Introduction
The objectives of this study were (1) to investigate the expressions of interleukin (IL)-17, RANKL (the receptor activator of NF-kappaB ligand), and osteoprotegerin (OPG) in root resorption areas during experimental tooth movement in rats, and (2) to determine the effect of IL-17 on the expressions of RANKL and OPG mRNA from human dental pulp cells.
Methods
Twelve male 6-week-old Wistar rats were subjected to an orthodontic force of 50 g to induce a mesially tipping movement of the maxillary first molars for 7 days. The expression levels of tartrate resistant acid phosphatase (TRAP), interleukin (IL)-17, IL-17 receptor (IL-17R), receptor activator of nuclear factor-kappa B ligand (RANKL), and OPG proteins were determined in dental pulp by immunohistochemical analysis. Furthermore, the effects of IL-17 on the expressions of RANKL and OPG mRNA were investigated using human dental pulp cells in vitro.
Results
In the experimental tooth movements in vivo, resorption lacunae with multinucleated cells were observed in the 50-g group. The immunoreactivities for IL-17, IL-17R, and RANKL were detected in dental pulp tissues subjected to the orthodontic force on day 7. Moreover, IL-17 increased the mRNA expression of RANKL from human dental pulp cells in vitro.
Conclusions
The results of this study suggest that IL-17 and RANKL may be involved in the process of orthodontically induced inflammatory root resorption in dental pulp cells.
Highlights
- •
Multinucleated cells were observed in the 50-g group in experimental tooth movements in rats.
- •
IL-17, IL-17R, and RANKL were found in dental pulp subjected to orthodontic force on day 7.
- •
IL-17 increased mRNA expression of RANKL and the ratio of RANKL/OPG from human dental pulp cells.
Orthodontically induced inflammatory root resorption (OIIRR) is a common complication associated with orthodontic tooth movement. This is an undesirable consequence that can cause permanent loss in the dental structure of the root apex. The pathogenesis is associated with the removal of necrotic tissue from the areas of the periodontal ligament (PDL) that have been compressed by an orthodontic load.
The RANKL/RANK/osteoprotegerin (OPG) system plays a critical role in inducing bone remodeling. The tumor necrosis factor-alpha-related ligand RANKL (receptor activator of nuclear factor-kappa B ligand) and its 2 receptors, RANK and OPG, have been shown to be involved in the bone remodeling process. RANKL is a downstream regulator of osteoclast formation and activation through which many hormones and cytokines produce their osteoresorptive effects. In the bone system, RANKL is expressed on the osteoblast cell lineage, and it exerts its effect by binding to the RANK receptor on osteoclast lineage cells. This binding leads to rapid differentiation of hematopoietic osteoclast precursors to mature osteoclasts. OPG is a decoy receptor produced by osteoblastic cells, which compete with RANK for RANKL binding. The biologic effects of OPG on bone cells include inhibition of the terminal stages of osteoclast differentiation, suppression of activation of matrix osteoclasts, and induction of apoptosis. Thus, bone remodeling is controlled by a balance between RANK and RANKL binding and OPG production.
Interleukin (IL)-17, initially referred to as cytotoxic T lymphocyte-associated antigen-8, is an inflammatory cytokine that is produced exclusively by activated T cells. IL-17 has been shown to be an important mediator of autoimmune diseases, including rheumatoid arthritis. IL-17 induces RANKL production by osteoblasts and was shown to be related to bone destruction in periodontitis. Moreover, Zhang et al reported that compressive force enhanced the expression of the IL-17 genes and their receptors in MC3T3-E1 cells, and also resulted in the induction of osteoclastogenesis. In addition, Hayashi et al reported that IL-17 induced by excessive orthodontic force stimulates odontoclastogenesis through IL-6 production in PDL tissues. Yamaguchi et al reported that the T-helper 17 cell response to excessive orthodontic force led to the progression of root resorption by increasing the expression of IL-17, RANKL, and RANK. Therefore, IL-17 in PDL tissues may contribute to OIRR during orthodontic tooth movement.
Considering the relationship between dental pulp and OIIRR, Yamaguchi and coworkers reported that IL-1 beta, IL-6, tumor necrosis factor-alpha, and RANKL were produced by substance P (inflammatory neuropeptide) released from human dental pulp cells in response to tooth movement. Previous reports have stated that human dental pulp cells are deeply involved in the progress of inflammation by orthodontic tooth movement and the incidence of OIIRR during orthodontic treatment.
However, little is known about the relationship among OIIRR, IL-17, and RANKL in dental pulp. In this study, the immunolocalization of IL-17, IL-17R, and RANKL was investigated in root resorption during experimental tooth movements in rats with the application of a heavy force (50 g) using an immunohistochemical analysis. Furthermore, the effects of IL-17 on the expression of RANKL mRNA were investigated using human dental pulp cells in vitro.
Material and methods
The animal experimental protocol in this study was approved by the ethics committee for animal experiments at Nihon University School of Dentistry at Matsudo in Japan (approval number AP11MD015). Twelve male (control, 6; orthodontic force, 6) Wistar rats (Sankyo Laboratory, Tokyo, Japan; age, 6 weeks; body weight, 180 ± 10 g) were used for the experiments. Experimental tooth movement was induced using the method reported by Asano et al. The period of the experiment was 7 days.
Tissue preparation and the immunohistochemistry were performed according to the method of Shimizu et al. The experimental period was set at 7 days after tooth movements were initiated. Each sample was sliced into 4-μm continuous sections in the sagittal direction and then prepared for hematoxylin and eosin staining and also for immunohistochemical staining. Dental pulp tissues in the distobuccal roots of the maxillary first molars were observed.
The antibodies for immunohistochemical staining were used as follows: (1) polyclonal anti-rabbit tartrate resistant acid phosphatase (TRAP) (Santa Cruz Biotechnology, Dallas, Tex; working dilution, 1:100), (2) polyclonal anti-rabbit IL-17 (Biolegend, San Diego, Calif; working dilution, 1:200), (3) polyclonal anti-rabbit IL-17R (Santa Cruz Biotechnology; working dilution, 1:200), and (4) polyclonal anti-goat RANKL and OPG (Santa Cruz Biotechnology; working dilution, 1:100). TRAP, IL-17, IL-17R, RANKL, and OPG were stained using the Histofine Simple Stain MAX-Po (G) and (R) kit (Nichirei, Tokyo, Japan) according to the manufacturer’s protocol. The Histofine Simple Stain MAX-Po (G) kit was used for TRAP, RANKL, and OPG, and the MAX-Po (R) kit was used for IL-17 and IL-17R.
Human dental pulp cells were prepared according to a modification of the method reported by Hosoya et al. Briefly, after obtaining informed consent from the donors, human dental pulp tissues were taken from the roots of the premolars extracted from 6 healthy young volunteers (3 boys, 3 girls; 14-16 years of age) during their orthodontic treatment and were used according to a protocol reviewed by the ethics committee of Nihon University School of Dentistry at Matsudo (number EC 11-022). For all experiments, human dental pulp cells were used at passages 6 through 9.
To examine the effects of recombinant human (rh)IL-17 (R&D Systems, Minneapolis, Minn) on the production of RANKL, human dental pulp cells were seeded onto 6-well dishes at a density of 1 × 10 6 cells per square centimeter. The cells were then cultured for up to 72 hours in α-MEM containing 1% fetal calf serum with 0, 0.1, 1.0, 10, or 100 ng per milliliter of rhIL-17.
Real-time polymerase chain reactions were performed with the method of Shimizu et al. Total RNA was isolated from cultured human dental pulp cells using a commercially available kit (RNeasy Mini kit; Qiagen, Tokyo, Japan) following the manufacturer’s protocol. Aliquots containing equal amounts of mRNA were subjected to real-time polymerase chain reactions. The primers used were as follows.
- 1.
RANKL: 5′-ATCGTTGGATCACAGCACATCAG-3′ and 5′-GGATGTCGGTGGCATTAATAGTGAG-3′
- 2.
OPG: 5′-AGCTGCAGTACGTCAAGCAGGA-3′ and 5′-TTTGCAAACTGATTTCGCTCTGG-3′
- 3.
GAPDH: 5′-GCACCGTCAAGGCTGAGAAC-3′ and 5′-TGGTGAAGACGCCAGTGGA-3′
The data were analyzed using the Smart Cycler software program (Multiplate RQ version 1.00; TaKaRa, Shiga, Japan), and the levels of the real-time polymerase chain reaction products corresponding to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) were confirmed to be the same in the experimental groups. Therefore, it was considered acceptable to assume that the amount of real-time polymerase chain reaction products reflected the level of each mRNA.
Statistical analysis
The values in each figure represent the means and standard deviations for each group. The Mann-Whitney U test was used to compare the means of the groups. Furthermore, 1-way analysis of variance tests were performed, with values of P <0.01 considered to indicate a significant difference.
Results
The body weights of the rats in both force groups decreased transiently on day 1 and then recovered. No significant differences between the 2 groups were observed (data not shown).
There were histologic changes in the periodontal tissues during tooth movement with the hematoxylin and eosin staining. In the control group (0 g), the rat dental pulp specimens were composed of relatively dense connective tissue fibers and fibroblasts that were horizontally aligned from the root dentin. The root surface was relatively smooth ( Fig 1 , A ). In the 50-g group, many root resorption lacunae with multinucleated odontoclasts were seen on the surface of the root on day 7 ( Fig 1 , B ).
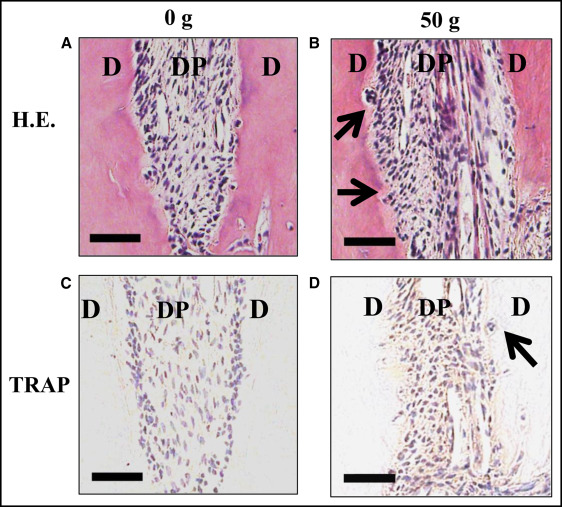
In the control group, resorption lacunae with TRAP-positive multinucleated osteoclasts were not observed on the surfaces of the root ( Fig 1 , C ). In the 50-g group, many resorption lacunae with multinucleated TRAP-positive odontoclasts were observed on the surface of the root on day 7 ( Fig 1 , D ).
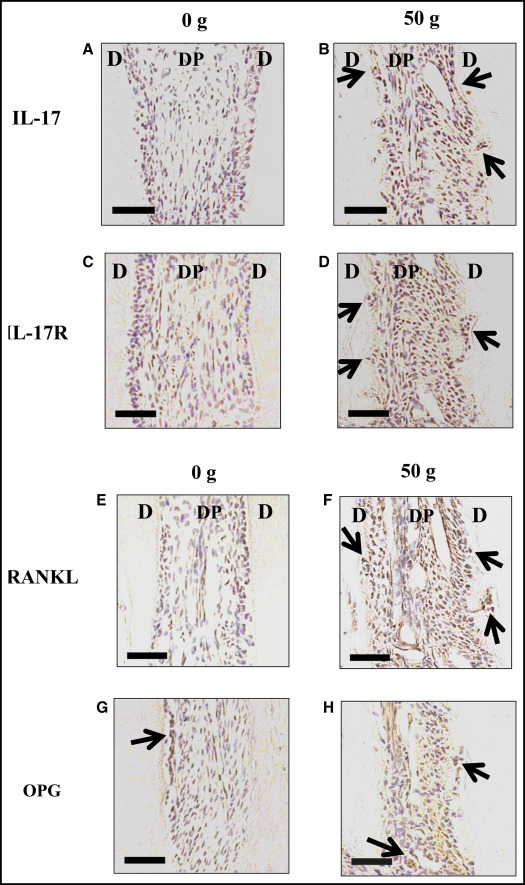
The protein expression levels of IL-17, IL-17R, RANKL, and OPG were tested.
The immunoreactivity tests of IL-17, IL-17R, and RANKL were performed on day 7 after tooth movement. IL-17-, IL-17R-, and RANKL-positive cells were rarely observed in the control group ( Fig 2 , A , C , and E ). In the 50-g group, many IL-17- and IL-17R-positive cells were observed ( Fig 2 , B and D ). Many RANKL-positive cells were observed on 7 day ( Fig 2 , F ). OPG-positive cells were observed in the control group and were increased in the 50-g group.

Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


