One-electron reduction of H2O2
Fenton reaction
Fe2 + + H2O2 → Fe3+ + ·OH + OH−
Harber–Weiss reaction
O2 · − + H2O2 → (metal catalyst) → O2 + ·OH + OH−
Reaction with semiquinone
tetrachlorosemiquinone + H2O2 → tetrachloroquinone + ·OH + OH−
Homolytic fission
Radiolysis of water
H2O + γ − ray → H· + ·OH
Photolysis of H2O2
H2O2 + UV → 2·OH
Sonolysis of water
H2O + ultrasound → H· + ·OH
To control the yield of hydroxly radicals, the reaction of hydroxyl radical generation system should be terminated appropriately when necessary. From this viewpoint, reaction involving homolytic fission is advantageous because the reaction can be terminated by cessation of irradiation of γ-ray, ultraviolet ligh or ultrasound. Since hydroxyl radicals have a very short lifetime (approximately 10−9 s) [44, 45, 25], residual toxicity would be negligible after the termination of reaction. On the other hand, it might be difficult to control the reaction of one-electron reduction of H2O2 during the disinfection treatment. The chemicals involved in the reaction of homolytic fission are basically safe because they are only H2O and H2O2. Indeed, a subcommittee of the US Food and Drug Administration concluded that H2O2 as a disinfectant is safe at concentrations of up to 3 % [46]. Although H2O2 is also one of ROS, it is expected that hydroxyl radicals can kill bacteria more effectively than H2O2 because hydroxyl radicals have much higher reactivity and oxidative power [47]. Considering the safety aspect of reaction energy, exposure of normal tissue to γ-ray should be avoided. Thus, radiolysis of H2O cannot be applied to disinfection treatment in vivo. The irradiation of ultraviolet light (UV), which is electromagnetic wave with a wavelength of <400 nm, to normal tissue would also cause adverse effect especially when UV light with a short wavelength known as UVB (280–315 nm) and UVC (100–280 nm) is used [48]. Besides UV light, we have found that visible blue light (wavelength: around 400 nm) can also photolyze H2O2 and it will probably be used without adverse effects as long as the treatment time is not so long. As for ultrasound irradiation, ultrasound device with a frequency of <5 MHz has been utilized in medical diagnostic imaging. Thus, the frequency of ultrasound itself is supposed to be safe. Therefore, we have been studying photolysis of H2O2 and sonolysis of H2O to develop new disinfection techniques to treat infectious diseases.
17.2.1 Photolysis of H2O2
In dental and medical fields, the bactericidal activity of 3 % H2O2 is well recognized and used as a disinfectant. However, the bactericidal effect is not sufficient to treat periodontal diseases as discussed above. To improve the bactericidal activity, we studied the effect of visible blue light irradiation of H2O2 (i.e. photolysis of H2O2). Ikai et al. used a laser with a wavelength of 405 nm at an irradiance of 940 mW/cm2 and demonstrated that the yield of hydroxyl radicals generated by photolysis of H2O2 increased with laser irradiation time [49]. The laser irradiation of bacterial suspensions in 1 M H2O2 (corresponds to approximately 3 % H2O2) resulted in a >4-log reduction of the viable counts of bacteria, such as Staphylococcus aureus, Aggregatibacter actinomycetemcomitans, Streptococcus mutans and Enterococcus faecalis, within 3 min of treatment [49]. Furthermore, treatment of S. mutans in an experimental biofilm also resulted in a >5-log reduction of viable counts within 3 min [49]. Concerning the periodontal pathogens other than A. actinomycetemcomitans, Ikai et al. also investigated the bactericidal effect of photolysis of H2O2 on Porphyromonas gingivalis. It was demonstrated that photolysis of 500 mM H2O2 killed P. gingivalis in an experimental biofilm with a >5-log reduction of viable counts within 30 s [50]. As for H2O2 solution, Oyamada et al. compared the bactericidal activity of photoysis of oxydol products (2.5–3.0 % H2O2 solution), which is an over-the-counter drug with quality guaranteed by Japanese Pharmacopoeia, to substitute them for H2O2 of reagent grade [51]. It was demonstrated that any of the oxydol products that have been already approved by an authority can be used for the disinfection technique in terms of bactericidal activity. Thus, based on these findings, we have been developing a therapeutic device for the treatment of periodontitis, and a clinical trial will be conducted in the near future. In the therapeutic device, H2O2 is released from the forefront of scaler tip of the device to the lesion site concomitantly with laser irradiation through an optical fiber during ultrasound scaling (Fig. 17.1).
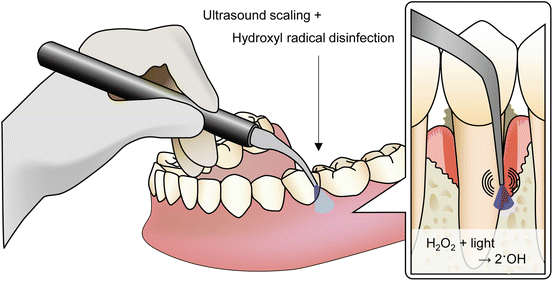

Fig. 17.1
Schematic illustration of the therapeutic device for the treatment of periodontitis. Mechanical removal of dental plaque by ultrasound scaling and chemical disinfection by hydroxyl radical generated by photolysis of 3 % H2O2 are performed at the same time
Nakamura et al. conducted a kinetic analysis and demonstrated that hydroxyl radicals generated by photolysis of H2O2 directly reacted with microorganisms [52]. In addition, it was demonstrated that the catalase activity of microorganisms influenced the microbial resistance to oxidative stress induced by photolysis of H2O2. Nonetheless, Candida albicans, a catalase positive yeast-like fungus whose catalase activity is more potent than that of catalase positive bacteria such as S. aureus could also be killed with a >4-log reduction of viable counts within 10 min when treated with the photolysis of 250 mM H2O2 using LEDs at an irradiance of 80 mW/cm2. This finding suggests that photolysis of H2O2 can effectively kill not only bacteria but also fungi. Shirato et al. evaluated the effect of thermal energy on the yield of and the bactericidal action of hydroxyl radicals generated by photolysis of H2O2 [53]. The results demonstrated that thermal energy accelerated the generation of hydroxyl radicals by photolysis of H2O2, which in turn resulted in a synergistic bactericidal effect of hydroxyl radicals and thermal energy. When photolysis of H2O2 was performed at 55 °C, S. aureus and E. faecalis were killed with a >5-log reduction of viable counts within 1 min even though the irradiance of light was 110 mW/cm2 and the concentration of H2O2 was 500 mM. This synergistic effect will be beneficial especially when it comes to cleaning of medical instruments or dental prostheses which can tolerate the temperature of 55 °C. Based on these findings, Kanno et al. applied photolysis of H2O2 to cleaning of removable dentures and conducted a clinical test [54]. They demonstrated that microorganisms in denture plaque were reduced by approximately 7-log within 20 min. In addition to the in vitro tests, in vivo antibacterial effect of photolysis of H2O2 was proven effective. Hayashi et al. evaluated antibacterial activity of photolysis of H2O2 on skin infection [55]. Infection of S. aureus was established in the full-thickness skin wounds of immunosuppressed rats. Then, the wound was treated by photolysis of 1 M H2O2. Two minutes treatment resulted in significant reduction in viable counts in comparison with the treatment by H2O2 alone. Thus, the disinfection technique based on photolysis of H2O2 is expected to be applied to a wide range of fields other than treatment for periodontitis.
Besides the bactericidal activity, it was demonstrated that photolysis of H2O2 caused lag of regrowth of the surviving bacteria after the disinfection treatment, known as the postantiboitic effect (PAE). Odashima et al. demonstrated that photolysis of 250 mM H2O2 with a treatment time of 10 s significantly delayed the regrowth of S. aureus colony on agar plate [56]. The PAE would be beneficial because it contributes to giving time for host immune defense system to overcome the infection. More importantly, it has been demonstrated that repeated treatment by photolysis of H2O2 does not induce bacterial resistance to this disinfection technique. Ikai et al. evaluated the risk of inducing bacterial resistance to disinfection treatment with photolysis of H2O2 using S. aureus, E. faecalis, Escherichia coli, Streptococcus salivarius, Pseudomonas aeruginosa, S. mutans, and A. actinomycetemcomitans [36]. The antibacterial activity against any of the bacterial species tested was not affected by repeated exposure to the disinfection treatment up to 40 times suggesting that the repeated treatment does not induce bacterial resistance.
Regarding the safety aspect of the disinfection system, Yamada et al. conducted an animal study in which histological analyses of oral mucosa and the full-thickness skin wounds in rats treated by the disinfection technique with photolysis of 1 M H2O2 were performed [57]. Since topical treatment had no detrimental effect on the oral mucosa and the healing process of full thickness skin wounds, it is expected that the acute locally injurious property of the disinfection technique is low. Moreover, Kanno et al. assessed the risk of carcinogenicity by using the hydroxyl radical for the treatment of oral infections by reviewing the literatures [58]. The reviewed studies reported possible involvement of hydroxyl radicals in some sort of chemically-induced mutagenicity and carcinogenicity. However, manifestation of carcinogenicity requires chronic exposure to the carcinogens that generate hydroxyl radicals, such as heavy metals. Thus, it was concluded that there is little or no risk of carcinogenicity as long as the hydroxyl radical is used as a disinfectant for the short-term treatment of oral cavity. Therefore, the disinfection treatment with photolysis of H2O2 is expected to be a novel alternative to antiseptics and antibiotics.
17.2.2 Sonolysis of Water
The antimicrobial effect of ultrasound has been studied in various fields, such as water treatment, food decontamination and medical disinfection [59]. The mechanism of killing microorganisms is mainly due to cavitation effect in which tiny gas bubbles formed in liquid medium by alternating compression and expansion collide violently, creating shock waves in a localized region with high temperature (5,500 °C) and pressure (50 MPa) [60]. When water is used as a liquid medium, the shock wave produced by the cavitation effect fragments the molecules of H2O into hydroxyl radicals and hydrogen atoms, which is so-called sonolysis of water [61]. Then, the localized heating and pressure together with the free radicals damage microorganisms resulting in cell death [60].
In this context, we examined fungicidal effect of ultrasound irradiation at a frequency of 1.6 MHz [62] to develop a treatment of onychomycosis (i.e. fungal nail infections). It was demonstrated that the ultrasound irradiation killed the dermatophytes, such as Trichophyton mentagrophytes and Trichophyton rubrum, with a yield of hydroxyl radicals that is proportion to ultrasound duration. In particular, when the treatment was performed at 50 °C, the fungi were killed with a 5-log of viable counts within 10 min. Thus, it is suggested that the ultrasound irradiation and heat treatment exerted a combination effect in fungicidal activity. This finding was in accordance with previous studies which evaluated the combination effect of heat and ultrasound on bactericidal activity [63, 64].
As for application of sonolysis of water to dentistry, other research groups have conducted studies. For instance, it has been demonstrated that ultrasound irradiation of 1.5 % (450 mM) H2O2 using a device for endodontic treatment generates hydroxyl radicals resulting in killing of E. faecalis [65]. However, the reduction of bacterial number within the treatment time of 90 s was less than 1-log. Thus, the clinical benefit of this treatment is still unclear. The other example is the ultrasonic scaler used for periodontal therapy. It has been reported that hydroxyl radicals are generated during scaling around ultrasonic scaler tip [66–68]. However, the role of hydroxyl radicals generated by ultrasonic scaler is still unknown.
The microbicidal activity of ultrasound irradiation varies dependently on the type of microorganisms treated (i.e. Gram-positive or negative bacteria, spore forming bacteria, and fungi). In addition, it has been reported that the microbicidal activity of ultrasound irradiation alone is relatively low (<1-log) and/or requires extended irradiation time (~60 min) unless it is used at high intensity [69]. Therefore, as suggested by Piyasena et al. [60], it would be beneficial to use ultrasound in conjunction with other disinfection techniques, such as heat treatment, antiseptics and antibiotics.
17.2.3 Other Hydroxyl Radical Generation Systems
More recently, we found that photo-irradiated polyphenolic compounds could generate hydroxyl radicals as well as H2O2. Since it has been reported that polyphenolic compounds exert antimicrobial effect via production of H2O2 by auto-oxidation in a liquid medium [70, 71], we tried to enhance the antimicrobial activity by means of exposure of polyphenolic compounds to blue light, which enhances the H2O2 production through photo-oxidation of the compounds resulting in hydroxyl radical production via photolysis of the resultant H2O2 (Fig. 17.2). Nakamura et al. demonstrated that hydroxyl radicals were generated when gallic acid, a polyphenolic compound, was irradiated with blue light (wavelength: 400 nm) at an irradiance of 80 mW/cm2 [72]. It was also demonstrated that S. aureus was killed by the photo-irradiation of gallic acid with a >5-log reduction of viable counts within 15 min. Since the bactericidal activity was attenuated by adding hydroxyl radical scavengers, such as dimethyl sulfoxide and sodium formate, it is suggested that the hydroxyl radical is the main contributor to the bactericidal effect. Later, they showed similar bactericidal activity of photo-irradiated proanthocyanidin which is also a type of polyphenolic compounds [73]. The advantage of this disinfection technique is that polyphenols are supposed to be safe for humans because they are edible compounds naturally occurring in fruits, vegetables, nuts, seeds, and flowers. In addition, since those polyphenolic compounds are noteworthy for its antioxidative activity [74, 75], they might alleviate the oxidative damage in the tissue after the disinfection treatment. Therefore, it is expected that this disinfection technique is applicable to the fields of medical and food sanitation.
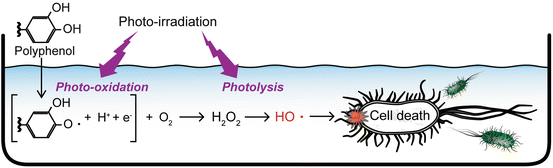

Fig. 17.2
Schematic illustration of the mechanism of bacterial cell death induced by photo-irradiation of polyphenolic compound
Based on the findings described above, we studied the combination effect of photolysis of H2O2 and photo-irradiation of proanthocyanidin. Since both disinfection techniques utilize the same light source (wavelength: around 400 nm), they can be performed at once if the liquid contains H2O2 and proanthocyanidin. Ikai et al. demonstrated that combination of H2O2 and proanthocyanidin worked synergistically to kill S. mutans when photo-irradiated [76]. This synergistic effect would probably contribute to shortening the treatment time and/or to reducing the concentration of H2O2 for more safety.
17.3 Summary
In the present review, we described our recent works on microbicidal effect of the artificially generated hydroxyl radicals. As discussed above, the microbicidal activity of hydroxyl radicals is sufficient to kill bacteria in an experimental biofilm as well as planktonic bacteria and fungi within a short-treatment time. At the same time, the safety aspect is also confirmed by an in vivo study and a literature review. Therefore, they can be applicable as novel disinfection treatments in medical/dental therapy. Further studies are in progress to understand the bactericidal mechanism of each disinfection system and also of a combination of each system for the more sophisticated medical devices.
Open Access This chapter is distributed under the terms of the Creative Commons Attribution Noncommercial License, which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
1.
Marsh PD, Nyvad B. The oral microflora and biofilms on teeth. In: Fejerskov O, Kidd E, editors. Dental caries. The disease and its clinical management. Oxford: Blackwell Munksgaard; 2003. p. 49–70.
2.
Socransky SS, Haffajee AD. Microbiology of periodontal disease. In: Lindhe J, Karring T, Lang NP, editors. Clinical periodontology and implant dentistry. 4th ed. Oxford: Blackwell; 2003. p. 106–49.
3.
Costerton JW, Lewandowski Z, Caldwell DE, Korber DR, Lappin-Scott HM. Microbial biofilms. Annu Rev Microbiol. 1995;49:711–45. doi:10.1146/annurev.mi.49.100195.003431.PubMedCrossRef
4.
Cargill KL, Pyle BH, Sauer RL, McFeters GA. Effects of culture conditions and biofilm formation on the iodine susceptibility of Legionella pneumophila. Can J Microbiol. 1992;38(5):423–9. doi:10.1139/m92-071.PubMedCrossRef
5.
Russell AD. Antibiotic and biocide resistance in bacteria: introduction. J Appl Microbiol. 2002;92(Suppl):1S–3. doi:10.1046/j.1365-2672.92.5s1.14.x.PubMedCrossRef
6.
Tenover FC. Mechanisms of antimicrobial resistance in bacteria. Am J Med. 2006;119:S3–S10. doi:10.1016/j.amjmed.2006.03.011.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


