Bone replacement grafts are widely used to promote bone formation and periodontal regeneration. Conventional surgical approaches, such as open flap debridement, provide critical access to evaluate and detoxify root surfaces as well as establish improved periodontal form and architecture; however, these surgical techniques offer only limited potential in restoring or reconstituting component periodontal tissues. A wide range of bone grafting materials, including bone grafts and bone graft substitutes, have been applied and evaluated clinically, including autografts, allografts, xenografts, and alloplasts (synthetic/semisynthetic materials). This review provides an overview of the biologic function and clinical application of bone replacement grafts for periodontal regeneration. Emphasis is placed on the clinical and biologic goals of periodontal regeneration as well as evidence-based treatment outcomes.
Bone replacement grafts are widely used to promote bone formation and periodontal regeneration. Conventional surgical approaches, such as open flap debridement, provide critical access to evaluate and detoxify root surfaces as well as establish improved periodontal form and architecture; however, these surgical techniques offer only limited potential in restoring or reconstituting component periodontal tissues. Bone grafting materials function, in part, as structural scaffolds and matrices for attachment and proliferation of anchorage-dependent osteoblasts ( Fig. 1 ). A wide range of bone grafting materials, including bone grafts and bone graft substitutes, have been applied and evaluated clinically, including autografts, allografts, xenografts, and alloplasts (synthetic/semisynthetic materials). Although not all bone grafting materials support the formation of a new periodontal attachment apparatus, there is conclusive evidence that periodontal regeneration is achievable with bone replacement grafts in humans.

The purpose of this review is to provide an overview of the biologic function and clinical application of bone replacement grafts for periodontal regeneration. Emphasis is placed on the clinical and biologic goals of periodontal regeneration as well as evidence-based treatment outcomes.
Periodontal regeneration: clinical and biologic goals
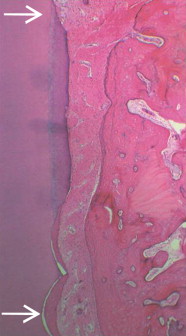
The biologic goal of periodontal regeneration is restoration of the periodontium to its original form and function. Periodontal repair is healing of the periodontium by tissue that does not fully restore the original functional anatomic and morphologic architecture. Following conventional surgical approaches, such as open flap debridement, repair of lost periodontal attachment occurs primarily through formation of a long junctional epithelium between the gingival flap and the previously diseased root surface. Epithelium is an efficient tissue for rapid wound repair, in part due to the comparatively high proliferation rate and effective adhesive mechanisms of epithelial cells. Regenerative therapy, in contrast, is designed to support regeneration of the attachment apparatus, namely the formation of bone, cementum, and periodontal ligament ( Fig. 2 ). Clinical outcome parameters consistent with successful regenerative therapy include reduced probing depth, increased clinical attachment level, and radiographic evidence of bone fill. At present, conclusive evidence of periodontal regeneration can be provided only through histologic evaluation of treated intraosseous defects using unambiguous reference points along the root surface (see Fig. 2 ).
Bone replacement grafts
Bone replacement grafts (bone grafts and bone graft substitutes) provide a structural framework for clot development, maturation, and remodeling that supports bone formation in osseous defects. Bone grafting materials also exhibit a variable capacity to promote the coordinated formation of bone, cementum, and periodontal ligament when placed and retained in periodontal defects. Bone grafting materials must possess the attributes of biocompatibility (lacking an immunogenic response) and osteoconductivity (providing a structure and surface topography that permit cellular attachment, proliferation, and migration). Bone replacement grafts may also possess other properties that support osteogenesis. Osteogenic grafting materials, such as cancellous bone/bone marrow, contain living cells that are capable of differentiation and formation of bone. Osteoinductive grafting materials, such as demineralized bone matrix (DBM), provide a biologic stimulus (proteins and growth factors) that induces the progression of mesenchymal stem cells and other osteoprogenitor cells toward the osteoblast lineage.
The Food and Drug Administration (FDA) regulates bone grafting materials that are intended to fill, augment, or reconstruct periodontal or bony defects of the oral and maxillofacial region. The regulatory control of the FDA includes human cells, tissues, and cellular-based products, under statutes published in Code of Federal Regulations (Federal Register, Title 21) that are intended for transplantation. Multiple classification systems have been used to organize bone replacement grafts, which commonly include source (eg, allograft), chemical composition (eg, calcium phosphate), and physical properties (eg, ceramic). Advances in material sciences, however, have increasingly blurred such boundaries between types of bone replacement grafts. For the purposes of this review, bone replacement grafts have been organized primarily according to source.
Autografts
The “gold standard” for bone grafting procedures historically has been particulate cancellous bone/bone marrow autografts, which can provide a rich source of bone and marrow cells that have osteogenic potential. Histologic findings from case reports substantiate the potential for autogenous bone/bone marrow grafts to support periodontal regeneration in humans. Multiple clinical considerations have limited the use of extraoral autografts, particularly from iliac crest, including the possibility of surgical complications and pain associated with donor site.
Autogenous bone is frequently harvested from intraoral sites, often from the same quadrant as the regenerative surgery. Intraoral donor sites, however, typically yield comparatively limited graft volume. Harvesting sufficient donor bone, therefore, as an osseous coagulum of cortical or cortical-cancellous bone, can necessitate the creation of additional intraoral surgical sites, thereby increasing the potential for surgical morbidity and discomfort.
Allografts
Bone allograft is the most frequently used alternative to autogenous bone for bone grafting procedures in the United States. Under FDA regulations, facilities engaged in procuring and processing human tissues for transplantation must ensure that specified minimum medical screening and infectious disease testing have been performed, and that records exist and are maintained to document screening and testing for each human tissue. The American Association of Tissue Banks also sets standards, inspects facilities, and accredits tissue banks in North America. There are no reports of disease transmission during the 30-year history of use of freeze-dried bone allografts in periodontal therapy.
Tissue banks process bone allografts using a variety of methods, some based on proprietary techniques, but most are based on similar underlying concepts that address cleansing, decontamination, antimicrobial treatment, dehydration, graft size, and terminal sterilization. Particulate bone allografts are provided as mineralized or demineralized products. Allogeneic bone that has undergone extensive demineralization is referred to as DBM or demineralized freeze-dried bone allograft (DFDBA). DBM exhibits the capacity to induce bone formation in nonorthotopic sites, such as muscle, and is considered to be osteoinductive. Bone demineralized to levels of approximately 2% residual calcium has been shown to provide maximum osteoinductive potential by assay systems, presumably due to exposure of bone morphogenetic proteins. Bowers and colleagues have provided conclusive histologic evidence that DBM supports periodontal regeneration in humans.
Xenografts
Xenografts are surgical grafts transplanted between different species. Two sources of xenografts are commercially marketed as particulate bone replacement grafts in clinical practice: bovine bone and natural coral. Anorganic bovine bone graft (ABM) is a naturally derived porous and deproteinized bovine bone mineral with comparable mineral composition and microporous structure to native humane bone. ABM has been shown to support significant gains in clinical attachment level and hard tissue fill in human intrabony defects. Available human histologic evidence provides proof-of-principle evidence that ABM and ABM/collagen can support periodontal regeneration in intrabony defects.
The calcium carbonate exoskeleton of coral species, such as Porites, can be converted to hydroxyapatite by hydrothermal exchange. Calcium carbonate-hydroxyapatite constructs can be produced by limited hydrothermal conversion. The porosity and pore size distribution of hydroxyapatite, which is dependent primarily on coral species, provides a osteoconductive scaffold that enhances bone formation and undergoes dissolution and resorption with bone remodeling. Ripamonti and colleagues have shown that coral-derived biomimetic matrices of different chemical compositions have the capacity to induce heterotopic bone formation in the nonhuman primate Papio ursinus .
Alloplasts
An alloplast is a biocompatible, inorganic synthetic bone grafting material. At present, alloplasts marketed for periodontal regeneration fall into 2 broad classes: ceramics and polymers. The composition, morphology, and surface topography of alloplasts provide the osteoconductive platform for promoting bone formation along the surface of the grafting material. The fate of an alloplastic bone grafting material is dependent primarily on its chemical composition, structure, and physical properties.
Ceramic-based bone grafting materials have been widely used for bone and periodontal regeneration, and function primarily through osteoconduction. Commercially available ceramic-based materials include calcium phosphates (eg, tricalcium phosphate and hydroxyapatite), calcium sulfate, and bioactive glass. These ceramics have also been considered osteointegrative, because of the tenacious, intimate bond formed between the new mineralized tissue and graft material. Ceramics can exist in fully amorphous or crystalline phases, with the same ratios of elemental substances (calcium and phosphorus). Differences in the degree of crystalline arrangement significantly impact the physical and biologic characteristics of the ceramic, including strength, modulus, and dissolution rate.
Synthetic and coral-derived porous hydroxyapatite has been shown to support significant clinical improvements in periodontal measures following implantation in intrabony defects. The porosity and degree of sintering of synthetic hydroxyapatite ceramics primarily determines the rate of biodegradation—crystalline, nonporous hydroxyapatite is essentially nondegradable. Human histologic evidence of ossification of the graft pores and the graft periphery of porous hydroxyapatite has been found in periodontal intrabony sites, with residual graft particles present 12 months following implantation. Porous hydroxyapatite appears to exhibit osteoconductive properties, as reflected clinically in bone formation; however, no evidence of periodontal regeneration has been shown.
β-Tricalcium phosphate (β-TCP) is a porous form of calcium phosphate, with similar proportions of calcium and phosphate to cancellous bone. As a bone grafting material, β-TCP supports improvements in clinical outcomes, including clinical attachment level, but histologic evidence reveals periodontal repair, primarily through the formation of a long junctional epithelial attachment, with limited new connective tissue attachment. Although the surface layer of β-TCP allows for bone deposition in orthopedic sites, including maxillary sinus augmentation, this ceramic has been shown to become fibroencapsulated when placed in periodontal intrabony defects, failing to stimulate new bone growth, with residual graft particles evident 18 months following treatment. Differences in the stoichiometric chemistry and structure of β-TCP may affect the rate of resorption and replacement with bone during healing.
Bioactive glass is a ceramic composed principally of SiO 2 . The original composition of bioactive glass approved by the FDA, designated 45S5, was composed of 46.1 mol% SiO 2 , 26.9 mol% CaO, 24.4 mol% Na 2 O, and 2.5 mol% P 2 O 5 . The original composition and fine structure has been extensively modified in an attempt to further enhance bioactive glass as a bone replacement graft. Bioglass can bond directly to bone through the development of a surface layer of carbonated hydroxyapatite in situ. This calcium phosphate-rich layer is thought to promote adsorption and concentration of osteoblast-derived proteins necessary for the mineralization of extracellular matrix. Bioactive glass has been shown to significantly increase clinical attachment level and hard tissue fill when implanted in intrabony defects. Although bioactive glass exhibits osteconductive properties as a grafting material in maxillary sinus and extraction socket, histologic analysis of human periodontal defects has revealed healing primarily by connective tissue encapsulation of the graft material and epithelial down-growth, with minimal evidence of new cementum or connective tissue attachment limited to the most apical part of the defect.
Polymers can be classified based on source: natural and synthetic. Natural polymers that have been used in the fabrication of bone grafting materials include polysaccharides (eg, agarose, alginate, hyaluronic acid, chitosan) and polypeptides (eg, collagen, gelatin); however, the structural properties of natural polymers, including a comparatively weak mechanical strength and variable rates of degradation, have limited their use as standalone bone grafting materials. Natural polymers may serve an important role in composite grafts, which incorporate a particulate or biologic, such as Bio-Oss Collagen (Geistlich Pharma AG, Wolhusen, Switzlerland).
Synthetic polymers (eg, poly(glycolic acid), poly( l -lactic acid), polyorthoester, polyanhydride) provide a platform for controlling the biomechanical properties of scaffolds as well as targeting drug delivery in tissue engineering. Polymers are more widely used as barrier materials in guided tissue regeneration (GTR) procedures for the treatment of periodontal defects. At present, several polymer systems are being used for bone and periodontal regeneration, including polylactic acid (PLA)-based polymers and copolymers. These polymers have proved to be effective in periodontal applications as barrier materials but can elicit inflammatory and foreign body reactions, especially following fragmentation secondary to bulk degradation. Synthetic polymers have an expanded use in orthopedic applications as injectable and solid resin-based products.
At present, a biocompatible microporous polymer containing polymethylmethacrylate (PMMA), polyhydroxylethylmethacrylate (PHEMA), and calcium hydroxide is available as a bone grafting material for the treatment of periodontal defects. This composite is prepared from a core of PMMA and PHEMA with a coating of calcium hydroxide. The polymer is hydrophilic and osteophilic, which purportedly aids in stabilization of the healing clot. A controlled study and case series reports provide evidence for the effectiveness of this polymeric grafting material in improving clinical parameters, relative to open flap debridement, in the treatment of periodontal intraosseous defects. Histologic evaluations revealed that the polymer was associated with minimal inflammation and infrequent foreign body giant cells, with evidence of both bone apposition and soft tissue encapsulation, at 1 to 30 months following implantation.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


