Fig. 2.1 Anterior view of oral cavity and oropharynx.
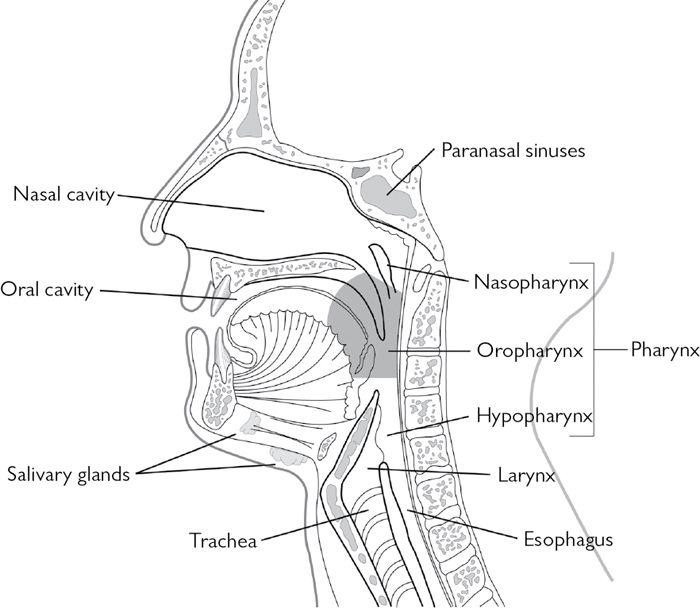
Fig. 2.2 Sagittal view of oral cavity and oropharynx.
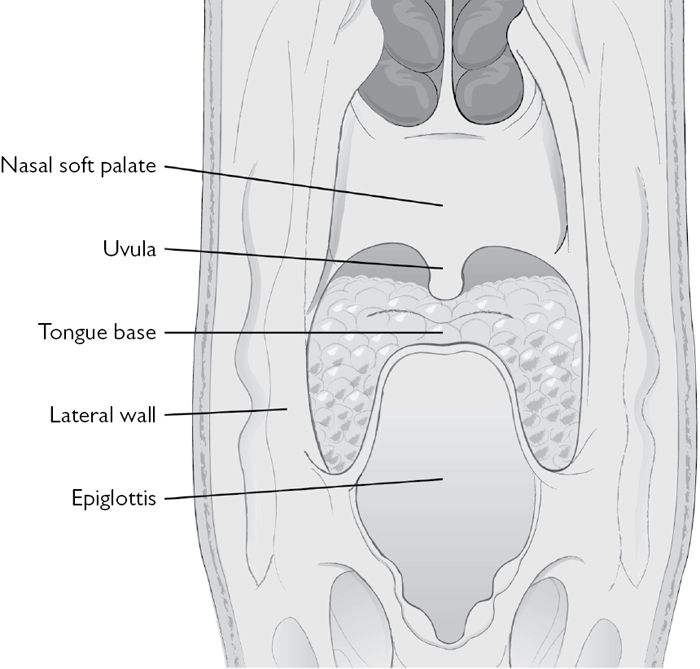
Fig. 2.3 Posterior view of oropharynx.
Epidemiology
• Around 7500 cases of all head and neck cancer types occur in the UK per annum, making it the 6th most common form of cancer.
• Approximately 2600 occur in the oral cavity and 900 in the oropharynx.
• In the USA, approximately 30,100 new diagnoses of oropharynx carcinomas are made and an estimated 7800 deaths occur.
• In India, oral cavity and oropharyngeal tumours comprise 40% of cancers.
• There is between 2 and 5 times greater incidence in men than women depending on site.
• Traditionally, it is most common in the 6th and 7th decades, although there is evidence that it is increasing in young adults.
Aetiology
• Smoking and consumption of alcohol are the two principal aetiological factors in this disease. These factors act separately but also synergistically in causing this form of cancer.
• Diet containing high proportions of vegetables and fruit might modulate carcinogenic effects and low body mass index (BMI) appears to increase the risk of oropharynx cancer.
• Human papilloma virus (HPV) is now recognized as an independent risk factor for oropharyngeal squamous cell carcinoma—HPV type 16 is the most prevalent genotype present in 87% of cases of oropharyngeal squamous carcinoma in one large study.
• Betel quid chewing is related to the high incidence of oral cancer in the Indian subcontinent.
Premalignant conditions
Conditions of definite premalignant potential
• Leukoplakia.
• Erythroplakia (Fig. 2.4).
• Chronic hyperplastic candidiasis.
Conditions associated with an increased risk of malignant transformation
• Lichen planus.
• Oral submucous fibrosis.
• Syphilitic glossitis.
• Sideropenic dysphagia.
These include conditions are that not themselves premalignant, but confer an ↑ rate of malignancy by the atrophy that they produce within the mucous membranes (see ![]() Lichen planus, p. 432, and
Lichen planus, p. 432, and ![]() Syphilis, p. 460).
Syphilis, p. 460).
Conditions erroneously considered to be premalignant
• Stomatitis nicotina.
• Habitual cheek biting.
• White sponge naevus.
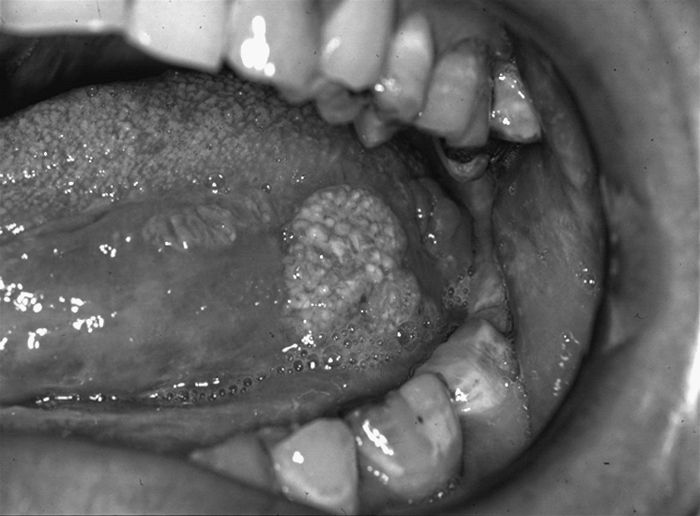
Fig. 2.4 Oral squamous cell carcinoma presenting in area of erythroplakia.
Diagnosis
This is made on the basis of clinical features and special investigations. Clinical features include symptoms and signs.
Symptoms
In early disease small tumours may produce very few or only specific symptoms, and are therefore difficult to detect. Presenting symptoms may include:
• Painless ulcer.
• Sore throat.
• Sensation of a foreign body in the throat.
• Change in voice, otalgia, or odynophagia (pain referred to the ear mediated via glossopharyngeal and vagus nerves).
• Lump in the neck.
• Weight loss.
With increasing tumour size, tongue movement may be impaired, affecting speech and swallowing. In late stage disease patients may complain of spontaneous bleeding and halitosis.
Signs
• Indurated ulcer (Fig. 2.5).
• Exophytic mass.
• Area of abnormal mucosa, e.g. red patch.
• Cervical lymphadenopathy: (Fig. 2.6) may be reactive or metastatic. A metastatic node will classically be:
• hard;
• non-tender;
• may be fixed to surrounding structures.
• Alteration of voice.
• Cranial nerve lesions:
• hypoglossal palsy (deviation towards affected side with wasting and fasciculation of ipsilateral side of tongue);
• vagus nerve palsy (impaired movement of soft palate, ipsilateral vocal cord paralysis);
• trigeminal nerve anaesthesia in territory supplied by lingual nerve and mental nerves.
Clinical examination must include a methodical examination of the oral cavity, as well as the anterior and posterior triangles of the neck. Fibre-optic naso-endoscopy must always be carried out when an oropharyngeal lesion is suspected. This can be carried out under topical anaesthesia.
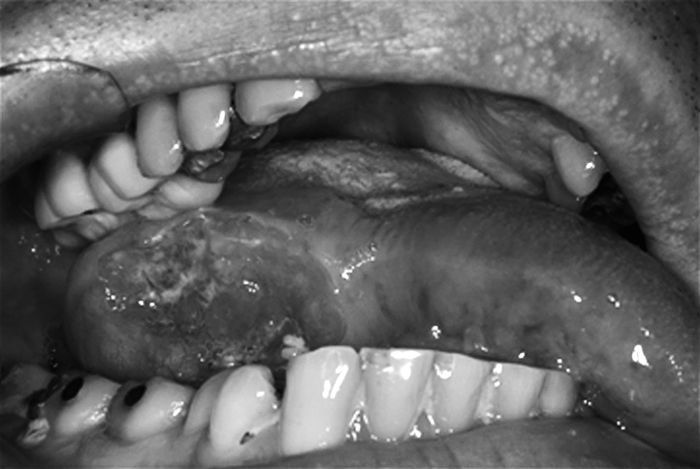
Fig. 2.5 Oral squamous cell carcinoma presenting as indurated ulcer.
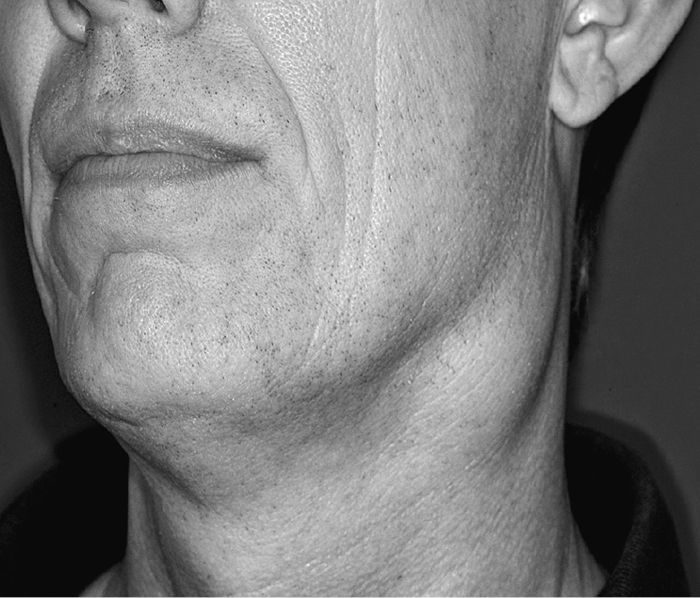
Fig. 2.6 Upper deep cervical lymph node enlargement from oral cavity primary.
Investigations
Plain radiography
Plain radiographs have a limited role to play in the diagnosis and pre-operative assessment of oropharyngeal tumours. Nonetheless, they are frequently performed as part of the diagnostic pathway, particularly in patients who present with non-specific symptoms.
The orthopantomogram
• Frequently undertaken as a screening investigation for patients presenting with facial pain.
• A rotational tomogram and, therefore, only of diagnostic value for those tissues that lie within the focal trough.
• Of little practical value in assessing soft tissue pathology.
• Bony pathology is only demonstrated where significant decalcification has occurred as a result of either pressure or direct invasion by a malignant process.
• Occasionally useful in diagnosing the much rarer osteosarcomas where characteristic features, including the ‘sunray’ appearance, are almost pathognomonic.
• Bone invasion by the more common squamous cell carcinomas is indicated by loss cortical outlines and irregular areas of radiolucency in either the mandible or maxilla (Fig. 2.7). Rarely resorption of dental roots can also be identified.
• Malignant transformation within pre-existing benign pathology cannot be excluded with plain radiology.
• Multiple lesions may occur with metastatic bony deposits, such as in myeloma or breast carcinoma.
• Main value of the OPG in patients with oropharyngeal malignancy is in aiding dental assessment prior to surgery or radiotherapy:
• planning of mandibular resections or access osteotomies must take into account the position of the teeth in order to minimize functional disability;
• extent of caries and periodontal disease can be determined in order to ensure that appropriate therapy including dental extractions are undertaken preferably at the examination under anaesthesia (EUA) stage, and certainly prior to the commencement of major therapeutic interventions.
• Finally, the OPG is used to confirm the appropriate reduction and fixation of access mandibulotomies and to ensure that, where mandibular reconstruction has been undertaken, bony contour, plate position, and accurate location of the condyle within the glenoid fossa have all been achieved.
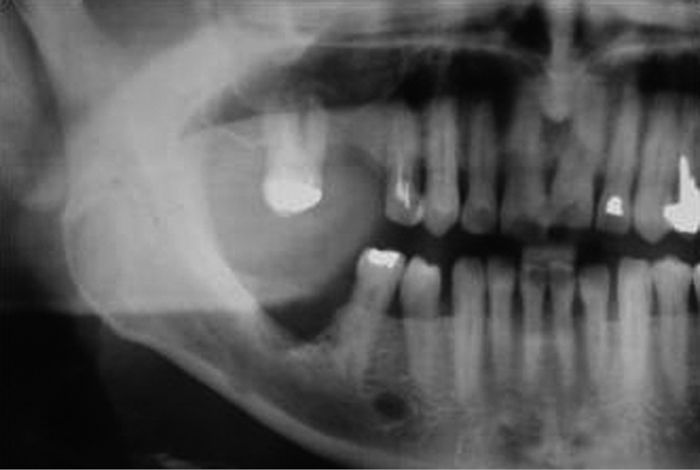
Fig. 2.7 Orthopantomogram demonstrating mandibular invasion in molar region.
Occipito-mental radiograph
• Seldom used in modern diagnostic practice outside the realms of facial trauma.
• Occasionally indicated in the investigation of facial pain thought to be of maxillary sinus origin.
• Opacity of maxillary sinus coupled with bony destruction is most likely to indicate a malignant process although long-standing chronic suppurative disease (e.g. aspergillosis) can give rise to similar appearances.
Chest radiograph (CXR)
The pre-operative CXR still has a number of important functions in the overall assessment of patients with oropharyngeal malignancy.
• Many patients present with cardiorespiratory co-morbidities and the CXR can form a valuable tool in supplementing clinical assessment of respiratory and cardiac function.
• Given the high incidence of post-treatment complications involving the cardiorespiratory system, a pre-treatment baseline CXR can provide invaluable information about the developing clinical picture.
• May demonstrate pulmonary metastases, although it is not as sensitive as other imaging modalities in the detection of smaller lesions. Nonetheless, a CXR that demonstrates widespread pulmonary metastases at the beginning of the diagnostic pathway may save a great deal of unnecessary time and investigation, and allows the patient to receive appropriate therapy at a much earlier stage.
• The role of CXR as part of the routine follow-up of patients following definitive therapy is less certain. In general, follow-up radiography is only recommended where symptoms dictate. Follow-up screening for pulmonary metastases or second primary malignancies is not recommended.
Contrast radiography
Sialography
Whilst sialography has no role to play in the planned assessment of a patient with known oropharyngeal malignancy, occasionally patients with such a malignancy may present with symptoms consistent with obstructive sialoadenitis. It is important to recognize that space-occupying lesions within the major salivary glands or extrinsic compression of the ducts may be identified on sialography.
Carotid angiography
High-resolution contrast MRI, duplex ultrasound scans (DUSS), and subtraction angiography have largely superseded the need for carotid angiography in the pre-operative assessment of oropharyngeal malignancy. Occasionally, however, where previous neck surgery or radiotherapy has been undertaken, angiography may be required in order to assess the patency and condition of the vascular tree in order to facilitate appropriate reconstructive choice. In highly vascular tumours embolization may be of value as an adjunct to surgery.
Barium swallow
This investigation can give useful information about tongue base lesions. It is of particular value when coupled with naso-endoscopy in the post-operative assessment of swallowing function.
Cross-sectional imaging
Computed tomography
• High-resolution CT scanning with and without contrast probably represents the best all-round imaging modality in the pre-operative assessment and staging of oropharyngeal malignancy.
• Details of bony anatomy and potential bone involvement by tumour are superior to MRI.
• Single CT scan of the head, neck, thorax, and upper abdomen allows a comprehensive staging scan to be undertaken rapidly at one sitting.
• In order to assess the para-nasal sinuses coronal scans can be undertaken or reformatted and give excellent detail regarding involvement of the orbit and cranial base. Sagittal scans can also be of value in the fronto-nasal area.
• CT is widely available and the criteria for determining the probability of nodal involvement are well developed.
• Main drawbacks of CT:
• distortion of images caused by amalgam artefact;
• limitations of imaging smaller lesions in mobile areas of the oropharynx.
• Whilst exposure doses have been significantly reduced with modern CT machines, this modality still has the disadvantage of subjecting patients to ionizing radiation.
• CT scan provides a high level of accuracy in assessing the involvement of lymph nodes in the neck (Fig. 2.8). The criteria for identifying positive neck nodes are:
• increase in size (short axis >8–10mm);
• central necrosis and rim contrast enhancement;
• extra-capsular extension;
• obliteration of surrounding fat planes.
• Surpasses plain radiography in diagnostic accuracy for determining the presence of pulmonary metastases or early primary lung tumours.
• Since image acquisition times with modern spiral scanners are short, there is little reason for not undertaking chest examination as part of a standard head and neck staging protocol.
• There remains the method of choice for radiotherapy treatment planning. However, an additional ‘planning CT scan’ is usually required with the patient positioned in the treatment position.
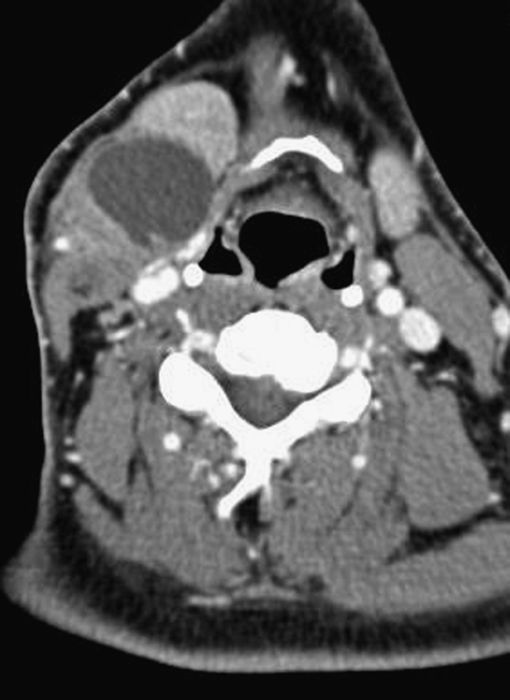
Fig. 2.8 Positive node identified on CT by imaging criteria.
Magnetic resonance imaging
• Superior to CT scanning for determining the depth and volume of primary soft tissue oropharyngeal tumours.
• Not susceptible to dental amalgam artefact.
• No exposure to ionizing radiation.
• Coupled with administration of gadolinium gives excellent information about potential proximity to vascular and neural structures.
• Acquisition times greater than for CT.
• Some patients find it unbearably claustrophobic and noisy.
• High false positive rate when assessing patients for recurrent tumour following therapeutic intervention.
• High-resolution MRI scans can be of particular benefit in identifying small naso-pharyngeal tumours and recurrence at the base of skull.
• Investigation of choice for assessing primary salivary gland malignancies and in particular is of value for identifying perineural spread.
• Gives almost equivalent accuracy to the CT for detecting cervical node metastases (criteria used to determine nodal involvement are similar to CT).
• Contraindicated in presence of ferrous implants.
Nuclear medicine
Bone scintigraphy
• Radioisotope scanning with technetium 99-labelled methylene diphosphonate (MDP) can be of benefit in determining whether early bony invasion exists.
• Computerized co-location on sectional anatomical images can be of benefit in identifying areas of bony invasion where no change is identified on either the CT or plain radiographs.
• Highly sensitive test and false positives may occur where there is active dental pathology, if there has been a recent biopsy, or where there is simply a periosteal reaction.
• Results of scintigraphy should be considered in the context of the clinical picture and other investigations. When positive, but where significant clinical doubt exists, an intra-operative periosteal strip should be performed before committing the patient to a segmental bone resection.
• Also of value in assessing the viability of microvascular free bone transfers in the post-operative period.
Positron emission tomography (PET)
• PET provides a functional assessment of tissue metabolism using radiolabelled fluoro-deoxy-glucose (FDG).
• Should give the maximum diagnostic information by distinguishing between the metabolic behaviour of cancer and normal tissues.
• Studies in the head and neck indicate that this test has a high diagnostic accuracy particularly for:
• detecting primaries of unknown origin;
• detecting tumour recurrence by distinguishing between active tumour and post-treatment scar tissue.
• Requires the use of radiolabelled products that only have a short half-life—therefore expensive.
• At present PET should be reserved for selected cases where a primary cannot be identified or where tumour recurrence is suspected, but cannot be detected by other methods, and patients should be considered for entry into ongoing clinical trials.
• It is important that no therapeutic intervention or biopsy has been recently undertaken in order to limit the false positive rate.
Ultrasonography
• Diagnostic ultrasound has become an important adjunct to the clinical assessment of neck masses—does not replace the requirement for either a CT or MRI scan since these give much more anatomical detail and permit staging of the neck, thorax, and abdomen.
• Accurate determination of the size and morphology of neck masses can be made, and in many cases normal and abnormal architecture of cervical lymph nodes can be assessed.
• When used in conjunction with fine needle aspiration cytology or biopsy (FNAC or FNAB) enhances diagnostic yields.
• DUSS of value in assessing the patency of the vascular tree and vascularity of the tumour.
• Does not expose the patient to ionizing radiation or strong magnetic fields.
• Simple to undertake.
• Highly operator dependent. Real-time imaging provides much more valuable information than can be conveyed in static images.
• Has a high diagnostic accuracy in the detection of salivary gland tumours; however, its ability to discriminate between benign and malignant tumours is less certain.
• If coupled with FNAC or FNAB, USS diagnostic accuracy for salivary neoplasms is improved, but high false positive and false negative rates for cytology still exist such that radical surgery cannot be justified on the basis of USS and cytology alone (however, demonstration of diffuse enlargement of gland may justify incisional biopsy to distinguish an inflammatory process from lymphoma). USS can be of benefit in monitoring patients with Sjögren’s syndrome and assess whether a discrete lymphoma is developing within sialectatic salivary tissue (see ![]() Sjögren’s syndrome, p. 186).
Sjögren’s syndrome, p. 186).
Fine needle aspiration for cytology or biopsy
FNAC
• Of benefit when abnormal squamous cells are identified from aspiration of a mass in the neck—triggers diagnostic pathway in search of a primary oropharyngeal malignancy. Where a metastasis from a mucosal malignancy is suspected, every attempt should be made to reach a sound diagnosis without resort to compromising the neck with open surgery.
• Where abnormal lymphocytes are identified suggesting lymphoreticular malignancy, cytology is rarely sufficient to determine the most appropriate therapy—next most appropriate diagnostic step is excision biopsy through a defensive neck incision (where the approach allow for a future neck dissection).
• Risk of seeding using FNAC is believed to be extremely remote—nonetheless, where there is unequivocal histological diagnosis of the primary tumour, and imaging of the neck demonstrates abnormal nodes on size or architectural criteria, any additional information obtained by FNAC is of questionable benefit as it is unlikely to influence therapeutic decisions.
FNAB
• Offers greater information regarding tumours because it provides not only evidence of abnormal cytology, but also information regarding the histology of the tissue.
• Occasionally will provide sufficient information regarding lymphomas to initiate treatment without resort to formal nodal excision biopsy.
• Theoretical risk of seeding using this technique, but there is no evidence to demonstrate a true risk.
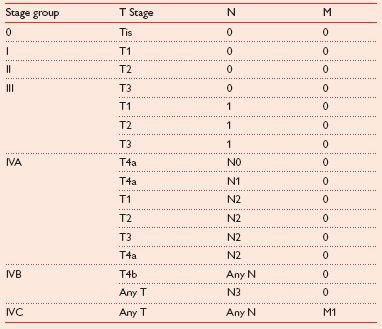
Staging
Oropharynx cancer is staged using the TNM system (tumour, lymph nodes, distant metastases). Staging can be further classified as clinical, radiological (based on imaging), or pathological (based on the outcome of histopathological examination of excised tissue).
T stage
The primary tumour is staged on the basis of surface extent (Table 2.1).
Table 2.1 T stage
| T stage | Tumour maximum surface diameter | ||
| Tx | Primary tumour cannot be assessed | ||
| T0 | No primary tumour | ||
| Tis | Primary lesion contains in situ cancer only | ||
| T1 | <2cm | ||
| T2 | >2cm <4cm | ||
| T3 | >4cm | ||
| T4a | Tumour invades larynx, deep/extrinsic muscles of tongue, medial pterygoid muscle, hard palate, or mandible | ||
| T4b | Tumour invades lateral pterygoid muscle, pterygoid plates, lateral nasopharynx or skull base, or encases carotid artery | ||
N stage
The cervical lymph nodes are staged as shown in Table 2.2.
Table 2.2 N stage
| N stage | Nodal status | ||
| Nx | Regional nodes cannot be assessed | ||
| N0 | No regional lymph node metastasis | ||
| N1 | Metastasis in a single ipsilateral node 3cm or smaller in maximum dimension | ||
| N2 | Metastasis in a single ipsilateral node >3cm, but 6cm or less in greatest dimension, or in multiple ipsilateral nodes 6cm or less, or in bilateral or contralateral lymph nodes 6cm or smaller | ||
| N2a | Metastasis in a single ipsilateral lymph node >3cm, but <6cm | ||
| N2b | Metastases in multiple ipsilateral nodes 6cm or smaller | ||
| N2c | Metastases in bilateral or contralateral nodes 6cm or smaller | ||
| N3 | Metastasis in a lymph node >6cm | ||
When evaluating nodal metastases clinically the actual size of the mass is measured taking account of the intervening soft tissues. Most neck masses >3cm are not single nodes, but confluent nodes or tumours in soft tissues of the neck.
M stage
Distant metastases are staged as shown in Table 2.3.
Table 2.3 M stage
| M stage | Distant metastasis | ||
| MX | Distant metastasis cannot be assessed | ||
| M0 | No distant metastasis | ||
| M1 | Distant metastasis | ||
Disease stage
Taking the TNM status together for a tumour, the stage of disease is as shown in Table 2.4.
Table 2.4 Disease stage
Principles of treatment
Treatment intent—to cure or palliate?
• Having established a diagnosis of oropharyngeal malignancy, the histological type, and the stage of disease it is important to take account of the overall physical and mental state of the patient.
• The relative risks and benefits of the various treatment options should be identified and it should be ensured that the patient, and preferably also their relatives and carers, fully understand the implications of treatment and are in a position to make an informed choice.
• Where remediable factors exist in terms of general co-morbidities these should be addressed prior to treatment decision.
• Treatment involves three potential modalities:
• surgery;
• radiotherapy;
• chemotherapy.
• Achieving loco-regional control is key and relates directly to overall survival.
• Management of the primary cancer can be either surgical or by radiotherapy, and remains controversial. Surgery is often combined with adjuvant radiotherapy based on histopathological outcomes of surgical resection.
• Ionizing radiation and chemotherapy are used increasingly for this patient group as a combined modality.
• Management of stage III and IV resectable oropharynx cancer—there has been only one prospective randomized controlled trial (RCT) comparing chemoradiotherapy with primary surgery plus adjuvant therapy.
• In early disease, surgery and primary radiotherapy are equally effective in eradicating primary disease.
• All surgical complications are well known to be far more prevalent and complex in the post-radiotherapy patient.
• Quality of life should be considered alongside the chances of survival.
• All members of the multidisciplinary team (MDT) should have the opportunity to express their views about treatment options from their own perspectives. A designated member of the team should take a lead role and ultimately be responsibility for the management of an individual patient’s care.
• A head and neck MDT should comprise of:
• oral and maxillofacial surgeons;
• ear, nose, and throat (ENT) surgeons;
• reconstructive surgeons (oral and maxillofacial surgery (OMFS), ENT, or plastic);
• specialist anaesthetists;
• clinical/medical oncologists;
• specialist nurses;
• speech and language therapists;
• palliative care physicians;
• specialist pathologists;
• dieticians;
• restorative dentists;
• dental hygienists;
• psychologists.
Having weighed the evidence and discussed all the treatment options with the patient it is vital that the intention of treatment is clearly understood by the whole team, patient, and carers. Where treatment is given with palliative intent it is important that this is targeted at the specific relief or prevention of symptoms, and that the treatment is designed to minimize side effects. Regular reassessment is required to ensure that therapeutic goals are being achieved and that the side effects of treatment are not outweighing the benefits. There must be a clear evidence-based stepwise strategy for managing common symptoms such as pain and nausea.
When appropriate, advance life directives should be discussed and an end of life resuscitation policy agreed with the patient and preferably carers.
Therapeutic options
The overwhelming majority of oropharyngeal cancers are mucosal squamous cell carcinomas. Most of the discussion regarding therapeutic options will be directed to the management of these relatively common tumours. There are three main treatment modalities to consider:
• Surgery.
• Radiotherapy.
• Systemic anti-cancer therapies (SACT).
These modalities can be used in isolation or combination. When used in combination surgical resection normally precedes adjuvant radiotherapy as post-irradiation surgical resection carries with it a much higher risk of complications. Primary chemoradiation may be indicated for certain tongue base and tonsillar tumours. However, the use of chemotherapy as a stand-alone primary modality or as adjuvant treatment with surgery is often only undertaken in the context of clinical trials. It is not possible to consider every permutation and combination, but the following factors all have a bearing on the choice of treatment:
• Site of primary tumour.
• Stage of disease.
• Proximity or involvement of bone.
• Treatment intent.
• Physical status of patient.
• Patient preference.
As far as the physical status of the patient is concerned a number of risk stratification models are available to assist the MDT in advising the patient. At the simplest level, the American Society of Anaesthesiologists (ASA) I–V scale gives a reasonable prediction of the risk of morbidity and mortality (see ![]() Anaesthesia, p. 376). Pre-operative risk stratification not only assists in choosing the modality of treatment and even the extent of surgery, but is also a prerequisite for meaningful comparative audit.
Anaesthesia, p. 376). Pre-operative risk stratification not only assists in choosing the modality of treatment and even the extent of surgery, but is also a prerequisite for meaningful comparative audit.
Surgery
There are a number of different options available under the banner of surgery:
• Conventional excision.
• Laser surgery.
• Thermal surgery.
The primary site
• Choice of primary modality treatment depends upon the tumour site and histological features, coupled with the stage of disease and ultimately patient choice.
• Size and location of tumour will determine whether a resection followed by primary closure or healing by secondary intention will be satisfactory, or whether reconstruction of the defect with a local or distant tissue will be necessary to restore form and function.
• In borderline cases there may have to be careful consideration of the trade-off between loss of function and the risks of the procedure.
Access to the primary tumour
Many tumours in the anterior part of the oral cavity can be accessed via the transoral route. This is ideal since the oral sphincter is maintained and no scars are produced above the jaw line. The cosmetic result is usually excellent. However, as tumours increase in size and as their position becomes more posterior it may not be feasible to undertake a safe resection via the transoral route. Three main alternatives exist to achieve access under these circumstances:
• Lip split and mandibulotomy (Fig. 2.9 and Fig. 2.10).
• A ‘pull through’ technique via the neck.
• For maxillary tumours, an upper lip and para-nasal incision (lateral infraorbital extension is rarely required and has a high complication rate).
• Transoral robotic surgery may be of benefit in performing ablative surgery, in otherwise inaccessible site. However this technique is still under evaluation and should be restricted to a small number of specialist centres.
There are several options for the lip skin incision. However, in most circumstances, some form of stepped incision is desirable both in the upper and lower lip. This disguises and lengthens the scar to prevent post-operative wound contraction, which otherwise distorts the vermilion border. After a lengthy operation the tissues can become oedematous by the time closure is undertaken and it is therefore advisable to mark the skin with methylene blue temporary tattoos prior to incision in order to ensure accurate apposition during closure. It is also important to ensure that the orbicularis muscles are correctly realigned in order to maintain an effective oral sphincter and prevent unsightly post-operative clefting of the lip.

Fig. 2.9 Operative photograph of lip split and mandibulotomy.
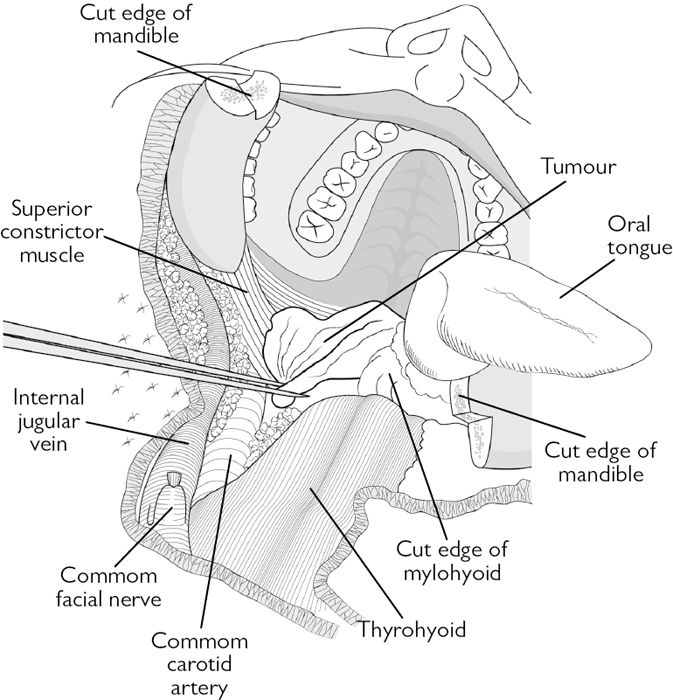
Fig. 2.10 Line diagram of Fig. 2.9.
Ablation
• Effective tumour ablation is achieved by ensuring that good visibility is maintained. This in turn results from appropriate access.
• In order to maximize the chances of achieving complete tumour resection with a clear margin of normal tissue the surgeon must employ both visual inspection and palpation.
• The method of cutting (cold steel, harmonic scalpel, laser, diathermy, or coblation) is, to some extent, a matter of personal preference. In many cases, a combination of techniques will be used.
• For some small and superficial lesions laser vaporization may be employed; however, this does not permit histological assessment of the adequacy of tumour resection. Similarly, lasers and thermal techniques, whilst reducing the amount of intra-operative bleeding, cause morphological distortion of tissues at the margins, which can lead to histological artefacts and some difficulty in assessing the adequacy of tumour resection.
• Coblation involves the generation of bipolar radiofrequency waves that generate tissue temperatures of around 60°C, much lower than temperatures generated by conventional diathermy. This is claimed to reduce post-operative pain, but the technique has been associated with ↑ levels of post-operative haemorrhage. It should therefore only be used by surgeons who have had specific training in its use.
• Use of intra-operative frozen sections to assist marginal clearance is controversial:
• given that the primary aim is tumour resection with a clearance preferably with clinical margin of 1cm (vital structures permitting) if the surgeon believes that close or positive margins could be possible then a wider resection should be undertaken if feasible;
• random frozen sections are unlikely to be able to identify positive margins;
• they may give a false sense of security to the surgeon and invariably prolong the operative time.
• Intra-operative tumour tissue marking has been attempted (e.g. toluidine blue), but this has limited value at detecting mucosal margin clearance because of the high false positive rate. The technique is unsuitable for detecting deep margin clearance (area where resection is most likely to be inadequate).
• Where bony resection is required extent is largely based on the clinical and radiological findings:
• extensive subcortical spread of tumour within the mandible is a relatively rare phenomenon, but will usually be suspected pre-operatively;
• prior to bone resection, a titanium reconstruction plate is fashioned to restore the contour of the resected bone (plate extended well beyond planned resection site in order to provide adequate fixation of the bone graft).
• For posterior oropharyngeal tumours it is important to visualize the great vessels of the neck up to the skull base and ensure that these are lateralized prior to resection of the primary tumour in order to avoid inadvertent damage.
• In rare instances where there is true tumour invasion of the major vessels, carotid resection is seldom warranted as the complications are severe, and neither palliation nor survival is enhanced.
• Following resection and prior to reconstruction, it is of vital importance that meticulous haemostasis is achieved:
• anaesthetist should be asked to restore the pre-operative blood pressure if hypotension has deliberately been employed;
• patient should be tipped head down to enhance identification of potential bleeding.
Photodynamic therapy
• Advocated as a technique that causes selective tumour destruction by cell apoptosis with minimal scarring and preservation of uninvolved tissue (thereby minimizing functional deficit).
• Photosensitizing agents currently available are insufficiently selective to prevent normal tissue damage.
• Patients must be protected from exposure to sunlight for several days.
• Following tumour ablation, the wound sloughs and heals by secondary intention and scarring.
• There does not appear to be any benefit of this technique over the more traditional resection methods.
The neck
• Several classifications of neck dissection (ND) are in existence.
• The term radical neck dissection should only apply to the classical ND described by Crile, which involves resection of the lymph nodes in level I–V of the neck together with sacrifice of:
• sternocleidomastoid muscle (SCM);
• spinal accessory nerve (SAN);
• internal jugular vein (IJV).
• All other neck dissections are selective and best described by the levels of lymph nodes resected, and which of the vital structures have been sacrificed, e.g. level I–IV with resection of IJV. This avoids confusion regarding the meaning of terms such as modified radical, functional, comprehensive, supra-omohyoid, and extended, which are all open to interpretation and lack clarity.
• Whether elective (staging ND in cN0) or therapeutic (ND in clinically or radiologically N+ disease) most neck dissections performed today preserve the vital structures of the neck to minimize functional deficit. The following structures are preserved unless they are directly invaded by tumour:
• SCM;
• carotid artery;
• IJV;
• SAN;
• laryngeal nerves;
• sympathetic chain;
• phrenic nerve;
• cervical plexus;
• hypoglossal nerve;
• mandibular branch of the facial nerve.
Neck access
There are four main types of incision described to access the neck:
• Apron.
• Wine glass.
• ‘H’.
• MacFee.
In most situations some form of the apron incision is the most appropriate. This should be raised in the sub-platysmal plane unless tumour invasion dictates otherwise. The subcutaneous blood supply is derived from a plexus of vessels with a vertical axis and therefore the MacFee (parallel transverse) incision, although preserving a bi-pedicle skin flap, does not make best use of the skin blood supply and produces poorer access to the neck. The ‘H’ and wine glass incision both give excellent access, but create points with reduced blood supply, which often lie over underlying vital structures. Where a previous incision has been made in the neck this should normally be excised and the neck incision planned around this.
Elective neck dissection (END)
• Where there is no clinical or radiological evidence of nodal involvement, END may be indicated because up to 30% of patients with tumours of the floor of mouth or tongue will have occult micrometastases.
• Access to the neck is also often required in order to facilitate microvascular reconstruction—under either of these circumstances a neck dissection will be indicated unless the patient is not fit enough for major surgery. When a simple local resection and ‘watch and wait policy’ can be employed.
• At present there is insufficient evidence to know whether any survival advantage is gained in performing END and what detriment in may have in terms of quality of life. However, it is hope that the ongoing UK-wide clinical trial ‘selective elective neck dissection’ (SEND) trial will address this issue. This is a prospective RCT comparing survival in patients who undergo either an END, or a ‘watch and wait policy’ for early carcinoma of the tongue. Study start date January 2007 with estimated primary completion in December 2015.
• Further benefit arising from END is accurate pathological staging of the neck that helps inform the need for adjuvant radiotherapy and increases prognostic accuracy.
• The extent of END determined by the site and size of the primary tumour, e.g. in anterior tumours:
• neck dissection should harvest levels I–IV (accounts for the possibility of skip lesions in level IV—particular phenomenon associated with primary tumours of the tongue and floor of mouth);
• incidence of spread to level IIb (above the spinal accessory nerve) is very low—therefore only included in large, posterior tumours;
• incidence of spread to level V in N0 neck is very low (1–3%)—therefore, level V not usually removed because morbidity to shoulder arising from devascularization of the spinal accessory nerve is high.
• If metastases are detected pathologically in levels I–IV (more than one positive node or any positive node with extracapsular spotted) then post-operative adjuvant radiotherapy is usually employed to include level V.
Sentinel node biopsy
• Advocated by some for the N0 neck.
• Injection of radiolabelled dye 1 day pre-operatively of the primary tumour area with intra-operative blue dye injected at the time of surgery.
• This then drains to the lymph nodes in the same order that tumour would spread.
• Gamma camera then used to determine the pattern of lymph node drainage for that tumour.
• Small incision made in the neck and the dye-bearing node(s) with high gamma signal excised and submitted for histology.
• If metastasis subsequently detected pathologically, formal neck dissection is undertaken at a second operation.
• Application of technique to oropharyngeal malignancies still under evaluation.
Therapeutic neck dissection
• Where the neck is N+.
• Where there is extensive disease in the neck with invasion of the SCM, SAN, and IJV, a radical ND is justified. On occasion it is necessary to extend the dissection beyond the boundaries defined by Crile (extended radical neck dissection (RND)).
• In other N+ cases a selective ND is indicated with preservation of as many vital structures as possible. In the majority of cases clear tissue planes between the vital structures and involved nodes are preserved, which means that some preservation of function is feasible.
• Occasionally it is not possible to safely resect involved nodes from a vital structure and, at that point, the ND should include the involved structure. This should be tailored to each individual patient. Whilst pre-operative imaging may give some idea about the likelihood of involvement, it is not infallible and often the decision will have to be made at operation.
• If this inclusion involves SCM, IJV, and SAN, and levels I–V, the ND is said to be ‘radicalized’.
• Care should be taken when undertaking the neck dissection to avoid unnecessary sacrifice of vessels that may have potential value for reconstruction—this should never take precedence, risking compromise of tumour resection.
Tracheostomy
The majority of patients who undergo resection of oropharyngeal tumours and ND do not require an elective tracheostomy provided that a level 3 critical care facility is available for the management of the patient in the immediate post-operative period. Where there has been a large oropharyngeal resection it is prudent to undertake a naso-endoscopic assessment of the airway prior to extubation. Occasionally, where the anaesthetist has assessed the intubation as difficult, it may be prudent to undertake an elective tracheostomy. Some units undertake elective tracheostomy where a bilateral ND or a large posterior resection has been carried out.
Where patients are taken back to theatre for re-exploration of the neck for flap insufficiency or bleeding it is usually wise to undertake a tracheostomy at that stage as swelling is likely to be much more significant (see ![]() Anaesthesia, p. 376).
Anaesthesia, p. 376).
Reconstruction
There have been major advances in this area over the last 3 decades. Previously, techniques available to repair defects following cancer ablation were limited. Patients were left deformed with little function. The use of free tissue transfer had transformed cancer treatment by extending what can be safely resected and with the resulting reconstruction, improved quality of life.
The oral cavity and oropharynx has many important functional roles. Resection of any tumour in this region may impact on function. The key functions to preserve with reconstruction of oropharyngeal cancer defects include:
• Speech.
• Swallowing.
• Eating.
• Chewing.
• Sensation.
• Cosmesis.
Reconstruction: issues to consider
The choice of reconstruction technique will depend on:
• Defect: site and size.
• Type(s) of tissue required.
• Patient factors:
• general fitness;
• existing medical problems;
• suitability of donor site;
• patient’s preference.
• Resources available.
General principles of reconstruction
• Replace like with like.
• Keep it simple.
• Immediate reconstruction is better than delayed.
• Minimize donor site morbidity.
• Resection to achieve clear margins must not be compromised to enable easier reconstruction.
Reconstructive ladder
The techniques available for repair are usually described as a reconstructive ladder, with simple techniques at the bottom rung (less effort/expertise) and more complex ones higher up. The success of reconstruction may be compromised by bleeding, haematoma, breakdown of wound, and infection. A more complex treatment, such as free tissue transfer, is an all or none phenomenon—its failure will result in a persisting defect, donor site morbidity, and a weaker patient.
Reconstructive techniques
The techniques available are described from a simple to more complex order.
Open wound
A laser bed wound is left open and heals with good mucosa coverage and minimal contracture. Conventional non-laser wounds, if left open, will heal slowly, with a combination of contracture and re-epithelialization. The resultant scar is poor, deformed, and may have a deleterious affect function.
Primary closure
• Ideal for small defects where the edges of the wound can be advanced for closure, enabling healing by primary intention.
• No donor site morbidity with maximal preservation of function and cosmesis.
• Use limited to small defects with lax adjacent tissues to advance for closure. Excessive tension to achieve primary closure may result in distortion, dehiscence and widened scar.
Graft
This involves using a piece of tissue (e.g. mucosa, skin graft, or bone) removed from its original donor site and transferred to a recipient site where it gains a new blood supply from the wound bed. The graft is usually harvested from the same individual (autograft), but may be from another individual of same species (allograft) or even different species (xenograft). A graft cannot be used if the recipient bed is absent (through and through defect) or hostile (no prospect of revascularization).
• Mucosal grafts:
• can be harvested from buccal mucosa (primary closure) or hard palate (allowed to re-epithelialize);
• good match for mucosal repair;
• easy to harvest;
• healing of donor site may cause further distortion of the oral cavity.
• Split-thickness skin graft:
• epidermis and part of dermis taken with a manual or electric dermatome knife;
• donor site left to re-epithelialize;
• high success rate as little tissue thickness is required for re-vascularization, but has minimal intrinsic features and may contract;
• donor site (e.g. thigh, upper arm) can be sore, requires dressing for several weeks, and has a paler appearance in the long term.
• Full-thickness skin graft:
• Reliable, but the success rate is slightly less than partial thickness counterpart;
• better appearance than partial-thickness graft;
• donor site (pre- or post-auricular, supraclavicular, abdomen) closed primarily.
• Bone:
• cancellous blocks (rich in osteogenic material), cortico-cancellous blocks, or cortical pieces;
• long-established technique that provides rigid scaffolding (osteoconductive), whereby the bony structure is gradually replaced by new bone formation;
• variety of donor sites, e.g. mandible, iliac crest, rib, tibia, or calvarium;
• can be used for repairing bony defects, and to augment or reconstruct discontinuity;
• as non-vascularized requires immobilization and a healthy vascular bed to take (radiotherapy will result in subsequent loss);
• in addition to generic operative side effects/possible complications (bleeding, haematoma, dehiscence, infection, and scarring), each donor site will have some specific morbidity, e.g. anterior iliac crest (numbness side of leg and groin, aching, and deformity).
• Cartilage grafts:
• used in reconstruction of composite ear or nose defects;
• usually harvested from the patient and has a good success rate if covered due to its low metabolic requirements;
• can be harvested from nose, ear, and rib as a composite graft with skin/mucosa.
• Nerve and vein grafts:
• occasionally required and often taken locally, e.g. as part of flap harvest, great auricular nerve, or external jugular vein;
• fascial grafts provide a band of firm tissue (from temporalis area or fascial lata of thigh) for use as slings or craniofacial dural repair.
Flaps
A flap is a piece of tissue retaining its attached vascular supply transferred to repair a defect within reach. Unlike a graft it is not dependent on initial re-vascularization since its blood supply is maintained. This also enables a more substantial bulk of tissue to be used, providing structure form and volume.
The vascular supply of a flap can be random or axial in pattern. The random pattern flap relies on subcutaneous vasculature. This limits the design on the length of flap before distal necrosis occurs (usually base width equal to length, but can be longer with the rich blood supply in the head and neck region). An axial pattern with specific associated vessels means a larger and longer flap can be raised safely.
• Local flaps: there are many local flaps that can be used for defects around the face, oral cavity, and pharynx, as well as neck. Examples include:
• local intra-oral options—buccal mucosal advancement/rotational flaps, tongue flap, palatal mucosal flap, and buccal fat pad flap. Suitable for small adjacent defects. Ease of harvest of a local flap and its matching tissue characteristics is offset by its limitation of size. Too large a harvest can distort anatomy and compromise function;
• naso-labial flap—random finger of skin taken from the naso-labial fold (primary closure of donor site with good resultant cosmesis) is turned into the oral cavity through a created opening in the cheek and buccal mucosa. Can be used to repair small defects (buccal, alveolus, and floor of mouth). The inferiorly based pedicle is divided 2–3 weeks after gaining co-lateral vascularization from the bed of the defect. When used bilaterally can repair anterior floor of mouth while retaining good function of the tongue;
• Karapandzic flaps—this sensate flap closes lower lip defects of up to 60% by advancing adjacent tissue, while preserving the peri-oral vascular ring and nerves (blunt muscular dissection technique). Microstomia can be a problem. There are numerous other rotational/advancement flaps for lip defects.
• Regional flaps: temporalis flap—muscle flap based on deep temporal vessels, reflected over the zygomatic arch to reconstruct small defects in the upper cheek, pharynx, and palate. Limited by its arc of rotation. Has a role in facial reanimation.
• Distant flaps:
• pectoralis major myocutaneous flap (PMMF)—workhorse, pedicled non-microvascular option in reconstruction of medium to large oral defects. Provides large muscle and associated chest wall skin based on the pectoral branch of thoraco-acromial artery. Often raised as a myocutaneous (muscle and skin) or muscle-only flap, reflected over the clavicle into the neck, and can reach up to the zygomatic arch. Donor site is primarily closed. Reliable and can be raised without need to change position of patient on table for oral cancer ablation. Its bulk is both advantageous (protect neck vessels, good for big defects) and disadvantageous (too rigid for defects requiring a more pliable repair). The shoulder function can be impaired when combined with radical ND;
• delto-pectoral flap—two-stage fasciocutaneous flap medially based on 2–3 perforating branches of the internal mammary artery. Limited by its reach into only the lower half of oral cavity and face. When collateral vascularization is achieved at 2–3 weeks, the tubed pedicle is divided and repositioned. The donor site defect is skin grafted (split-thickness). Superseded by other options but worth considering as a fall back in rare circumstances.
• Free flap: involves harvesting a flap of soft tissue or bone with its associated vascular supply, detaching it and re-anastomosing with donor vessels in the neck using microsurgical techniques (loupes or microscope). Surgery is complex and technically challenging. However, the type of tissue available for repair is no longer restricted by its ability to reach the defect. Success rate is high (around 95%) in specialized units. Post-operative monitoring is essential as a haematoma or venous thrombosis can threaten the viability of the flap.
There are many flaps suitable for oral and oropharyngeal defects following cancer ablation.
Four common free flaps used
Radial forearm flap
This is a distal skin paddle of the forearm based on a pedicle of the radial artery, vena commitantes, and/or superficial subcutaneous vein (Fig. 2.11
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


