Chapter 18 Nasal obstruction and sleep- disordered breathing
1 FUNCTIONAL VALVULAR ANATOMY
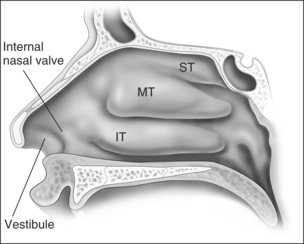
Anatomic models (Figs 18.1 and 18.2) in combination with objective measurements have shown that nasal airflow follows a parabolic curve directed superiorly through the nostril, upwards through the nasal cavity past the turbinates, and posteriorly to the nasopharynx during inspiration.1,2
The internal nasal valve area is the narrowest part of the nasal passage and is the major source of nasal resistance in normal patients. Four structures compose the internal nasal valve: the upper lateral cartilage superiorly, nasal septum medially, pyriform aperture inferiorly, and the head of the inferior turbinate posteriorly.3 The narrowest portion of the internal nasal valve area is the region between the septum and the caudal border of the upper lateral cartilage. This structure is approximately 10–15° in Caucasian subjects and can be wider in subjects of African and Asian descent. Patients with internal nasal valve angles of <10° are more prone to internal nasal valve collapse on inspiration.
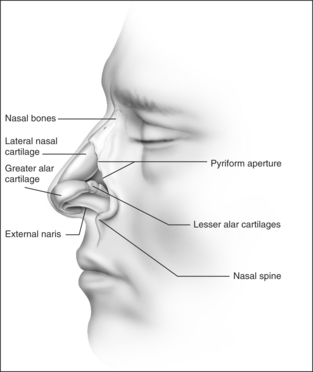
The external nasal valve is composed of the nares and the nasal vestibule. The nasal vestibule lies just inside the external naris and is located caudal to the internal nasal valve area. The septum is located medially along with the columella, and the alar sidewalls are lateral to the vestibule. Vibrissae are located within the vestibule under the lateral crus and function as a filter for the inspired air. They also serve to direct the air posteriorly into the nasal cavity and to slow the inspired air.4 The nares are composed of the alar margin, the soft tissue triangle, the columella and the nasal sill. The position of the medial crural footplates, the nasal spine and the caudal septum may all change the location and shape of the columella, thereby influencing the nares.
The internal and external nasal valves function together to deliver a smooth air current to the nasal cavities for humidification. The nasal valves can have a greater influence during deep inspiration when the nostrils flare and the diameter of the external nasal valve is increased. The Bernoulli principle is responsible for this effect in that the intraluminal pressure in the internal valve area decreases when the airflow is increased through it. The cartilaginous structure of the nose serves as a counterbalance to this tendency toward internal nasal valve collapse. The investing nasal musculature consists of elevator muscles, including the procerus, levator labii superioris alaeque nasi and anomalous nasi. The depressor muscles include the alar nasalis and the depressor septi nasi while the compressor muscles include the transverse nasalis and the compressor narium minor. There are also minor dilator muscles. The alar muscles contract and dilate the internal nasal valve area in order to keep the lumen open. Throughout normal nasal function, the internal nasal valve area should remain unchanged,5 although collapse demonstrable only during forced inspiration does not require intervention.6
The otolaryngologist should be aware of those characteristics that contribute to nasal obstruction. Those patients who have pre-existing or traumatic septal deviation can suffer nasal obstruction. Insufficient support of the alar rim and alar lobule may lead to external valve collapse on inspiration. Short nasal bones and a long upper cartilaginous vault; narrow, projecting nose; slit-like nostrils; exaggerated supra-alar creases; visible pinching of the lateral wall with inspiration; thin cartilages and skin; and cephalically positioned lateral crura which provide minimal support to the alar margins are all attributes that the surgeon should consider when planning surgical correction.7
2 NASAL OBSTRUCTION AND SDB
Case reports and case series as early as the 1890s showed an association between the nasal valve and sleep-disordered breathing (SDB). Today, we are all familiar with the fact that nasal obstruction and SDB often co-exist. Among patients referred to sleep clinics, studies have shown that subjective or objective nasal obstruction is a risk factor for SDB.2–4 Approximately 15% of patients with SDB also have nasal obstruction,8 but there is no correlation between the severity of obstructive sleep apnea and an objective measure of nasal resistance – acoustic rhinometry.9 Lofaso analyzed cephalometrics, body mass index and posterior rhinometry and showed that daytime nasal obstruction is an independent risk factor for obstructive sleep apnea.10 Others have explored the relationship between nasal obstruction and SDB showing that there is no association between SDB severity and the severity of the nasal obstruction.11–15
Beyond a simple association, however, nasal obstruction often plays a major role in SDB and its treatment. Nasal obstruction itself may contribute substantially to SDB by being an independent source of airway obstruction. The evidence supporting the role of nasal obstruction in SDB comes from a variety of sources. Normal patients have been shown to experience sleep disturbances – including sleep stage disruption as well as new-onset snoring and even mild obstructive sleep apnea – after acute, complete bilateral nasal obstruction such as abrupt occlusion of their nose.16 One study demonstrated that SDB was related to nasal cross-sectional area objectively assessed using acoustic rhinometry and both titrated nasal continuous positive airway pressure (CPAP) and the Respiratory Disturbance Index (RDI); however, these associations were only present in patients with normal Body Mass Index (BMI).17
Besides the simple effect of nasal obstruction on breathing patterns during sleep, the nose is a major conduit for treatment of obstructive sleep apnea with positive airway pressure therapy. Nasal obstruction can therefore interfere with treatment. Lafond and Series showed that nasal obstruction can increase the required continuous positive airway pressure in obstructive sleep apnea patients.18 They induced an increased nasal resistance with histamine and demonstrated that this increased flow limitation in two commonly used CPAP devices. Zozula and Rosen also showed that nasal obstruction can affect positive airway pressure therapy tolerance and adherence in their study examining and classifying reasons for CPAP non-compliance.19
2.1 INCREASED AIRWAY RESISTANCE
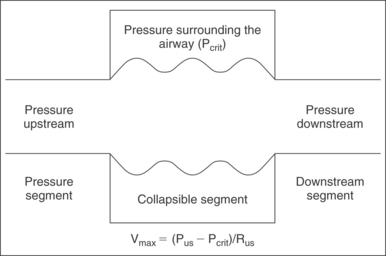
Applying the Starling model to the pharynx (Fig. 18.3), maximal airflow is based on three factors: upstream pressure, resistance in the upstream segment, and the extraluminal pressure. Increased nasal resistance is an increase in resistance of the upstream segment and therefore reduces flow through the collapsible tube, the pharynx. The maximal velocity of air through the nose is proportional to the pressure surrounding the tube (Pcrit) subtracted from the upstream pressure (Pus) and is inversely proportional to the resistance of the upstream segment (Rus).
< div class='tao-gold-member'>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses