The initial assessment and intensive care treatment of the injured patient are extremely broad topics. Complete textbooks by renowned traumatologists and intensivists have already been written on each of these topics. Our goal in producing this chapter is to give a broad overview that may serve as a reference to oral surgeons who may encounter trauma and intensive care treatment as part of their practice. As with any field, the arenas of trauma and critical care continue to expand; as such, our focus is on general principles and concepts rather than specific techniques and practices. The selected readings list can be used to delve into topics in more detail.
INITIAL ASSESSMENT OF THE INJURED PATIENT
Prior to the development of the Advanced Trauma Life Support (ATLS) course in 1978, delivering care to injured patients was inconsistent at best. The traditional tenets of detailed history-taking, thorough physical examination, diagnostic testing, and formation of a detailed differential diagnosis are not applicable to the care of the injured patient. Instead, rapid identification and correction of life-threatening injuries must be paramount in the treatment of trauma patients. The ATLS process emphasizes a systematic approach to the evaluation and treatment of the injured patient, focusing on the following steps:
- 1.
Primary survey—identification and treatment of life-threatening injuries
- 2.
Resuscitation and cessation of ongoing hemorrhage
- 3.
Secondary survey—head-to-toe evaluation of the patient
- 4.
Definitive care, including operative and intensive care
PRIMARY SURVEY
In the severely injured patient, a rapid, systematic evaluation prioritizes those injuries most likely to cause harm. The primary survey is used to identify and treat all immediately life-threatening injuries. Since this task can be overwhelming in the case of the multiply injured patient, an “ABCDE” mnemonic has been developed and is shown in Table 4-1 .
| A | Airway maintenance while maintaining adequate cervical spine control |
| B | Breathing and adequate oxygenation and ventilation |
| C | Circulation and control of ongoing hemorrhage |
| D | Disability assessment and neurologic evaluation |
| E | Exposure of the patient and environmental control |
Following this algorithm can keep a care provider from being distracted by more obvious, but perhaps not life-threatening, injuries.
Airway
The first priority in evaluating an injured patient is to ensure that the airway is patent and can be maintained. While assessing the patient’s airway, take care to maintain cervical spine immobilization. There have been many documented cases of severe cervical spine injury presenting with an intact neurological examination; therefore, all injured patients should be presumed to have a spine injury until proven otherwise. If appropriate immobilization devices need to be removed, a member of the trauma team should maintain the patient’s head and neck in neutral position using inline stabilization techniques, avoiding hyperextension or flexion.
Airway obstruction can be caused by foreign body, aspiration, tongue position, facial fractures, edema, expanding hematoma, or tracheal/laryngeal injuries. Simple maneuvers, such as placement of airway devices (both nasal and oral), jaw thrust, or chin lift, may establish an airway temporarily. However, establishment of a definitive airway, in the form of a secured tube placed in the trachea through which adequate oxygenation and ventilation can be achieved, should be considered in some patients. Patients with impending respiratory failure—those who have significant tachypnea or who are unable to maintain adequate oxygenation or ventilation—may require early placement of a definitive airway. Likewise, patients who are unable to protect the airway with adequate cough and gag reflexes need a definitive airway placed early in the evaluation. Bag-valve mask support of the spontaneously breathing patient may be necessary while preparations for a definitive airway are made. Both nasotracheal and orotracheal tubes can be used. Nasotracheal tubes are generally better tolerated in the awake patient but are smaller in diameter and require spontaneous respiratory effort on the part of the patient for placement. Evidence of midface trauma is a contraindication to nasotracheal tube use. Orotracheal tubes may require the administration of paralytics for placement. Prior to inducing paralysis, adequate means of oxygenation and ventilation must be established in the event that definitive tube placement is unsuccessful. Direct visualization of the airway with bronchoscopy may be useful in the placement of a difficult airway, and placement of the definitive airway over an Eschmann obturator or Cook airway exchange catheter may be useful in some situations. Adequate oxygenation and ventilation must be maintained during intubation or use of airway adjuncts. Inability to do so, particularly after several unsuccessful intubation attempts, should prompt the placement of a surgical airway, such as cricothyroidotomy.
Breathing
Once an adequate airway is established, the patient can be evaluated for oxygenation and ventilation. Visual inspection can determine if movement of the chest wall is symmetric. Paradoxical movement of the thoracic cage, where a portion of the chest wall becomes depressed during inspiration, can indicate a flail segment with rib fractures in more than one location. In such cases, significant underlying pulmonary contusion may contribute to inadequate gas exchange. Point tenderness or bony crepitans often indicates rib fractures, and the presence of subcutaneous emphysema should raise the index of suspicion for pneumothorax or, in cases of massive subcutaneous emphysema, major bronchial injury. Tracheal deviation may be a late sign of tension pneumothorax. Loss of breath sounds may indicate pneumothorax, hemothorax, or inappropriate endotracheal tube placement.
It is no coincidence that airway and breathing are closely tied together, and diagnostic separation of the two is often difficult. Most of the immediately life-threatening injuries identified in the primary survey occur in these two areas. The presence of a pneumothorax can jeopardize airway and breathing. A pneumothorax results from the violation of the visceral pleura, allowing air to escape into the pleural space. The resultant collapse of the lung parenchyma often results in tachypnea or low oxygen saturation ( Figure 4-1 ).
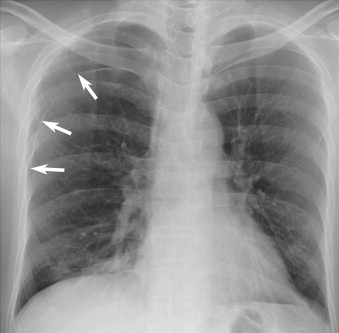
A tension pneumothorax is more critical. It is caused when air entering the pleural cavity is unable to escape, and the amount of air entrapped in the pleural space increases with each subsequent inspiration. The resultant shift of the mediastinum can cause obstruction of venous return through the superior and inferior vena cava ( Figure 4-2 ).
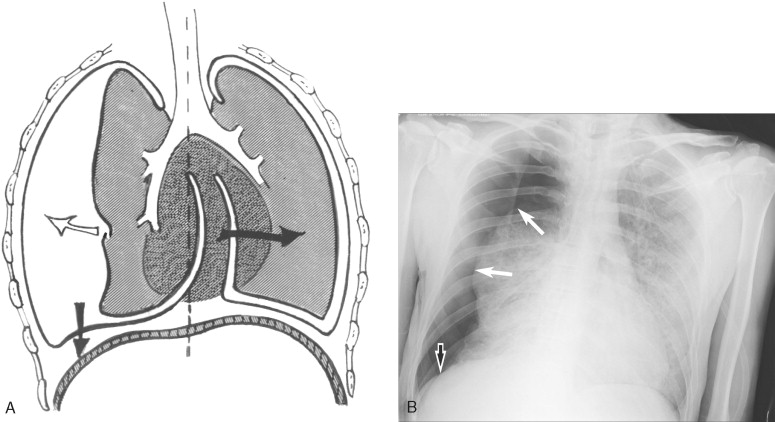
As this obstruction reduces filling of the heart, hypotension results and rapidly leads to cardiac arrest if untreated. Thus, the diagnosis of tension pneumothorax is only rarely made with radiographic examination. An injured patient with absent or decreased breath sounds along with hypotension and impending arrest should empirically be treated with needle decompression of the ipsilateral hemithorax. Successful needle decompression by insertion of a 14-gauge IV catheter into the second intercostal space in the mid-clavicular line is accompanied by a satisfying rush of air, and it converts the tension pneumothorax to a simple pneumothorax, which can be treated with tube thoracostomy after the primary survey is complete ( Figure 4-3 ).
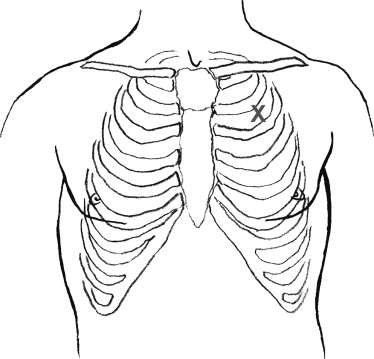
It is important to note that the application of positive pressure ventilation to a simple pneumothorax may convert it to a tension pneumothorax. To prevent the reaccumulation of a tension pneumothorax, take care that the catheter does not become dislodged or occluded after needle decompression. Another significant pneumothorax is the open pneumothorax, which presents as a sucking chest wound, in which air enters and exits the chest cavity through a penetration or laceration of the chest wall. This condition is treated by applying a sterile, occlusive dressing that is only secured on three sides. The dressing acts as a one-way valve, allowing air in the pleural cavity to escape while preventing air from reentering. Again, this maneuver converts the open pneumothorax to a simple pneumothorax, which will eventually require tube thoracostomy for evacuation. In cases of open pneumothorax, operative intervention to close the chest wall defect is often necessary as well.
Decreased or absent breath sounds should also raise the suspicion for significant hemothorax. Since a significant percentage of the patient’s blood volume can be contained in a single hemithorax, massive hemothorax often presents with signs of hypovolemic shock as well as oxygenation and ventilation deficits. Placement of tube thoracostomy can alleviate any collapse of the pulmonary parenchyma if the hemothorax is associated with pneumothorax and can allow simultaneous quantification of ongoing blood loss.
Circulation
Once adequate oxygenation and ventilation are established, the hemodynamic status of the patient should be assessed. Tachycardia and hypotension should raise the suspicion for significant blood loss. As a rule, palpable pulses in the radial, femoral, and carotid regions indicate systolic pressures of 80, 70, and 60 mm of Hg, respectively. Any obvious sources of hemorrhage, particularly in the extremities or scalp, can be temporarily controlled with direct pressure. Other, less obvious, sites of blood loss include the thorax, abdomen, retroperitoneum, pelvis, and long bone injuries, particularly femur fractures. Two 16-gauge or larger peripheral intravenous lines should be placed for volume administration and resuscitation. The four classes of hemorrhagic shock are outlined in Table 4-2 .
| Class I | Class II | Class III | Class IV | |
|---|---|---|---|---|
| Estimated blood loss (% blood volume) | <15% | 15-30% | 30-40% | >40% |
| Pulse | normal | ↑ | ↑↑ | ↑↑↑ |
| Blood pressure | normal | normal | ↓ | ↓↓↓ |
| Urine output | normal | ↓ | ↓↓ | absent |
| Mental status | normal | agitated | confused | obtunded |
* Up to 15% blood volume loss shows little or no clinical manifestation. By the time hypotension is detected, as much as one third of the circulating blood volume has been lost.
It is important to note that loss of up to 15% of a patient’s blood volume occurs without changes in pulse, blood pressure, or urine output. Documented hypotension (Class III shock) is associated with a loss of over 30% of the patient’s blood volume and must be identified and treated expeditiously. Identifying the patient in class II shock may be difficult, since the findings of mild tachycardia and anxiety may be explained by pain, catecholamine release, drugs, or other intoxications. In addition, the young athletic patient or the elderly patient receiving beta adrenergic blocking agents may not manifest the classic signs of tachycardia with hypovolemic shock. In these cases, monitoring of urine output can aid in determining the degree of hypovolemia. The kidneys, which normally receive 20-25% of cardiac output, are a highly regulated vascular bed. Overall intravascular volume depletion causes decreased renal blood flow and decreased urine output. Minimal urine production in the adult is roughly 0.5 mL/kg/h, and adequate volume resuscitation will rapidly be reflected in the urinary output production. Resuscitation is initiated by administering 2 L of crystalloid fluid, preferably lactated Ringer’s solution, after which blood products are infused. Clinical improvements in tachycardia, blood pressure, mental status, and urine output can be used as signs of adequate resuscitation when more intensive monitoring is not available. Lack of improvement, or transient improvement, in these parameters suggests ongoing hemorrhage.
In general, significant blood loss can occur externally or in one of five compartments: thorax, abdomen, retroperitoneum, pelvis, or thighs. The hemodynamically unstable patient should be evaluated for blood loss into each of these regions by physical examination or other diagnostic studies. Whereas spinal cord injury can cause loss of sympathetic tone with resultant hypotension, neurogenic shock is characterized by hypotension associated with bradycardia rather than tachycardia. The patient who presents with obvious cord injury, tachycardia, and hypotension has an unidentified source of blood loss that must be discovered.
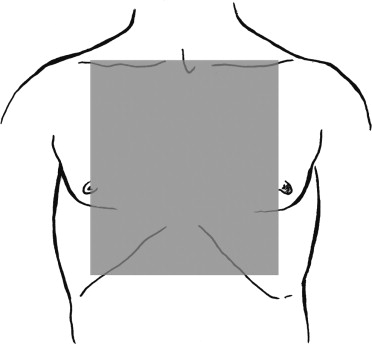
Transient responses to fluid boluses may also indicate the presence of cardiac tamponade. Rapid accumulation of fluid in the pericardial sac can compromise cardiac filling and result in hypotension and arrest. In the acute setting, as little as 100 mL of fluid can cause tamponade. Although blunt cardiac rupture causing tamponade is rare, penetrating wounds, particularly stab wounds within the “mediastinal box” (the area bordered by the midclavicular lines, the sternal notch, and the rib border), should raise the index of suspicion for cardiac tamponade in a patient who deteriorates clinically ( Figure 4-4 ). Treatment with pericardiocentesis may temporarily alleviate the tamponade until definitive treatment is accomplished.
Disability
The primary survey neurological assessment identifies any severe injury that requires immediate intervention or further evaluation. Pupillary size and responsiveness, lateralizing signs, or evidence of spinal cord injury should be assessed, with careful documentation of the level of injury. The Glasgow Coma Score (GCS) is a rapid, simple method of determining the patient’s level of consciousness, is reproducible, and has been shown to be predictive of patient outcome ( Table 4-3 ).
| Points | |
|---|---|
| Eyes (E) | 4—opens eyes spontaneously |
| 3—opens eyes to voice | |
| 2—opens eyes to pain | |
| 1—no eye opening | |
| Motor (M) | 6—follows commands |
| 5—localizes to pain | |
| 4—withdraws to pain | |
| 3—abnormal flexion (decorticate) | |
| 2—extensor response (decerebrate) | |
| 1—no movement | |
| Verbal (V) | 5—coherent and oriented |
| 4—confused conversation | |
| 3—inappropriate words | |
| 2—incomprehensible sounds | |
| 1—no response |
* The best response elicited in each category is assigned the appropriate point value. The patient’s GCS = E + M + V.
Besides direct traumatic brain injury, other causes of decreased level of consciousness must be taken into account. Inadequate cerebral oxygenation or perfusion, alcohol ingestion, narcotic or paralytic administration, and hypoglycemia can also cause altered mental status. However, in the acutely injured patient, changes in level of consciousness should be attributed to traumatic brain injury until proven otherwise. The patient’s neurologic condition should be monitored closely during the primary survey; waxing and waning mental status, or the classic findings of a lucid interval associated with epidural hematoma, can mask sudden neurological deterioration. Any change in level of consciousness should also prompt a rapid reevaluation of the patient’s oxygenation, ventilation, and perfusion status.
Exposure and Environment
Finally, in preparation for the secondary examination, the patient should be completely undressed so a more detailed examination of the entire body can be performed. During this portion of the evaluation, take care to avoid patient hypothermia. It is a common problem, occurring in up to 70% of trauma patients. Patients who have received paralytics (e.g., for intubation) are especially susceptible, since the natural shivering mechanism for generating heat has been eliminated. Radiant heat losses and administration of cold fluid also contribute to hypothermia. Hypothermia decreases the efficacy of the clotting cascade; even a decrease of a few degrees of core temperature can result in a marked decrease in clotting ability. This reduction in clotting function can be critical in the injured patient. The use of warming devices, environmental heating, and administration of warmed fluids can help to reverse these trends and should be considered early in the evaluation of the injured patient ( Figure 4-5 ).
ADJUNCTS TO THE PRIMARY SURVEY
Electrocardiograph and saturation monitoring should be performed immediately and evaluated frequently during the primary survey. After the primary survey is complete, a decompressive gastric tube can be placed. Note that placement of a gastric tube decreases but does not eliminate the possibility of aspiration. If there is evidence of midface or cribriform plate injury, nasogastric placement should be avoided. In the awake patient, orogastric placement may not be tolerated and can induce vomiting. Another useful adjunct is the urinary catheter, which can be used to monitor renal perfusion and volume status. If there is evidence of urethral disruption (meatal blood, perineal ecchymosis, high-riding prostate on rectal examination), a transurethral catheter should not be placed. Appropriate limited radiographs can also be obtained at this time, including anteroposterior chest and pelvic films and a lateral cervical spine film.
Often during the primary survey and resuscitation, enough information is obtained to indicate the need for transfer of the patient to another facility. The transfer process can be initiated while further evaluation and resuscitative measures are performed; however, the transfer of the patient to a facility more readily equipped to deal with the injured patient should never be delayed to complete further diagnostic studies. Physician-to-physician communication prior to the transfer is essential. Before continuing with the secondary evaluation or transfer of the patient, a brief review of the primary survey should be performed to ensure that no potentially life-threatening situations (e.g., tension pneumothorax on positive pressure ventilation or migration of endotracheal tube) have developed. Although the mnemonic for the primary survey suggests a sequential nature, many of these therapies occur simultaneously in practice, especially when adequate personnel are present. However, in situations where personnel or experience is limited, following the primary survey guidelines can prevent a care provider from being distracted by obvious but not life-threatening injuries and may improve patient outcome.
SECONDARY SURVEY
The secondary survey is not initiated until the primary survey is completed, resuscitation is established, and the patient is demonstrating normalized vital functions. Only then is a systematic, head-to-toe, physical examination completed with identification of all non-life-threatening injuries. Additional radiographic studies can be performed as indicated from the physical examination. Any incomplete portions of the medical history should be obtained at this time.
Head and Neck
The secondary survey begins with evaluating the head and any concomitant neurological injury. The injured patient should be presumed to have an unstable cervical spine injury. Absence of a neurological deficit does not exclude injury to the cervical spine. Consequently, the patient’s neck should remain immobilized until injury has been excluded by radiographic and clinical examination. General inspection of the head and neck for lacerations, abrasions, contusions, and deformities suggestive of fractures should be undertaken. Ocular examination should include evaluation of pupillary size, reactivity, hemorrhage, acuity, and muscular entrapment. Although distended neck veins are often cited as the hallmark of cardiac tamponade, the use of spine immobilization devices often precludes accurate and frequent examination of these signs, and the diagnosis of cardiac tamponade often must be made on clinical suspicion alone.
Chest
Completion of the chest examination begun in the primary survey includes continued visual inspection and palpation of the chest wall, clavicles, and sternum. Auscultation in a noisy environment may obscure subtle findings; however, decreased unilateral breath sounds should raise suspicion for a malpositioned endotracheal tube, pneumothorax, or hemothorax. Muffled cardiac sounds, often cited as a hallmark of cardiac tamponade, are also often difficult to ascertain in the emergency situation.
Abdomen
Abdominal injuries must be identified and treated aggressively. However, they can be difficult to diagnose since many intra-abdominal injuries do not necessarily present with abdominal pain or discomfort. Although serial abdominal examinations can be used to exclude intra-abdominal injuries, this method requires a cooperative patient without significant distracting injuries. In the patient with altered mental status, whether from head injury, intoxication, or other drug ingestion, physical examination may not be reliable, and more objective evaluation of the abdomen should be performed if the patient is stable (see Adjuncts to the Secondary Survey). Although the timely identification of some injuries may be paramount (e.g., those that cause significant intra-abdominal hemorrhage), the specific diagnosis is not as important as recognizing that an injury exists and that surgical intervention may be necessary.
Pelvic and perineal examination should be performed as part of the abdominal examination. Assessment of pelvic stability to anteroposterior and lateral compression should be performed only once, and excessive manipulation of the unstable pelvis should be avoided, since this can lead to further hemorrhage and significant blood loss. The most expeditious maneuver to stabilize the pelvis is to wrap a sheet tightly across the pelvis at the level of the trochanters until more definitive stabilization can be obtained. The perineum should be examined for contusions, lacerations, hematoma, and urethral blood. Rectal examination should be performed before the placement of a urinary catheter. Vaginal examination should also be performed, particularly to assess for injury if perineal bleeding is seen. Pelvic fractures can cause vaginal lacerations and should be treated as open fractures.
Musculoskeletal System
The extremities should be inspected for lacerations, contusions, and deformities suggestive of fractures. Peripheral pulses should be examined and compared for symmetry. In general, limb deformities should be evaluated with appropriate radiographs and reduced as soon as possible in order to alleviate patient discomfort and limit the amount of blood loss from the fracture. External hemorrhage from the limb should be controlled with direct localized pressure. A well-placed digit with a minimal amount of pressure can be more effective than a bulky dressing with generalized pressure to the area. Absence of peripheral pulses in a fractured limb should prompt rapid realignment of the limb. Persistent decrease in pulse examination should be attributed to vascular injury until determined otherwise.
Fat embolism, most often resulting from long bone fractures, usually does not develop immediately. This syndrome usually presents with progressive respiratory and neurologic decline and is sometimes associated with thrombocytopenia and petechiae, particularly on the thorax and abdomen. Treatment consists of fracture repair and supportive respiratory care.
The musculoskeletal examination should include palpation of the vertebral column, noting any areas of point tenderness or step-offs. In the awake, alert patient, the absence of pain or tenderness along the spine virtually excludes the presence of significant spinal injury. In the patient with altered mental status, appropriate radiographs must be taken to exclude spinal cord injury. If these films are inconclusive, spine immobilization should continue until fracture or injury is excluded, although efforts should be made to remove the patient from the spine board in a timely fashion to prevent development of decubitus ulcerations.
ADJUNCTS TO THE SECONDARY SURVEY
Specialized diagnostic procedures that are used to confirm suspected injury should only be performed after life-threatening injuries have been treated and the patient is hemodynamically stable. If the primary survey indicates the need for a more specialized facility, transfer of the patient should not be delayed for completion of any other studies.
Computerized tomography (CT) of the head is a rapid, noninvasive means of assessing neurologic injuries and should be performed in any patient with changes in mental status. Head CT is very effective at diagnosing intracranial injuries such as hemorrhage, contusion, shear injury, and edema. Effacement of the ventricles or midline shift suggests significant cerebral edema and may be an indication for intracranial pressure (ICP) monitoring, depending on the clinical status of the patient. The Monro-Kellie doctrine states that the volume of intracranial contents, composed of cerebrospinal fluid (CSF), blood, and brain parenchyma, remains constant. Although compensatory adjustments for mass effect from edema or clot exist, they are relatively limited. After this mass exceeds approximately 100 mL, there is a rapid increase in ICPs that can lead to cessation of blood flow. The primary focus of treatment for patients with suspected traumatic brain injury is to prevent secondary injury by ensuring adequate oxygenation and perfusion of the central nervous system.
Spine films should be used in conjunction with physical examination. If these are inadequate, CT may be necessary. The presence of ligamentous injury may not be apparent on CT scan, and a clinical tertiary examination should be performed when the patient’s mental status allows. Until spine fractures are excluded, particularly those of the cervical spine, spinal precautions such as backboard immobilization or log rolling should be used.
Several diagnostic adjuncts are available for the evaluation of blunt abdominal injuries. Diagnostic peritoneal lavage (DPL) requires the placement of an intraperitoneal catheter to evaluate for the presence of intra-abdominal blood. There is significant potential for iatrogenic injury in inexperienced hands, and DPL should be reserved for individuals comfortable with this technique. Recently, the focused abdominal sonogram for trauma (FAST) has largely replaced DPL as a rapid, non-invasive technique for evaluating the peritoneal cavity for abnormal fluid, which indicates abdominal hemorrhage in the case of most injured patients. Although this method is rapid and noninvasive, its sensitivity depends on operator expertise and may not detect small amounts (<200 mL) of peritoneal fluid. Abdominal CT is the most specific study available for identifying abdominal injuries. The use of intravenous contrast is preferable since it is more effective in delineating solid organ injury and can also be used to evaluate for ongoing hemorrhage and arterial extravasation. In most patients, the use of oral contrast is unnecessary and delays completion of the study. In addition to identifying intraperitoneal hemorrhage, CT can be used to examine the retroperitoneum and pelvis for evidence of injury or hemorrhage. Computerized tomography should only be used in those patients who are hemodynamically stable and should not delay transport of the patient to an appropriate trauma facility if needed.
Extremity films should be obtained as directed by clinical examination. Areas of point tenderness, swelling, ecchymosis, or deformity should be imaged to detect fractures. Any bony injuries associated with lacerations should be considered open fractures, and urgent treatment by an appropriate orthopedic surgeon is needed.
DEFINITIVE CARE
Once the determination has been made that the care of the injured patient exceeds the available resources or requires specialized facilities or personnel, arrangements should be made for transfer to a more appropriate institution. The primary survey should be completed by stabilizing the patient and initiating resuscitation. Contact with the accepting physician can be initiated by ancillary personnel, but there is no substitute for physician-to-physician communication. The accepting physician may request other interventions prior to transfer (e.g., placement of tube thoracostomy for stable pneumothorax if the patient is to be transported by air). All documentation, including laboratory values and radiographic studies, should be sent with the patient, since repetition of studies at the accepting institution exposes the patient to additional radiation and further delays definitive care. Most importantly, completion of radiographic studies or other secondary survey adjuncts should never delay transfer.
PENETRATING INJURIES
Most trauma patients seen at civilian centers will be injured through blunt rather than penetrating mechanisms. However, in this age of increasing violence and availability of handguns, penetrating injuries are becoming more commonplace and at some institutions can account for up to 40% of all injured patients seen.
Although the principles of ATLS are applicable to both blunt and penetrating injuries, penetrating injuries are more likely to require surgical intervention. In general, the most common mechanisms of penetrating injuries are stab wounds or impalements as well as gunshot wounds. In general, stab wounds are more likely to cause localized injury, and knowledge of the anatomy surrounding the wound can be useful in predicting injured organs. Foreign bodies, particularly those involved in impalement injuries, should not be removed in the emergency department, since release of tamponade may result in significant hemorrhage. Gunshot wounds have the potential to injure organs far from the external wound, and it is not uncommon to find missiles that have traveled through more than one body cavity, making operative prioritization even more difficult. As such, determination of the number and location of wounds, including careful evaluation of the patient’s back, is necessary. If the patient is stable, plain films can assist in determining the trajectory of the missile, particularly if there is an odd number of wounds. Because these mechanisms differ widely, the following is a generalized review of penetrating injuries that should be individualized for the patient, weapon, trajectory, and hemodynamic status.
Head and Neck
Penetrating injuries to the head are often lethal, since the inability of the cranium to expand to accommodate blood and brain edema can result in rapidly elevated ICPs. Standard ATLS protocols should be followed with particular attention to minimizing secondary anoxic brain injury while appropriate neurosurgical intervention is obtained. Penetrating brain injury is frequently associated with coagulopathy, and these patients must be aggressively monitored and given appropriate coagulation factors as needed.
Neck injuries are often managed based on location. Zone II injuries, extending from the angle of the mandible to the cricoid cartilage, are most amenable to surgical intervention. Even external wounds that appear innocuous can cause injury to vascular structures, upper airway, or esophagus; thus, any penetrating injury that violates the platysma should be carefully evaluated by an experienced surgeon. Although zone II injuries formerly mandated surgical exploration, more recent studies have examined the use of less-invasive modalities for evaluating these structures. Angiography, CT angiogram, esophagram, and esophagoscopy have also been used to diagnose injuries with varying degrees of sensitivity and specificity. Penetrating wounds to zone I (clavicle to cricoid) and zone III of the neck (angle of mandible to base of skull) are much less amenable to surgical intervention and often require special diagnostic and radiographic studies to exclude injury.
Chest
Penetrating injuries to the thorax can result in a range of injuries from relatively benign lung parenchymal injuries to lethal cardiac wounds. Stab wounds to the chest should not be explored, since the potential for creating pneumothoraces or dislodging stable blood clots is high. Wounds to the “mediastinal box” are more likely to result in cardiac injury. Suspicion for cardiac tamponade must be high in these cases, particularly because many patients with cardiac wounds do not arrive in extremis. Pericardial lacerations may vent the pericardium and present as a hemothorax, or the patient may have tamponade that is initially well compensated. Tension pneumothorax and massive hemothorax are other causes of hemodynamic deterioration in the injured patient, and decompression with needle or tube thoracostomy should be performed expeditiously. Initial evacuation of more than 1500 mL of blood or continued hemorrhage of greater than 200 mL/h warrants surgical exploration.
Abdomen
Stab wounds to the anterior abdominal wall can be evaluated with exploration under local anesthetic. Although enlarging the incision may improve exposure, this technique is often difficult, particularly in the obese patient. Violation of the muscle fascia raises the likelihood of peritoneal injury, and surgical consultation is advised. Radiographic adjuncts, such as ultrasound and CT scan, may miss injuries if only a small amount of intraperitoneal fluid is present. In the awake, alert patient, serial abdominal examinations may assist in determining the need for exploratory laparotomy. Because of the degree of musculature in the flank and back, stab wounds to these areas are less likely to cause intraperitoneal injury. CT with triple radiographic contrast (intravenous, oral, and rectal) can help delineate the retroperitoneal structures and determine if operative intervention is needed.
Thoracoabdominal Wounds
Because of the normal excursion of the diaphragm, some penetrating injuries may cause damage both in the thorax and abdomen. Anteriorly, wounds below the nipple should be evaluated for abdominal injury as well as chest injury. Posteriorly, wounds below the tip of the scapula (approximately the level of T8) should be considered as potential abdominal as well as thoracic wounds. Stab wounds in these regions are particularly difficult to evaluate, and triple contrast CT scan or diagnostic laparoscopy may be of benefit in selected cases.
Extremities
Penetrating injuries to the extremities should be evaluated for fractures, vascular injuries, and nerve injuries. Fractures should be reduced, particularly if vascular compromise is noted. Patients with open fractures should be given antibiotics and tetanus prophylaxis. Wounds in close proximity to vascular structures should be evaluated for expanding hematoma, evidence of arterial or venous hemorrhage, and distal extremity pulses as applicable. Gunshot wounds have the potential to create secondary blast injury to vascular structures, and peripheral pulses should be carefully examined.
ICU CARE OF THE TRAUMATICALLY INJURED PATIENT
As part of their definitive care, many patients with multiple injuries will be admitted to an intensive care environment. Common reasons for admission include respiratory insufficiency, potential loss of airway, hemodynamic instability with the need for ongoing resuscitation and intensive monitoring, and decreased level of consciousness in association with a traumatic brain injury. As a general rule, any patient who exceeds the level of monitoring and nursing or respiratory care readily available on an inpatient patient care floor or patients with threatened airway should be admitted to an ICU setting.
Intensive care of the trauma patient is best approached with a multidisciplinary team including intensivists, trauma surgeons, nurses, respiratory care practitioners, critical care pharmacists, and nutritionists. Care of these patients is frequently complex, with injuries that may cross many organ systems and require the involvement of multiple sub- and super-specialists. In this setting, the trauma surgeon coordinates care among multiple consultants.
Delivery of optimal ICU care may require a great deal of time and resources. As the saying goes, “The devil is in the details” (see Volume II, Chapter 6 ). This is especially true of the ICU. Proper intensive care of the trauma patient requires meticulous attention to detail. In this setting, a systematic approach to the patient by organ system is quite useful ( Table 4-4 ). This helps the ICU team and physicians to approach daily rounds and problem solving in a logical and methodical pattern. In the remainder of this chapter, we will highlight issues within each organ system frequently faced by traumatically injured patients in the ICU. This section is meant to serve as an introduction for the oral and maxillofacial surgeon to ICU care.
| Respiratory | Infectious disease |
| Cardiovascular | Injury specific needs |
| Fluids, electrolytes, nutrition | Pain control and sedation |
| Gastrointestinal | Tubes and lines |
| Hematological | Prophylaxis |
| Renal | Rehabilitation |
| Endocrine |
* Trauma patients may have active issues in multiple areas. Use of an organ systems approach assists in clarification of management strategy and maintaining attention to detail.
RESPIRATORY SYSTEM
Intubation and Mechanical Ventilation
Many trauma patients in the ICU require intubation for airway protection and mechanical ventilation. Common indications for intubation and mechanical ventilation include hypoxia, hypercapnea, inability to control secretions, potential loss of airway, need for deep sedation, and the need to remove work of breathing from a patient’s overall metabolic demand.
Common clinical indicators to guide intubation include inability of the patient to speak in full sentences, use of accessory respiratory muscles, a feeling of air hunger or being smothered, weakened inspiratory effort, tachypnea, and altered mental status with decreased GCS score and depressed level of consciousness. The monitoring of vital signs and use of pulse oximetry and arterial blood gas analysis are useful adjuncts. Persistent hypoxemia with arterial oxygen saturations less than 90% and the presence of respiratory acidosis (pH <7.25) or hypercapnea (PaCO 2 >50 mm Hg in non-COPD patients) are strong indicators of the need for intubation and mechanical ventilation. It is important to remember that semi-elective intubation is nearly always less stressful for the patient and involved caregivers than is emergency intubation. Respiratory insufficiency is best treated in this fashion with early intubation and mechanical ventilation rather than allowing this to progress to respiratory failure. Semi-elective intubation also allows adequate preparation of patient and equipment with subsequent minimization of potential complications at the time of intubation.
Following intubation, the patient is oxygenated and ventilated. Many different modes of ventilation, such as assist control (AC) or synchronized intermittent mechanical ventilation (SIMV), may be used. Use of specific modes and methods of mechanical ventilation varies widely among critical care physicians, is at times a controversial topic, and likely matters little as long as the patient is treated in a method that is viewed as “lung protective.” With this in mind, it is useful to remember that the goals of ventilator support are relatively simple and revolve around ensuring adequate oxygenation and ventilation for the patient. Adequacy of oxygenation is determined by oxygen saturation, with a target of greater than 92% saturated hemoglobin. Ventilation is considered appropriate when a normal pH is achieved from a respiratory source. When approaching issues with oxygenation and ventilation, various aspects of ventilatory support may be manipulated to achieve these goals. As a general rule, oxygenation can be improved by increasing FiO 2 or positive end expiratory pressure (PEEP). The respiratory component of pH can be altered by changing minute ventilation (minute ventilation = respiratory rate × tidal volume), with increasing minute ventilation leading to decreased PaCO 2 and subsequently increased pH, and decreasing minute ventilation leading to increased PaCO 2 and decreased pH. This simplified approach allows correction of the vast majority of acute ventilation and oxygenation issues.
Liberation from Mechanical Ventilation
As the patient improves, the level of ventilatory support should be decreased as tolerated until minimal ventilator settings are reached. This is commonly known as “weaning” but is perhaps better conceptualized as “liberation from mechanical ventilation.” Many weaning methods have been described, but none have proven superior. Prior to extubation, the following should be considered: (1) Does the patient have adequate ventilatory and oxygenation ability to allow removal of external ventilatory support, (2) does the patient’s mental status allow for adequate airway protection, and (3) has the underlying condition that led to the patient being intubated and ventilated resolved adequately to allow removal of such support? Answers to the first question may be determined from the results of a trial of unassisted breathing and calculation of the rapid shallow breathing index (RSBI). RSBI is the ratio of respiratory rate to tidal volume. An RSBI of less than 80 suggests that the patient may tolerate removal of mechanical ventilation, while an RSBI of greater than 105 suggests that the patient will not tolerate removal of mechanical ventilatory support. Answers to the other questions must be determined using clinical judgment.
Acute Respiratory Distress Syndrome
Acute respiratory distress syndrome (ARDS), also known by the sobriquets of “Da Nang Lung” and “non-cardiogenic pulmonary edema,” was first described in the late 1960s. ARDS is characterized by the presence of an inflammatory state in the lungs, leading to an alveolar capillary leak with accumulation of an exudative material in the alveoli. The pathophysiology state of ARDS is different than that of cardiogenic pulmonary edema in that it is a state of inflammation rather than hydrostatic edema. This leads to impaired alveolar gas exchange. Subsequently, oxygenation is impaired and pulmonary compliance decreases. Patients with ARDS may be quite challenging for the ICU team to treat and require substantial ventilatory support. Diagnostic criteria for ARDS and its related condition acute lung injury (ALI) are given in Table 4-5 .
| Category | ALI | ARDS |
|---|---|---|
| PaO 2 /fraction of inspired oxygen ratio | <300 | <200 |
| Chest radiograph | Infiltrates consistent with pulmonary edema | Infiltrates consistent with pulmonary edema |
| Estimated pulmonary artery occlusive pressure | <18 mm Hg | <18 mm Hg |
Clinically, ARDS is associated with decreased oxygenation and the presence of infiltrates on chest radiograph. Patients with ARDS should be ventilated using strategies for lung protection ( Table 4-6 ).
| Category | Protocol |
|---|---|
| Ventilator mode | Volume targeted assist control |
| Tidal volume | 6 mL/kg (based on gender and height) |
| Plateau pressure | No more than 30 cm H 2 O |
CARDIOVASCULAR SYSTEM
Shock
A substantial proportion of trauma patients admitted to the ICU will be suffering from shock. Shock is best conceptualized as cellular hypoxia, which in turn leads to loss of cellular membrane potential, with resultant swelling and damage. The etiology of shock states can be broadly categorized using a schema first described by Alfred Blalock, MD, in the 1930s: cardiogenic, septic, neurogenic, and hypovolemic. Trauma patients often present with hypovolemic shock resulting from hemorrhage. Neurogenic shock or cardiogenic shock may also be present. The diagnosis of shock states in trauma patients can be made more challenging by the presence of more than one category of shock. For example, a patient injured in a fall who has neurogenic shock resulting from a cervical spinal cord injury may also have hypovolemic shock from a large splenic laceration. A primary goal of early definitive care of the trauma patient is resuscitation from shock. Guidelines for and endpoints of resuscitation are discussed elsewhere in this text.
SIRS and MODS
The magnitude of injury sustained by trauma patients as well as the severity of shock and need for blood and blood component transfusion may lead to systemic inflammatory response syndrome (SIRS). SIRS is the result of an exuberant pro-inflammatory response and is defined by two of the following: fever or hypothermia, tachycardia, tachypnea, hyperventilation, leukocytosis or leukopenia, or bandemia. SIRS can be conceptualized as similar to a local response to an insult, except extended to a whole body level.
SIRS that involves organ dysfunction is termed multiple organ dysfunction syndrome (MODS) . This may progress to multiple organ failure. Mortality in patients with SIRS and MODS is directly related to the number of organ systems that fail, and ranges from approximately 40% for one-organ system to more than 80% for four-organ system failure. Strategically, SIRS and MODS are treated by reversing the inciting event, if possible, then ensuring resuscitation and adequate oxygen delivery.
Blunt Myocardial Injury
A subset of trauma patients may be hemodynamically compromised as a result of blunt myocardial injury, also known as cardiac contusion. This injury typically results from a direct blow to the chest and may occur in the absence of overlying rib or sternal fractures. Diagnosis of this condition may be challenging. The most common clinical sign is sinus tachycardia, which is relatively nonspecific in this patient population. Patients with suspected blunt myocardial injury and ECG changes, which may include ST segment elevation or conduction delays, should be closely monitored until these findings resolve. Patients presenting with hemodynamic compromise will often have right heart failure as a result of the injury. In this setting, resuscitation and inotropic support, along with afterload reduction, may be necessary.
Atrial Fibrillation
After sinus tachycardia, the most common tachydysrhythmia present in trauma patients in the ICU is atrial fibrillation. With the aging of the U.S. population, a larger proportion of trauma patients present with pre-existing chronic medical conditions such as atrial fibrillation. In addition, an underlying propensity for this dysrhythmia may be unmasked by catecholamine release, fluctuating electrolyte levels, fluid overload, or fluid depletion.
Atrial contraction contributes as much as one-fourth of the ventricular end diastolic volume. Loss of this “atrial kick,” as occurs in atrial fibrillation, is well tolerated by itself. In the setting of a non-compliant left ventricle, as in long-standing hypertension, or with the presence of a rapid ventricular response, atrial fibrillation may lead to lowered cardiac output and hemodynamic compromise.
In the treatment of atrial fibrillation with rapid ventricular response, it is more important to control the ventricular response component than convert the heart to sinus rhythm. If the patient is hemodynamically unstable, protocols for advanced cardiac life support (ACLS) should be followed. If the patient is hemodynamically stable, rate control may be achieved pharmacologically with several different classes of agents, including calcium channel blockers, beta-adrenergic receptor blockers, amiodarone, and digoxin. Once rate control is achieved, the cause of the dysrhythmia should be considered and reversed if possible. If the patient remains in atrial fibrillation for more than a brief period, anticoagulation should be initiated if not contraindicated because of increased risk of atrial mural thrombosis with distal thromboembolism.
FLUIDS, ELECTROLYTES, AND NUTRITION
Electrolytes
In the initial period following a traumatic injury, electrolytes should be monitored at least daily. Common practice is to follow these parameters even more frequently during acute resuscitations. Alterations in serum levels of electrolytes are common and many ICUs develop protocols to guide replacement therapy.
Nutrition
The importance of nutrition in the critically ill patient can never be overstated. The presence of inadequate nutrition in the traumatized patient is common and is associated with increased rates of complications, wound failure, and death. In addition, the metabolic state present in trauma leads to increased daily needs for calories and protein.
A “rough” estimate of basal energy expenditure (BEE) can be determined by the following equation:
In critically ill patients, caloric needs may increase substantially over BEE. More accurate determination of caloric requirements can be achieved using either indirect calorimetry or the Harris-Benedict equation.
Indirect calorimetry uses specialized equipment, also known as a metabolic cart. This equipment measures pulmonary exchange of carbon dioxide and oxygen, allowing the determination of respiratory quotient as well as BEE. Indirect calorimetry can be performed at the bedside in the ICU.
Caloric needs may also be estimated by the Harris-Benedict equation, which states:
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


