http://evolve.elsevier.com/hatrick/materials.
Adhesive materials are used on a daily basis in the modern dental practice. Adhesive materials are beneficial for many restorative and preventive procedures. They provide a seal between the tooth and the restoration and seal the dentinal tubules to reduce postoperative sensitivity. They enhance retention of the restoration so that more conservative preparations can be made, and they may increase the strength of the prepared tooth to resist fracture. Materials bonded to tooth structure may themselves be more resistant to fracture than materials that are not bonded. Adhesives are used for a wide variety of dental procedures. The dental assistant and the dental hygienist must be familiar with the terms and processes used in bonding of various restorative and preventive materials, to be knowledgeable, effective members of the dental team. The dental assistant will be involved in helping the dentist perform bonding procedures many times each day. In some states trained hygienists and assistants may etch tooth structure and apply bonding agents and place sealants and composite resins. The dental hygienist may perform periodontal treatments that might affect bonded restorations. Therefore, it is important that the allied oral health practitioner understand the properties and handling characteristics of the bonding materials and the processes involved in their use.
Basic Principles of Bonding
In dentistry, the term bond, or bonding, is used to describe the process of attaching restorative materials, such as a composite resin, to a tooth by adhesion (the attraction of atoms or molecules of two different contacting surfaces). When describing cosmetic restorations such as porcelain or composite veneers, patients often use the term “bonding,” for example, “The dentist is bonding my front teeth.” Bonding also is the basis for several other dental procedures, such as the placement of orthodontic brackets and fixed retainers. It is used to describe some of the materials used in the process of placing restorations. For example, bonding resin is placed on the etched tooth surface before light-curing. Manufacturers often use the word “bond” in the trade names of their bonding resins, such as Prime & Bond NT (Dentsply International) and OptiBond Solo Plus (Kerr Corporation).
Preparation for Bonding
The first step in the bonding process involves preparation of the surface of the tooth or the restoration (or both) to receive the material that will be bonded to it. Preparing the tooth surface usually includes removing plaque and debris, and then etching or conditioning the enamel or dentin (or both) with an acid. The most commonly used acid is phosphoric acid in concentrations ranging from 10% to 38%. Acid removes mineral from the surface to create roughness or microscopic porosity.
Bonding to the Etched Surface
When a resin bonding agent or primer is flowed over the etched surface, it penetrates into the microscopic pores. When it hardens (cures or polymerizes), it creates projections called resin tags that lock into the tooth, creating a mechanical bond called micromechanical retention. The resin bonding agent will then chemically bond to other resins placed over it, such as composite resin. The chemical bond, called a primary bond, is a true adhesion between atoms or molecules of the composite resin and the bonding resin. The chemical bond is stronger than a physical bond, called a secondary bond, which is a weak physical attraction between two surfaces such as the adhesion of paint to a metal surface. Roughening the metal surface by sandblasting increases the adhesion of the paint by mechanical retention, much in the way that acid etching roughens the surface of the enamel to achieve mechanical retention.
Surface Wetting
Acid etching also increases the ability of liquids to wet the surface of the tooth by creating high surface energy. High surface energy can attract contaminants (such as saliva), so good isolation is important. If saliva contamination occurs, drying the surface will leave residues that will interfere with the bond. The surface must be re-etched. Good wetting increases the intimate contact of the bonding resin with the etched tooth structure, improving the penetration of resin to form tags and thereby improving the bond. High surface energy helps to attract the resin to the etched surface. Surfaces that are poorly wetted will cause beading of the liquid, similar to water on a newly waxed car. Each bead of water stands up on the surface of the car with a high angle of contact. On an unwaxed car, the water easily spreads out and has a low angle of contact (Figure 5-1). Bonding agents are usually not very viscous (thick), so they will flow readily and wet the etched surface.
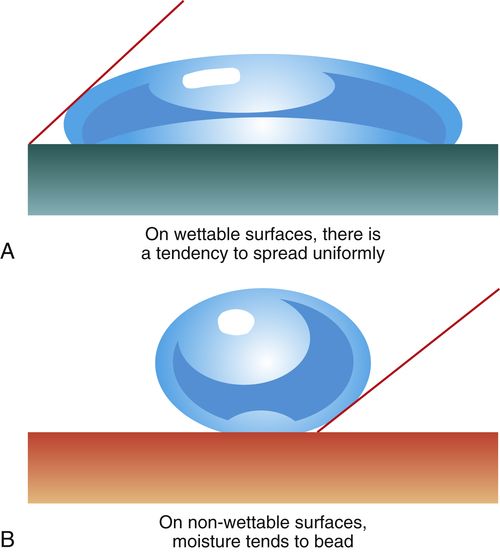
FIGURE 5-1 Acid etching of enamel increases its ability to be wetted by a resin bonding agent, resulting in a stronger bond.
A, A low contact angle indicates good wetting as the liquid spreads over the surface. B, A high contact angle indicates poor wetting as the liquid beads on the surface like water on a waxed car.
Bond Strength
The strength of the bond obtained is usually measured by determining the force needed to separate the two joined materials. The force needed to break the bond is divided by the cross-sectional area of the bonded surfaces to arrive at the value for the bond strength. Most bond tests pull the bonded materials apart (tensile bond strength) or apply forces at approximately 90 degrees to the bonded interface of the materials until the bond fails (shear bond strength). The value for the bond strength is reported as megapascals (MPa). One megapascal equals 150 pounds per square inch (psi). Choosing materials with good bond strengths to tooth structure can enhance the longevity of the restoration and potentially allow for more conservative preparations, because cutting into healthy tooth structure to create mechanical locks can be minimized.
Bonding to enamel usually achieves consistently high bond strengths of around 30 MPa (4500 psi). The bond strength to dentin is usually less than to enamel and varies according to how deep into the dentin the cavity preparation extends. The dentin near the dentinoenamel junction (DEJ) has fewer dentinal tubules (about 15,000 to 20,000/mm2), occupying 14% of the dentin surface, and they are smaller in diameter than in the dentin closer to the pulp. Deeper dentin contains more tubules (about 45,000 tubules/mm2) and they are larger in diameter, occupying 20% to 30% of the dentin surface. Fluid flows from the pulp into the tubules on a constant basis. Therefore, deeper dentin will be wetter dentin from the flow of pulpal fluid through the tubules. Wetter dentin with more holes (tubules) is more difficult to bond to consistently than is shallower dentin.
Durability of the Bond
How long the bond lasts is more important than how high the initial bond strength is. Over time, exposure of the bonding agents to moisture may cause them to degrade (hydrolyze). In addition, repeated stresses on the bond caused by chewing pressures and temperature changes that cause different amounts of expansion and contraction between the restoration and the tooth structure (measured by the coefficient of thermal expansion) will gradually cause fatigue failure of the bond. (Fatigue failure is similar to taking a piece of metal and repeatedly bending it back and forth until it breaks.) When composite resin is placed and polymerized, it shrinks and can put stress (as much as 20 MPa or 3000 psi) on the bond of the resins to the tooth. In addition, hot and cold foods or beverages can cause composite resin to expand and contract much more so than the tooth (about four times more). If the bond fails, the restoration could leak, causing sensitivity in the tooth or leading to recurrent caries.
It is thought that proteolytic enzymes called matrix metalloproteinases can cause breakdown of the resin bond to dentin. To stop the effect of these proteolytic enzymes, chemicals that inhibit them are being tested. One of these inhibitors is chlorhexidine. If the research confirms its efficacy, the durability of the bond may be enhanced by scrubbing the dentin with chlorhexidine before bonding. Further testing and a protocol on how to use it appropriately are needed.
Enamel Etching
Michael Buonocore introduced etching of enamel into dentistry in the 1950s. Enamel is composed of thousands of rods (prisms) that extend from the dentin to the tooth surface in a radial fashion. Each rod has many millions of crystals composed of hydroxyapatite with about 20% carbonate inclusions. These carbonate imperfections add to the solubility of the crystals in acid. Proteins, lipids, and water in small quantities are found in microscopic spaces between the crystals. Etching of enamel removes a small portion of the surface, reduces the ends of the enamel rods, and opens porosities between adjacent rods (Figure 5-2). The durability and strength of the bond to the enamel depends on how well the etch pattern is developed. Among the different acids tested, phosphoric acid provides the best etch pattern.
Etching Times
The enamel of permanent teeth is usually etched for 20 to 30 seconds with 37% phosphoric acid. Although etching times as short as 10 seconds appear to give good clinical results in some teeth, research results suggest that 20 to 30 seconds is optimal. Highly mineralized teeth may be more resistant to etching and may require up to 60 seconds of etching. Primary teeth should be etched for longer periods (60 seconds or more), because the surface of the enamel has a prism pattern that is not as well structured, is considered aprismatic (without a regular prism pattern), and is more resistant to deep resin tag formation. The etched surface should have a frosty appearance when dried (Figure 5-3). However, when a cavity preparation involves the etching of both enamel and dentin, and the preparation is left slightly moist for wet dentin bonding, it cannot be determined whether the enamel has a frosty appearance.
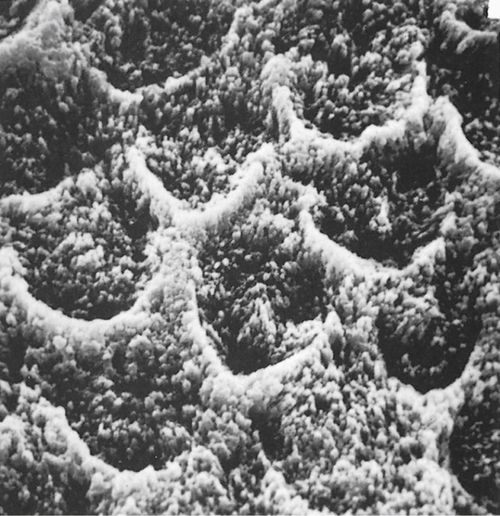
FIGURE 5-2 An etched enamel surface as seen in this scanning electron micrograph has numerous peaks and valleys that provide retention and greatly increase the surface area for bonding. (From Phillips RW, Moore BK: Synthetic resins. In: Elements of Dental Materials for Dental Hygienists and Dental Assistants, Philadelphia, 1994, Saunders.)
Etchant Liquid or Gel
The acid etchant comes as a liquid and a gel. Often coloring agents are added so the practitioner can see where the etchant is on the tooth. Liquid etchants are usually applied with a brush, a small cotton pellet, or a small sponge. Gels are more popular because they stay in place, whereas liquids tend to run without control. Gels contain silica as a thickener. They are usually applied by brush or dispensed from a syringe through a fine needle or brush tip. The recommended rinsing time for acid gels is approximately 10 seconds or longer. Rinsing times shorter than 5 seconds may not remove residual silica. Rinsing times for liquid etchants can be shorter—5 to 10 seconds.

FIGURE 5-4 Cutting of tooth structure with a rotary instrument forms a layer of cutting debris called the smear layer, as seen in this scanning electron micrograph. It is removed by acid etching so that it does not interfere with the formation of a bond. (Courtesy of Grayson Marshall, University of California School of Dentistry [San Francisco, CA].)
Dentin Etching
Smear Layer
The dentin has a higher water and organic content (about 50% by volume) than does enamel (only about 13% by volume). It contains a collagen matrix woven throughout the mineral component (hydroxyapatite) and a system of dentinal tubules through which fluids from the pulp flow. When a cavity preparation is cut with rotary or hand instruments, a layer of cutting debris forms on the surface of the cut dentin and enamel. This layer, called the smear layer, is composed mostly of cut tooth structure and may also contain plaque, bacteria, saliva, and even blood (Figure 5-4). The smear layer sticks tenaciously to the surface, plugs the openings of dentinal tubules, and cannot be washed off with use of an air-water spray. The smear layer is about 2 microns (μm) thick and interferes with the formation of a bond to dentin. Bonding systems that use phosphoric acid to etch the enamel and dentin dissolve the smear layer and rinse it away. Bonding systems that use acidic primers to etch the enamel and dentin penetrate the smear layer and incorporate it into the bonding agent, since there is no rinsing used with these systems. Regardless of the etching method, the smear layer is removed or modified so the bonding agents can penetrate the etched surfaces. Early bonding systems that did not etch the dentin had low bond strengths to dentin because the smear layer was in the way.
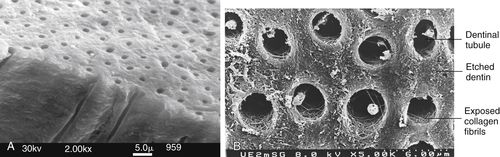
FIGURE 5-5 A, Normal dentin with dentinal tubules. B, Acid etching of the dentin removes some of the mineral exposing the collagen fibers of the matrix, as seen in this scanning electron micrograph. (A, from Sakaguchi, Powers: Craig’s Restorative Dental Materials, ed 13, St. Louis, Elsevier. B, from Dr. Jorge Peridigao, University of Minnesota School of Dentistry, Twin Cities, MN.)
Phosphoric Acid Etching
Etching dentin with phosphoric acid dissolves the smear layer first, and then portions of the hydroxyapatite crystals from the surface of the dentin, creating a porous surface and exposing the meshwork of collagen fibrils that are part of the dentin matrix (Figure 5-5, B). Because dentin is not as highly mineralized as enamel, it should be etched for shorter periods, typically for 10 seconds. With a 10-second etch mineral is removed up to 5 μm in depth from the area between the tubules (intertubular dentin) and from around the periphery of the tubules (peritubular dentin) as well as in the opening of the tubules. Acid that goes into the tubules is neutralized by the fluids that flow from the pulp. When mineral is removed, it leaves a roughened, porous surface (but not the same as with enamel, because there are no rods or prisms).
When etching both enamel and dentin as in a coronal cavity preparation, it is best to apply the acid to the enamel first for 10 seconds, and then to the dentin for 10 seconds. That way enamel will be etched for a total of 20 seconds and dentin only 10 seconds. Etching dentin for 20 seconds or longer opens the tubules too wide and removes hydroxyapatite mineral to too great a depth. Overetching will expose too much collagen matrix, causing it to act as a thick barrier and making it more difficult to coat the dentin and seal the tubules with the resin bonding agent. Overetching dentin can result in a weaker bond and in posttreatment sensitivity.
Moist Dentin for Bonding
The acid is removed by rinsing for at least 10 seconds. The excess water is removed by a gentle stream of air. However, the dentin is left slightly moist so that it glistens but without any puddles of water. It is critical at this stage not to overdry the dentin. The dentin surface must be moist to keep the collagen fibrils fluffed up. If the dentin is dried too much, the collagen fibrils collapse and form a dense surface that occludes the tubules and blocks adequate penetration by the dentin bonding resin. On the other hand, too much water remaining makes it difficult for the resin to displace the water trapped in the collagen fibrils and produces a potential source for leakage. Both overdrying and underdrying can produce an incomplete sealing of the dentinal tubules and a much weaker bond results, because the dentin-resin interface will fracture more easily. A good dentinal seal helps eliminate bacterial leakage and postoperative sensitivity.
Enamel and Dentin Bonding
Enamel Bonding Resins
Bonding agents are low-viscosity resins that flow well into the microscopic porosities and irregularities of the etched surfaces. When bonding to enamel alone, the process is much simpler than bonding to dentin. Etching of enamel creates a high-energy, low-tension surface that makes the surface easier to wet. Bonding to enamel alone requires only a low-viscosity liquid resin monomer that will penetrate into the spaces on and between enamel rods created by acid etching. The liquid may be simply an unfilled resin or may include small amounts of very fine filler particles to enhance the strength of the resin. A high-energy surface attracts the molecules in the resin bonding agent to improve penetration into the porous, etched enamel.
Resin Tags
When the resin is cured by a chemical process or by light activation, it locks into the microscopic spaces and irregularities, producing resin tags that are 10 to 20 μm long and 5 to 6 μm in diameter (Figure 5-6). The resin tags secure the resin to the enamel and create a very strong bond (shear bond strength of more than 20 MPa). The length of the resin tags is determined in part by the orientation of the etched enamel rods. If the enamel on the surface of the tooth is etched (as with bonding orthodontic brackets), then the rods are etched on their ends and the resin penetration is deep. In a class I cavity preparation, the sides of the enamel rods on the walls of the preparation have been exposed. When the sides of the rods are etched, the penetration of the resin is much shallower (about 5 to 10 μm long). Even though there is a difference in the length of the resin tags between end-etched rods and side-etched rods, the bond strengths are not significantly different.
Contaminants on the surface, such as saliva or blood, can dramatically lower the strength of the bond to the enamel. This is one major reason why good isolation is so important. If enamel becomes contaminated after etching, it must be re-etched for 10 to 15 seconds before the bonding process is continued. Because most restorative procedures involve etching both enamel and dentin, a single bonding resin that can be used on both the etched enamel and dentin is preferred.
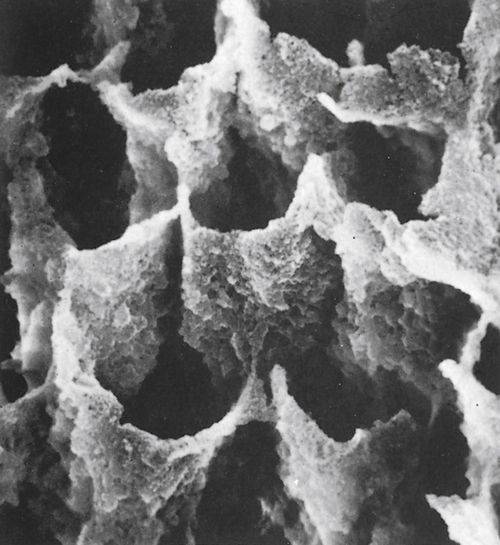
FIGURE 5-6 A resin bonding agent placed on etched enamel penetrates the porous surface and forms resin extensions or tags that lock into the enamel and form a mechanical bond. This scanning electron micrograph shows the resin tags left after the enamel was dissolved away. (From Phillips RW, Moore BK: Synthetic resins. In: Elements of Dental Materials for Dental Hygienists and Dental Assistants, Philadelphia, 1994, Saunders.)
Dentin Bonding Resins
Resin Components
Bonding resins can be viewed as two components. The first is a resin primer that penetrates etched dentin and enamel and lays down a resin layer. The primer is composed of hydrophilic (water-tolerating) monomers and molecules that allow it to penetrate water. HEMA (2-hydroxylethyl methacrylate) is commonly used as a primer monomer. The second component is an adhesive resin that is applied over the primer. The adhesive resins commonly used are bis-GMA (bisphenol-A-glycidyl dimethacrylate), TEGDMA (triethylene glycol dimethacrylate), and urethane dimethacrylate (see Chapter 6 [Composites, Glass Ionomers, Compomers]).
The two resins chemically bond to each other, that is, the initial resin bonding material prepares (or primes) the tooth surface, much in the way that a primer is applied to wood before painting so that the paint will adhere better. The second resin then chemically bonds to the primer. In an effort to simplify the bonding technique, many manufacturers have combined the primer and bonding resin into one bottle to eliminate one step in the process.
“Wet” Dentin Bonding
Bonding to moist dentin (“wet” dentin bonding) was the first technique to achieve good bond strengths to dentin. The dentin is kept moist to keep the collagen fibrils from collapsing into a thick mat that blocks penetration of the bonding agents. A primer is more important on dentin than enamel because the primer contains hydrophilic groups that penetrate wet, etched dentin and keep the meshwork of collagen fibrils expanded so that the adhesive resin can penetrate.
Solvents
For the resin to penetrate through the water, it must be dissolved in a solvent that can penetrate water and carry the resin with it. The solvents allow the resins to penetrate water on the dentin and in the dentinal tubules, and to penetrate around collagen fibrils and into porosities in the tooth surfaces created by etching. The solvents are primarily acetone, ethanol (ethyl alcohol), or a combination of ethanol and water. In general, the solvent is the largest portion of the bonding agent, making up 60% or more of the material. Acetone is a highly volatile solvent. Its rapid evaporation may require that two or more coats of the bonding resin be applied to ensure adequate sealing of the dentin. (An example of a bonding agent with acetone is Prime & Bond NT [Dentsply International].) Ethanol evaporates more slowly, so it may need a longer drying time. (An example of a bonding agent with ethanol is OptiBond Solo Plus [Kerr Corporation].) All bottles of bonding agents should be recapped immediately after the material is dispensed, to prevent evaporation of the solvent, which leads to gradual thickening of the resin with less ability to penetrate etched dentin. Unit-dose (single-use) packaging of bonding agents avoids some of these problems associated with bonding agents in bottles, along with infection control issues.
After the primer is placed, it is dried before it is cured, to remove the solvent and the remaining water. The resin adhesive bonds to the primer and provides a resin-rich layer that will chemically bond to hydrophobic (water-repelling) resin restorative materials such as composite resin that are placed over it. See Procedure 5-1 for bonding to enamel and dentin.
Self-Etching Technique
Bonding systems that do not use phosphoric acid etching instead use an acidic primer that etches and primes in the same step. Acidic primers use water as a solvent. The water allows the acid component to ionize and become active for etching tooth structure. Water can also rewet the collagen fibrils if they have collapsed from drying to allow penetration by the primer. No rinsing and drying is needed with these materials.
Bonding Systems
History of the Development of Bonding Systems
Bonding agents for enamel and dentin have undergone rapid changes over the past four decades (Box 5-1). The composites that became commercially available in the early 1960s were placed without etching or bonding agents. By the beginning of the 1970s, dentists were beginning to etch the enamel with acid and place a self-cured, unfilled bonding resin on the enamel only. This is considered to be the first generation of bonding agents. A number of different acids were tested, but phosphoric acid gave the best results. Great concern was expressed about putting a strong acid (like phosphoric acid) on dentin for fear of damaging the pulp. The dentin was covered with a liner such as calcium hydroxide for protection.
By the late 1970s and early 1980s, both enamel and dentin bonding resins (second generation) were being used, and they were light-cured. The dentin still was not etched, so the bonding resin placed on dentin was merely bonding to the smear layer. The resulting bonds to dentin were very weak bonds (2 to 6 MPa). Later in the 1980s, it was discovered that the smear layer was interfering with the ability of the bonding resins to bond to dentin. Acidic components of the bonding systems (third generation) were used to remove the smear layer, but the dentin was not adequately etched and it was dried. So, bonding resins did not penetrate the dentin surface in a meaningful way. Therefore, bond strengths were still relatively low (bond strength of 12 to 15 MPa).
By the start of the 1990s, etching both enamel and dentin with phosphoric acid (called total etch) was an accepted technique. Not only was the smear layer removed, but the surface of the dentin was etched and kept moist, allowing for the penetration of hydrophilic resin primers into etched dentin surface. The bond strength to dentin increased to about 20 to 30 MPa. The “wet” dentin bonding technique was introduced in the United States by John Kanca. Primer and bonding resins were applied separately (two bottles—fourth generation) or combined into one bottle (fifth generation) and were called total-etch systems.
By the latter part of the 1990s, primers with acidic components that could etch enamel and dentin (called self-etching systems) were introduced and were light-cured or dual-cured (both light and chemical cures). Separate steps for etching with phosphoric acid and rinsing and drying were eliminated with the self-etching primers. This was the sixth generation of bonding systems. These systems had primer and bonding resin in separate bottles.
In the early 2000s, improvements were made in self-etching materials so that components were contained in one bottle (“all in one” adhesives—seventh generation) and did not require mixing. More recently, self-etch flowable composites (e.g., Vertise Flow; Kerr Corporation) have been developed that incorporate the bonding agent into the composite.
Each generation of adhesive systems has consisted of better materials or simpler procedures. The variety of combinations of primers and adhesives has gotten simpler but can still be confusing to the beginner until some experience in using the materials is gained. An easier way to view the current adhesive systems rather than by their generations is to categorize them into one of two basic groups: total-etch bonding systems (also called etch-and-rinse) or self-etch bonding systems (Table 5-1). The first three generations of adhesive systems are no longer commercially available, and therefore this discussion is limited to those still available.
Total-Etch Bonding Systems (Generations 4 and 5)
Total-etch (also called all-etch or etch-and-rinse) refers to phosphoric acid etching of both enamel and dentin with a separate step that includes rinsing off the acid and lightly drying the tooth to leave the dentin moist. Dentin is composed of approximately 70% mineral and 30% collagen and water. When the mineral is removed from the surface of the dentin by the acid etchant, collagen fibrils are exposed and the remaining mineral is roughened on its surface. After the acid is rinsed off, the dentin is left slightly moist (glistening but no water puddles), so that the collagen fibrils stay fluffed up. If the dentin is dried too vigorously, the fibrils collapse into a thick, dense layer that prevents the resin from penetrating through the collagen to reach the etched dentin mineral.
TABLE 5-1
Bonding Systems: Application and Commercial Products
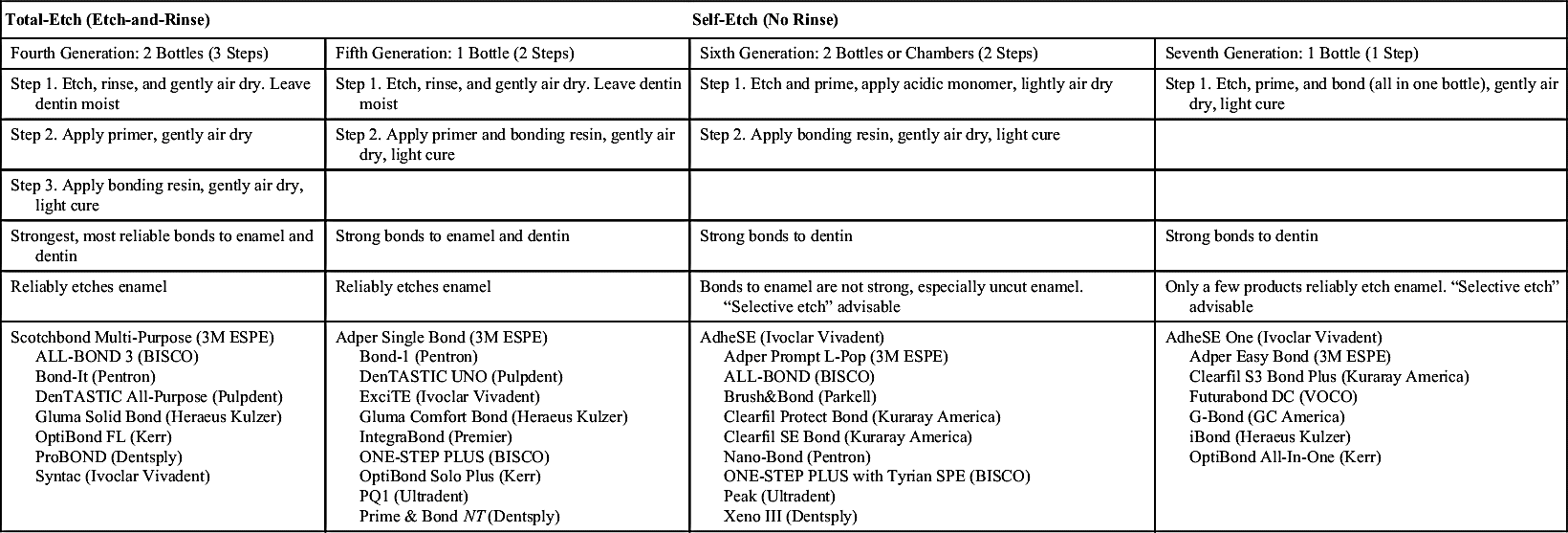
Adapted from Anusavice KJ, Shen C, Rawls HR: Table 12-1. In: Phillips’ Science of Dental Materials, ed 12, St. Louis, 2013, Elsevier.
The total-etch technique became successful when hydrophilic (water-loving) resin monomers (2-hydroxyethyl methacrylate, or HEMA) were added to the primer and adhesive. The hydrophilic monomers facilitated the penetration of the adhesive resin into the moist dentinal tubules and peritubular dentin. When dentin bonding resins are applied to moist dentin, the solvents in which the resins are dissolved allow the resins to penetrate through the water and around the fluffed-up collagen fibrils, into the dentinal tubules and etched dentin surface. Drying with air is done at this stage to remove the volatile solvents from the resin and any remaining water. The resin is then light-cured (or chemical-cured).
Hybrid Layer
The layer that is formed by the intermixing of dentin bonding resin with collagen fibrils and the etched dentin surface is called the hybrid layer (first described by Japanese researchers Fusayama in the late 1970s and Nakabayashi in the early 1980s), because it is a combination (or hybrid) of dentin components and resin. The resin-rich hybrid layer facilitates bonding of the composite resin to the tooth through a chemical resin-to-resin bond (Figure 5-7).
All of the current bonding systems work by micromechanically locking into the etched enamel and by forming a hybrid layer with the dentin.
Because the serous-like fluid in the dentinal tubules neutralizes the etching acid, the walls of the dentinal tubules are etched mostly around the opening and not very far into the tubules. As a consequence, long resin tags extending into the tubules do not add much to the retention of the resin. Most of the retention comes from penetration into the etched mineral of the intertubular and peritubular dentin and around the collagen fibrils. Most systems use light-cured resins.
Two-Bottle Adhesive Systems (Fourth Generation)
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses