Introduction
The aim of this study was to evaluate the cytotoxicity of 3 orthodontic acrylic materials and 2 manipulation methods.
Methods
The orthodontic acrylic materials Orthocryl EQ (Dentaurum, Ispringen, Germany), Orthoplast (Vertex Dental, Zeist, The Netherlands), and O-80 (Imicryl, Konya, Turkey) were prepared with 2 polymerization methods (doughing and spray on). Totally, 60 cylinders (5 × 2 mm), fabricated by using a different acrylic and method, were divided into 6 groups. Gingival fibroblasts were isolated from gingival connective tissue of systemically healthy subjects. Materials were incubated in Dulbecco’s modified eagle’s medium culture medium (Biological Industries, Beit Haemek, Israel) for 72 hours according to ISO 10993-5 standards (surface area to volume ratio of the specimen to cell-culture medium: 3 cm 2 /mL). Gingival fibroblasts were maintained with Dulbecco’s modified eagle medium containing 10% fetal bovine serum. A real-time cell analyzer (RT-CES, xCELLigence; Roche Applied Science, Mannheim, Germany, and ACEA Biosciences, San Diego, Calif) was used to evaluate cell survival. After seeding 200 μL of the cell suspensions into the wells (20,000 cells/well) of the E-plate 96, gingival fibroblasts were treated with bioactive components released by the acrylic materials (1/1 and 1/2 dilutions) and monitored every 15 minutes for 121 hours. For the proliferation experiments, the statistical analyses used were 1-way analysis of variance (ANOVA) and Tukey-Kramer multiple comparisons tests.
Results
There was no significant difference between the cell indexes of the control and study groups for the 1/1 and 1/2 dilutions at 21 and 32 hours. When evaluated at 68 hours, all 1/2 dilutions of acrylic materials showed statistically insignificant differences ( P >0.05) except for Orthoplast ( P <0.05). But all acrylic materials were different from the control group in the 1/1 dilutions ( P <0.001). At 121 hours, all test groups were significantly different from the untreated control group ( P <0.001).
Conclusions
The results indicate that the long cycle increased the cytotoxicity of the tested materials, and there was no significant difference between the spray-on and doughing methods on cytotoxicity.
Acrylic resins, or polymethylmethacrylates (PMMAs), are commonly used primarily for dentures and orthodontic appliances. In addition, PMMAs are used for individual impression trays and early orthopedic appliances for patients with cleft lip and palate and as splints for orthognathic surgery.
Although acrylic resins can be classified as thermo-polymerized, auto- polymerized, microwave-polymerized, and photo-polymerized, auto-polymerized resins are the most popular materials in orthodontics because of their low cost and ease of use. Despite its advantages, PMMA can cause irritation, inflammation, and allergic reactions in the oral mucosa. Toxic compounds such as methylmethacrylate and methacrylic acid in the chemical composition of the acrylic resin can cause hypersensitization and allergy in orthodontic patients and the dental team. Furthermore, acrylic materials have been found to be potentially toxic, carcinogenic, mutagenic, and estrogenic.
Methyl esters of methacrylic acid are the basic modules of PMMA, but there are many other components in acrylics used for prosthetic dentistry and orthodontics. Heat-polymerizing denture acrylics are generally based on PMMA, whereas light-polymerizing and microwave-polymerizing products are derived partly from PMMA and also from urethane dimethacrylates. The polymerization of chemically curing or auto-polymerizing acrylics (generally orthodontic acrylic) that set at room or oral temperature is triggered by a redox system.
Currently, PMMA resins are widely used in orthodontics for removable and auxiliary fixed appliances as well as Hawley retainers. There are different methods of manipulation to produce these appliances. For manipulation, generally 2 methods are described. The spray-on method, in which the polymer is saturated by its monomer, is widely used in orthodontics, and the doughing method, also a cold-cured technique, in which powder and liquid are mixed together, is commonly used in prosthodontic dentistry.
Although most acrylic materials used consist of PMMA, many authors have discussed the polymerization process involved in converting monomer to polymer, because adequate polymerization is a crucial factor in maximizing the physical properties and biocompatibility of acrylic materials resins. Some orthodontic appliances are kept in contact with the oral mucosa for a long period, so the biologic effects of methylmethacrylate (MMA) must be considered. MMA is considered an allergen and can cause local adverse reactions, such as erythema, burning sensations, edema, fissures, necrosis, pain, and even some systemic reactions.
Furthermore, the developments and improvements of the orthodontic acrylic materials are satisfying and incredible, but the biocompatibility of these acrylics is still a problem for orthodontists. In addition, few data are available in the literature regarding the biocompatibility of commercially available orthodontic acrylics and the length of time after polymerization. Orthodontists are using many acrylic materials, and newer orthodontic acrylic materials present new challenges because of the potential for interactions.
A number of published studies have investigated the biocompatibility of acrylic denture base materials. The majority of these studies concluded that acrylic materials are cytotoxic.
No comprehensive data are available in the orthodontic literature regarding the toxicity of orthodontic acrylic materials. The evaluation procedures for biologic investigation, especially of the acute local toxicity of medical and dental materials, are regulated by ISO standard 10993-5. The manufacturers of these materials possess comprehensive test data. Therefore, the aim of this study was to evaluate the cytotoxic effects of 3 orthodontic acrylic materials with 2 manipulation methods.
Material and methods
The orthodontic acrylic materials selected were Orthocryl EQ (Dentaurum, Ispringen, Germany), Orthoplast (Vertex Dental, Zeist, The Netherlands), and O-80 (Imicryl, Konya, Turkey). Details related to these products and their components are listed in Table I .
| Brand | Components | Chemical composition | Manufacturer | Setup proportion for doughing | Time |
|---|---|---|---|---|---|
| Orthoplast | Liquid | Methylmethacrylate, crosslinker, accelerator | Vertex | Liquid:1 mL | Dough: 9 min |
| Powder | Methyl methacrylate | Powder: 2.1 g | Polymerization: 20 min | ||
| Orthocyl EQ | Liquid | Methylmethacrylate,ethylene glycol dimethacrylate, N,N-dihydroxyethylene-p-poluidine | Dentaurum | Liquid:1 scale | Dough: 3-5 min |
| Powder | Polymethylmethacrylate | Powder: 2.5 scale | Polymerization:15 min | ||
| O-80 | Liquid | Methylmethacrylate | Imicryl | Liquid:1 scale | Dough: 9 min |
| Powder | Methacrylate copolymers | Powder: 2.5 scale | Polymerization: 20 min |
Test specimens were prepared according to the manufacturers’ instructions in standard discs made of polytetrafluoroethylene, measuring 5 mm in diameter and 2 mm in thickness. All specimens were prepared and handled under aseptic conditions to limit any biologic contamination of the cell culture tests. The specimens were prepared between polyester film and glass slabs to minimize the oxygen inhibition and maximize the surface smoothness.
The 3 orthodontic acrylic materials (Orthocryl EQ, Orthoplast, and O-80) were prepared with 2 manipulation methods (doughing and spray on). Totally, 60 cylinders (5 × 2 mm) fabricated with different acrylics and methods were divided into 6 groups. Ten samples were prepared for each group for cytotoxicity testing. The samples were immersed in 7 mL of culture medium for 24 hours at 37°C to extract residual monomer or cytotoxic substances. The culture medium containing the material extracts was sterile and filtered for use on the cell cultures. The test procedures for this investigation were regulated by ISO standard 10993-5.
The xCELLigence system (Roche Applied Science, Mannheim, Germany, and ACEA Biosciences, San Deigo, Calif) consists of 4 main components: the impedance-based real-time cell analyzer (RTCA), the RTCA single plate station, the RTCA computer with integrated software, and disposable E-plate 96. The RTCA single plate station fits inside a standard tissue-culture incubator. The electronic impedance of the sensor electrodes was measured to allow monitoring and detection of physiologic changes of the cells on the electrodes. The voltage applied to the electrodes during the RTCA measurement was about 20 mV (root mean square). The impedance measured between electrodes in each well depends on electrode geometry, ion concentration in the well, and whether the cells are attached to the electrodes. In the absence of cells, electrode impedance is mainly determined by the ion environment both at the electrode-solution interface and in the bulk solution. In the presence of cells, cells attached to the electrode sensor surfaces act as insulators and thereby alter the local ion environment at the electrode-solution interface, leading to increased impedance. Thus, the more cells that are growing on the electrodes, the larger the value of the electrode impedance. The data expressed in cell index units can be exported to Excel software (Microsoft, Seattle, Wash) for any type of mathematical analysis.
For the cell cultures, gingival fibroblasts were isolated from gingival connective tissues of systemically healthy subjects. The gingival fibroblasts were obtained from samples of gingiva removed for crown lengthening purposes. All patients gave informed consent before providing the samples. This protocol was approved by the ethics committee of the Faculty of Dentistry at Selcuk University in Turkey. Briefly, the gingival tissues were cut into small pieces, rinsed with the biopsy medium, placed in tissue culture dishes, and incubated in the biopsy medium in a humidified atmosphere of 95% air and 5% carbon dioxide at 37°C overnight. The following day, the biopsy medium was replaced with the culture medium (Dulbecco’s modified eagle medium [Biological Industries, Beit Haemek, Israel] with 10% fetal bovine serum, 100 units/mL penicillin, 100 μg/mL streptomycin). After reaching confluency, the cells were passaged with 0.25% trypsin and 0.1% ethylene diaminotetraacetic acid. Gingival fibroblasts were used between the fourth and sixth passages for all experiments.
In the preparation of the materials, 5 × 2-mm cylinders of each material were fabricated and set for 2 days at 37°C. The materials were incubated in DMEM culture medium for 72 hours according to ISO 10993-5 standards (surface area-to-volume ratio of the specimen to cell-culture medium: 3 cm 2 /mL). Gingival fibroblasts were maintained with DMEM containing 10% fetal bovine serum. The xCELLigence system was used to evaluate cell survival, according to the instructions of the supplier. After seeding 200 μL of the cell suspensions into the wells (20,000 cells/well) of the E-plate 96, gingival fibroblasts were treated with bioactive components released by the acrylic materials (1/1 and 1/2 dilutions) and monitored every 15 minutes for 121 hours.
The morphology of the gingival fibroblasts was evaluated after treatment with bioactive components of the acrylic materials by using an inverted microscope.
Statistical analysis
All calculations were obtained by using the RTCA-integrated software of the xCELLigence system. The RTCA software performs a curve-fitting of selected “sigmoidal dose-response equation” to the experimental data points. Data are represented as mean (mmol/L) ± SEM (n = 5).
For proliferation experiments and gene expressions, the statistical analyses used were 1-way analysis of variance (ANOVA) and Tukey-Kramer multiple comparison tests. The data are represented as means and standard deviations. A value of P <0.05 was considered to be statistically significant.
Results
First, we determined the optimal concentrations for cell proliferation and viability measurements. To this end 40,000, 20,000, 10,000, and 5000 cells per well were seeded in the E-plate 96, and the impedance was determined ( Fig ).
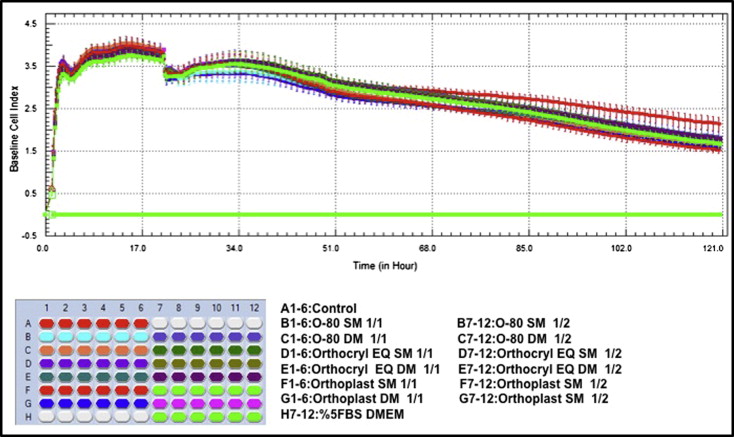
According to ANOVA, when left to elute for 21 and 32 hours, there were no significant differences between the cell indexes of the control and study groups for the 1/1 and 1/2 dilutions. ( Table II and Fig ).
| Group (n = 6) | Concentration | 21 hours mean ± SD |
32 hours mean ± SD |
68 hours mean ± SD |
121 hours mean ± SD |
|---|---|---|---|---|---|
| Control | 3.60 ± 0.09 | 3.50 ± 0.13 | 2.93 ± 0.09 | 2.34 ± 0.16 | |
| O-80 SM | 1/1 | 3.65 ± 0.22 | 3.35 ± 0.25 | 2.60 ± 0.09 † | 1.88 ± 0.09 † |
| O-80 SM | 1/2 | 3.75 ± 0.15 | 3.58 ± 0.28 | 2.86 ± 0.07 | 2.04 ± 0.12 † |
| O-80 DM | 1/1 | 3.70 ± 0.06 | 3.60 ± 0.14 | 2.62 ± 0.07 † | 1.84 ± 0.11 † |
| O-80 DM | 1/2 | 3.72 ± 0.20 | 3.59 ± 0.24 | 2.84 ± 0.11 | 2.00 ± 0.10 † |
| Orthocryl EQ SM | 1/1 | 3.67 ± 0.24 | 3.50 ± 0.28 | 2.60 ± 0.10 † | 1.93 ± 0.07 † |
| Orthocryl EQ SM | 1/2 | 3.69 ± 0.15 | 3.58 ± 0.24 | 2.89 ± 0.10 | 2.02 ± 0.05 † |
| Orthocryl EQ DM | 1/1 | 3.69 ± 0.18 | 3.52 ± 0.17 | 2.57 ± 0.03 † | 1.93 ± 0.08 † |
| Orthocryl EQ DM | 1/2 | 3.70 ± 0.08 | 3.61 ± 0.10 | 2.79 ± 0.06 | 2.01 ± 0.04 † |
| Orthoplast SM | 1/1 | 3.81 ± 0.20 | 3.56 ± 0.24 | 2.59 ± 0.08 † | 1.72 ± 0.07 † |
| Orthoplast SM | 1/2 | 3.74 ± 0.05 | 3.54 ± 0.16 | 2.76 ± 0.07 ∗ | 1.90 ± 0.09 † |
| Orthoplast DM | 1/1 | 3.85 ± 0.05 | 3.33 ± 0.16 | 2.57 ± 0.07 † | 1.80 ± 0.06 † |
| Orthoplast DM | 1/2 | 3.89 ± 0.04 | 3.62 ± 0.12 | 2.81 ± 0.08 | 1.95 ± 0.09 † |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


