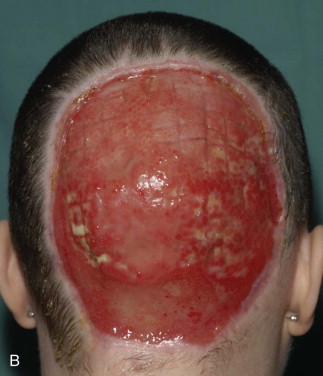
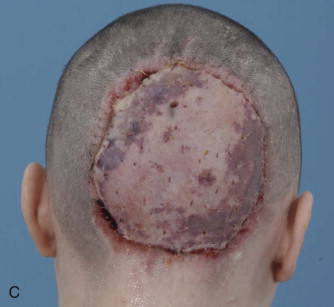
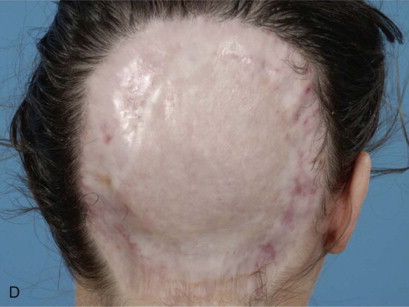
Large Open Wounds to the Scalp and External Cheek.
Despite the unappealing properties of poor color match and contracture, split-thickness grafts are frequently used to obtain initial healing of the defect. They may also be applied in medically compromised patients to decrease the morbidity associated with larger pedicled flap procedures or microvascular free tissue transfer ( Figure 120-2 ).
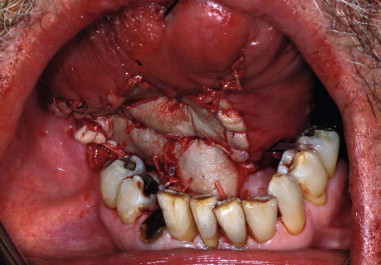
Buccal Mucosa, Partial Tongue, Limited Floor of Mouth Reconstruction.
Due to the lack of esthetic concerns intraorally, split-thickness grafts are often used in an attempt to resurface defects. Contracture is expected, and therefore grafting is undertaken for smaller defects of the aforementioned sites. For larger defects, flaps would be ideally recommended to improve the function and bulk of the resected areas ( Figure 120-3 ).
Exposed Muscle from a Pedicled or Microvascular Flap in the Head and Neck Region.
Pedicled flaps or even microvascular tissue transfer can create areas of exposed muscle that cannot be closed over, primarily due to soft tissue swelling and tension or concerns over pedicle blood supply. Some grafts may be considered temporary measures to be excised at a later date if necessary.
Covering Exposed Paratenon or Muscle in Microvascular-Free Flap Harvest (Common Areas Include Radial Forearm and Fibula Donor Sites).
Donor tissue sites may frequently create a defect in areas that possess minimal esthetic needs. Controversy continues to exist over the use of full-thickness versus split-thickness grafts to cover these defects. Many accept that split-thickness grafts offer a greater chance of graft success, especially when placed over paratenon (radial forearm defect) due to decreased vascularity.
Oral Rehabilitation
Maxillectomy with Use of Obturator.
Several authors believe that a split-thickness skin graft within the buccal or labial defect of a maxillectomy can be used to provide a scar band that can stabilize an obturator. The contraction that occurs with the healing of a skin graft is used advantageously by the maxillofacial prosthodontist for additional retention.
Oral Rehabilitation
Vestibuloplasty with Floor of Mouth Lowering.
Preprosthetic site preparation has been utilized to improve tissue attachment and subsequent denture retention. With the increasing use of microvascular free tissue transfer for managing ablative defects, there is an increased need for flap revision, debulking, and skin grafting. Minimizing the soft tissue thickness allows for more successful oral rehabilitation with dental implants.
Full-Thickness Donor Sites
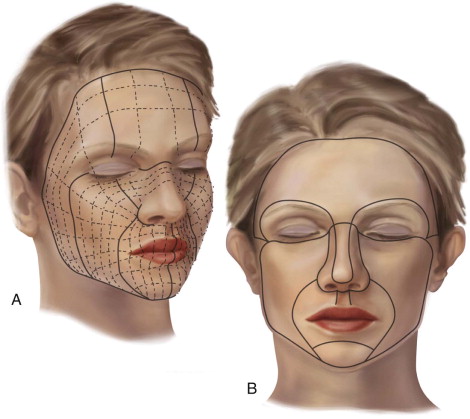
To satisfy cosmetic requirements of the face, full-thickness grafts can offer optimized color match, consistency, and texture. A multitude of harvest sites can allow for inconspicuous residual scars, including the upper lid, preauricular, postauricular, nasolabial, supraclavicular, and upper neck ( Figure 120-4 ). When esthetic demands are not paramount, any body site can be utilized, including the abdomen, thigh, upper arm, and even skin from an avulsive injury or from resection of soft tissue flaps. As these sites require primary closure, elliptical incisions are used. The elliptical incisions parallel the relaxed or resting skin tension lines of the face or neck and are oriented perpendicular to the lines of maximum extensibility. Often, excess tissue is harvested to allow for tension-free closure while avoiding the creation of standing cutaneous (cone) deformities (“dog ears”) at the edges of the harvest site. The pinch test is an easy tool that can be used to ensure optimal harvest with minimal tension closure.
Full-Thickness Recipient Site Applications
Given the improved success rates of healing with split-thickness skin grafts, the full-thickness skin graft is targeted primarily to higher esthetic needs of the head and neck. A full-thickness graft can replace a shallow defect with like tissue that allows minimal to no discernible depression to the surgical defect area. Use of a bolster dressing is imperative to adapt the graft to the defect and avoid untoward shear stresses. After placement and healing of the graft, subtle irregularities in color and texture can often be managed with selective dermabrasion.
Soft Tissue Defect
Small Defects in High Esthetic Areas.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


