Erythropoietic protoporphyria (EPP) is an inherited blood disorder in which formation of the heme group of hemoglobin is defective. Specifically, a deficiency of the enzyme ferrochelatase leads to the accumulation of protoporphyrin, resulting in often painful photosensitivity of the skin and tissues. The prevalence of EPP is estimated at 1:75,000 to 1:200,000. Photosensitivity is exhibited upon exposure to light with specific wavelengths through the creation of reactive oxygen products (oxidants), activation of the complement system, and mast cell degranulation. The aim of this article is to report the orthodontic treatment of an 11-year-old boy with EPP, a Class III skeletal relationship, and an anterior crossbite. Orthodontic treatment established normal overbite and overjet. Short-term periodontal and dental tissue responses to treatment were noted. Extra care was needed when collecting photographic and radiographic records for this patient and during some treatment procedures to avoid causing a photosensitive reaction of the skin or oral mucosa.
Porphyrias are a group of metabolic disorders resulting from defective porphyrin and heme synthesis. They are classified as erythropoietic or hepatic, depending on which organ site expresses the defect in heme synthesis; by the clinical manifestations of cutaneous photosensitivity; or by neurovisceral symptoms. Erythropoietic protoporphyria (EPP) was first described by Kosenow and Treibs (as cited in Lecha ) and further by Magnus et al and is a subset of the erythropoietic porphyrias with a prevalence between 1:75,000 and 1:200,000. EPP is an inherited disorder of the heme metabolic pathway characterized by accumulation of protoporphyrin in blood, erythrocytes, and tissues, resulting in cutaneous manifestations of photosensitivity. The accumulation of protoporphyrin is a result of a deficiency in the ferrochelatase enzyme. Protoporphyrin accumulates in red blood cells and their precursors and eventually diffuses out of the red blood cells into the plasma as the red blood cells mature. Excessive protoporphyrin accumulates in the vascular and perivascular areas and absorbs light energy at 320 to 595 nm on the exposed areas of skin. Peak absorption is in the range of 400 to 410 nm (the Soret band). The energy absorption causes the formation of reactive oxygen products, the activation of the complement system, direct and indirect degranulation of mast cells, and chemotaxis of polymorphonuclear leucocytes.
EPP typically first presents symptoms in early infancy after sun exposure; it is characterized by cutaneous manifestations of acute painful photosensitivity leading to erythema and edema. The severity of the disorder varies significantly, from extreme stinging, itching, and burning sensations to petechiae and, uncommonly, blistering. Affected children might cry and scream for hours, often throughout the night, without an obvious reason. The pain has been described as like “a lighted match being held against your skin”; a 6-year-old child, after crying for a full weekend, asked his mother to “please cut my hands off and let me go to sleep.” Recurrent episodes of unexplained hand and facial swelling can warrant further investigation. It is often diagnostic that the affected person attempts to relieve the pain by applying cold water to the skin.
Because there is no cure for EPP, treatment is focused on prevention and symptom management. Exposure to light within the noxious range should be avoided, with protective clothing and sunscreens to limit photosensitivity. Even sunlight through window glass can precipitate photosensitivity. Other therapies to relieve photosensitivity have been tried, but most lack studies that definitively show clinical relevance ; however, cholestyramine therapy might be helpful to increase fecal excretion of protoporphyrin.
Because of the lack of sunlight exposure and therefore decreased endogenous vitamin D production, supplementation of vitamin D (1,25 dihydroxycholecalciferol) is required to prevent deficiency and to maintain normal bone metabolism and homeostasis. Protoporphyrin blood concentrations and liver function are monitored with regular blood testing, and appearance is assessed for jaundice. Protoporphyrin is not water soluble; therefore, excretion is facilitated by its solubilization in the liver followed by biliary secretion. When the protoporphyrin solubilizing capacity of the liver is exceeded, the result can be hepatic failure, which occurs in approximately 10% of affected persons ; this can be fatal without prompt liver transplantation. During clinical procedures, operating and room lighting must be filtered to avoid inducing severe burns. Dental curing lights emit light wavelengths of approximately 480 nm and should also be considered a potential danger to EPP patients.
This case report describes the nonextraction orthodontic treatment and management of a patient with EPP.
Diagnosis and etiology
The patient, a boy, age 11 years 10 months, sought an orthodontic consultation for his anterior crossbite. His medical history included EPP, allergy to penicillin, and concerns about medications because of the hepatic and kidney associations with EPP. He took vitamin D daily. No liver issues were diagnosed before or during treatment.
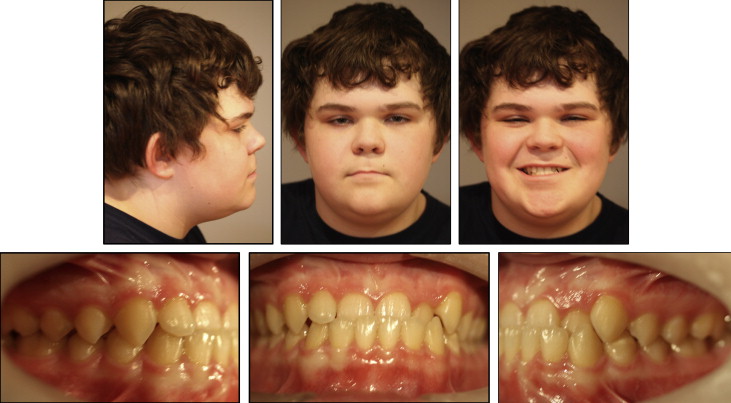
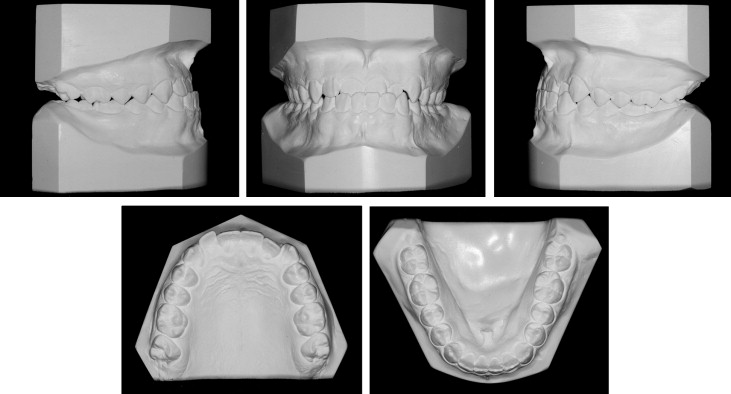
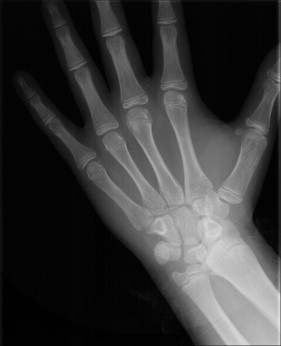
His notable facial attributes included a concave facial profile, a canted mandibular lip on smiling with otherwise normal facial symmetry and facial thirds, a functional shift of the mandible (1 mm anteriorly and 2-3 mm superiorly) ( Fig 1 ). Notable dental attributes included a super Class I molar relationship, a partial anterior crossbite, a −1.8-mm overjet, a 3-mm overbite, adequate mandibular and inadequate maxillary arch lengths, and good dental hygiene ( Fig 2 ). The cephalometric radiographs were analyzed using software (version 11.5; Dolphin Imaging and Management Solutions, Chatsworth, Calif). The adductor sesamoid was present on the hand-wrist radiograph, and the patient appeared to display a cervical vertebral maturation stage 2; therefore, it was likely that pubertal growth was imminent or already attained ( Figs 3-5 ).
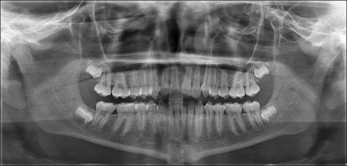
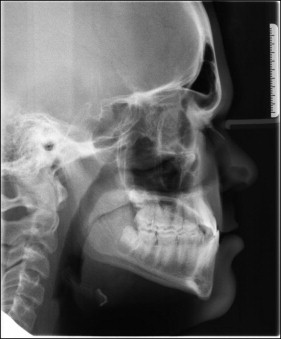
The skeletal analysis involved components from the Indiana University School of Dentistry, American Board of Orthodontics, Bjork, Bjork 2, Michigan Surgery, and Litt Biodynamic analyses (version 11.5; Dolphin Imaging and Mangement Solutions). This variable analysis was used because of the short anterior cranial base, the altered position of nasion, and the decreased angulation of the S-N line because any cephalometric measurement associated with nasion was most likely skewed. The skeletal attributes included a hypodiverent tendency, normal lower face height, decreased ANB and A-B/OP (Wits), and increased SNA and SNB. The relative rather than literal values of SNA and SNB were considered because of the altered position of nasion and the decreases in sella-nasion-basion and SN-articulare angulation. A decreased SN-Ba angulation tends to translate into increased values for SNA and SNB. The maxillary position determined by condylion to anterior nasal spine displayed maxillary retrusion, whereas condylion to pogonion had a normal mandibular position. The maxillary incisors exhibited a decreased inclination relative to the palatal plane and an increased inclination relative to the Frankfort horizontal. The mandibular incisors showed retroclination and increased protrusion relative to the A-point–pogonion line ( Table 1 ). No clicking or pain was reported in the right or left temporomandibular joint. The maximum opening was slightly restricted at 39 mm for a soft end stop, and maximum opening was 49 mm. Lateral excursive and protrusive movements were normal (8-10 mm); the mandible deviated to the left on closure. Tooth and condylar morphologies on the panoramic radiograph showed no overt pathology.
| Pretreatment | Posttreatment | Mean | SD | Dev Normal | |
|---|---|---|---|---|---|
| Skeletal measurements | |||||
| N-A-Pg (°) | −8 | −9.5 | 5.5 | 4.5 | −3 |
| N-A || HP (mm) | 3.6 | 4.5 | −2 | 3.5 | 1.6 |
| N-B || HP (mm) | 13.4 | 16.8 | −8.5 | 4.5 | 4.9 |
| N-Pg || HP (mm) | 13.8 | 18.4 | −9 | 5 | 4.6 |
| A-B || HP (mm) | 9.8 | 12.2 | −6.5 | 2.5 | 6.5 |
| A-B || OP (mm) | −5.5 | −6.4 | −0.5 | 2 | −2.5 |
| FH-SN (°) | 2.1 | 2 | 6 | 4 | −1 |
| SNA (°) | 87.1 | 88.2 | 82 | 3 | 1.7 |
| SNB (°) | 91.7 | 93.8 | 80 | 4 | 2.9 |
| ANB (°) | −4.6 | −5.7 | 2 | 1.5 | −4.4 |
| OP-HP (°) | −6.6 | −8.7 | 9 | 2.5 | −6.2 |
| PP-HP (°) | −13.3 | −13 | 0.5 | 3 | −4.6 |
| MP-HP (°) | 14.3 | 14.2 | 25 | 5 | −2.1 |
| Palatal-maxillary occlusal plane (°) | 9.9 | 6.1 | 10 | 4 | 0 |
| Mandibular occlusal-mandibular plane (°) | 24.6 | 24.5 | 20 | 5 | 0.9 |
| Chin angle (Id-Pg-MP) (°) | 74.8 | 73.9 | 70 | 5 | 1 |
| Y-axis (S-Gn-FH) (°) | 50.1 | 49.5 | 60 | 3 | −3.3 |
| Gonial/jaw angle (Ar-Go-Me) (°) | 131.2 | 130.5 | 124.4 | 6.7 | 1 |
| Chin angle (Id-Pg-MP) (°) | 74.8 | 73.9 | 70 | 5 | 1 |
| Anterior cranial base (SN) (mm) | 68.6 | 69.6 | 73.9 | 3 | −1.7 |
| Posterior cranial base (S-Ar) (mm) | 38.4 | 39 | 33.5 | 4 | 1.2 |
| Ramus height (Ar-Go) (mm) | 50.4 | 53.8 | 45.3 | 4.5 | 1.1 |
| Length of mandibular base (Go-Pg) (mm) | 75.3 | 82.7 | 73 | 3 | 0.8 |
| Maxillary unit length (Co-ANS) (mm) | 83.1 | 82.8 | 90 | 5 | −1.4 |
| Mandibular unit length (Co-Pog) (mm) | 113.4 | 116.8 | 113 | 8 | 0.1 |
| Harvold (CoPog)-(CoANS) (°) | 30.3 | 34.1 | 20 | 3 | 3.4 |
| Maxillary skeletal (A-Na perp) (mm) | −0.7 | 0.1 | 0 | 3.1 | −0.2 |
| Mandibular skeletal (Pg-Na perp) (mm) | 5.4 | 9.3 | −4 | 5.3 | 1.8 |
| MP-SN (°) | 21.3 | 21.2 | 33 | 6 | −1.9 |
| Upper face height (N-ANS) (mm) | 45.4 | 46.9 | NA | NA | NA |
| Lower face height (ANS-Me) (mm) | 61.7 | 65.5 | NA | NA | NA |
| Upper face height (N-ANS/[N-ANS+ANS-Me]) (%) | 42.4 | 41.7 | 43 | 100 | 0 |
| Lower face height (ANS-Me/[N-ANS+ANS-Me]) (%) | 57.6 | 58.3 | 57 | 100 | 0 |
| Ba-S-N (°) | 124.7 | 124.1 | 130 | 100 | −0.1 |
| SN-Ar (°) | 120.1 | 119.5 | 124 | 5 | −0.8 |
| Dental measurements | |||||
| 1/(HP) (°) | 119.6 | 133.8 | 110 | 6.5 | 1.5 |
| /1(HP) (°) | 78.6 | 80 | 60 | 4.5 | 4.1 |
| 1/-(UL) (mm) | 2.4 | −0.2 | 2.5 | 1 | −0.1 |
| IMPA/1 (MP) (°) | 87.1 | 85.8 | 90 | 5 | −0.6 |
| Interincisal angle (U1-L1) (°) | 139 | 126.2 | 130 | 6 | 1.5 |
| /1(A-Pg) (mm) | 5.4 | 6.2 | 2.5 | 2 | 1.4 |
| /1(N-Pg) (mm) | 3.2 | 3.5 | 3.5 | 2.5 | −0.1 |
| U1-SN (°) | 112.6 | 126.8 | 102.6 | 5.5 | 1.8 |
| U1-ANS (mm) | 26 | 26.6 | 30 | 5 | −0.8 |
| U6-PP (UPDH) (mm) | 21 | 22.3 | 20 | 3 | 0.3 |
| L1-MP (LADH) (mm) | 39.5 | 42 | 40 | 3 | −0.2 |
| U1-Me (mm) | 5.7 | 5.6 | 35 | 3 | −9.8 |
| L6-MP (LPDH) (mm) | 27.4 | 30.7 | 30 | 3 | −0.9 |
| Pr-N-A (°) | 3.9 | 7 | 2 | 1 | 1.9 |
| U1-palatal plane (°) | 106.3 | 120.8 | 110 | 5 | −0.7 |
| U1-NA (mm) | 6.1 | 11.4 | 4 | 2.7 | 0.8 |
| GoGn-chin line (°) | 78.1 | 76.4 | 70 | 5 | 1.6 |
| L1-GoGn (°) | 90.3 | 88.3 | 94 | 7 | −0.5 |
| L1-NB (mm) | 2.3 | 2.6 | 4 | 1.8 | −0.9 |
| Pg-NB (mm) | −1.2 | −1.3 | NA | NA | NA |
| Overjet (mm) | −1.9 | 1.7 | 2.5 | 2.5 | −1.8 |
| Overbite (mm) | 3.6 | 1.8 | 2.5 | 2 | 0.5 |
| Soft-tissue measurements | |||||
| Facial convexity (G′-Sn-Po′) (°) | −1.5 | −4.1 | 13.5 | 5.5 | −2.7 |
| UL protrusion (UL-SnPg′) (mm) | 2.9 | 2.5 | 3.5 | 1.8 | −0.3 |
| LL protrusion (LL-SnPg′) (mm) | 5.7 | 3.5 | 2.8 | 1.7 | 1.7 |
| UL to E-plane (mm) | −5.2 | −6.2 | −3 | 2 | −1.1 |
| LL to E-plane (mm) | 0.2 | −2.5 | −2.5 | 2.5 | 1.1 |
| ILG (HP) (mm) | 1.2 | −0.7 | NA | NA | NA |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


