Introduction
Proper diagnosis and management of eruption disturbances remains challenging but is critical to a functional occlusion. The objective of this study was to establish definitive criteria to differentiate and diagnose eruption disorders, specifically primary failure of eruption (PFE) and ankylosis.
Methods
Sixty-four affected persons were placed into 3 cohorts: PFE diagnosed through confirmed PTH1R mutation (n = 11), PFE diagnosed based on clinical criteria (n = 47), and ankylosis diagnosed based on clinical criteria (n = 6). These groups were assessed to identify clinical features that differentiate PFE and ankylosis.
Results
Ninety-three percent of the subjects in the genetic and clinical PFE cohorts combined (n = 58) and 100% in the genetic PFE cohort had at least 1 infraoccluded first permanent molar. Additionally, a novel functional PTH1R mutation, 1092delG, was identified and linked to PFE in the deciduous dentition.
Conclusions
An infraoccluded, supracrestal first molar is a hallmark of PFE, often involving both arches in the permanent or deciduous dentition, and with unilateral or bilateral affection, infraoccluded second premolar or second molar, and multiple affected adjacent teeth. Our results further suggest that PFE and ankylosis might be clinically indistinguishable without knowledge of prior trauma, treatment history, genetic information, or obliteration of the periodontal ligament space.
The process of eruption in the human dentition is complex and poorly understood. Although advances in molecular biology have increased our understanding of the mechanisms underlying dental eruption, the clinical correlations remain elusive. Consequently, our understanding of eruption is based on multiple, yet differing, theories that are both controversial and ill-supported. Eruption disorders therefore create a rare and unique diagnostic challenge for general dentists seeking to monitor the dental development of their patients, but it is particularly complex for orthodontists and pediatric dentists who must manage these patients. The gestalt of this challenge is a lack of definitive clinical diagnostic criteria to distinguish between different types of eruption disorders and differentiate them from idiopathic delayed dental development.
Disturbances in dental eruption can occur for many reasons. Among these are 2 clinical problems that form the central basis of this article: primary failure of eruption (PFE; OMIM: 125350) and ankylosis. Ankylosis is histologically defined as the fusion of cementum to bone in at least 1 area lacking a periodontal ligament space. The resultant occlusion has a tooth that ceases to erupt, drift, or move despite normal adolescent growth or orthodontic traction. In contrast to ankylosis, PFE does not include fusion of the cementum to the bone but is marked by a disturbance in the eruption mechanism itself, causing a nonankylosed tooth to fail to fully or partially erupt. Surgeons extracting teeth diagnosed as PFE have noted that the tooth is mobile in the socket, further differentiating these teeth from ankylosed teeth (Tim Turvey, personal communication, December 3, 2012). PFE was initially described based on its clinical appearance. This description of PFE is the most comprehensive diagnostic indicator to date and includes the following features: it primarily affects posterior teeth; it affects all teeth posterior to the most anteriorly affected tooth; the occlusion manifests as a lateral open bite; and the teeth do not respond favorably to orthodontic forces. A common clinical dilemma is distinguishing PFE from ankylosis—mainly because the disorders have a similar clinical appearance and developmental fate. There is some evidence that PFE is often misdiagnosed as ankylosis. It is important, however, to recognize that ankylosis and PFE dictate distinct treatment modalities, and inaccurate diagnosis could significantly alter the treatment success. The misdiagnosis and mismanagement of either eruption disorder could result in inappropriate and extended treatment, significant financial burdens, patient frustration, and an inferior occlusal condition.
The fact remains that PFE is a rare, yet handicapping, disorder in which the treatment options are unclear and unpredictable. The few current treatment options to improve the occlusion of a patient with PFE include small segmental osteotomies and prosthetic restoration of the occlusion. However, no treatment or limited esthetic treatment is often the best option because orthodontic treatment with a continuous archwire, even after extracting the most severely affected teeth, results in exacerbation of the lateral open bite by intrusion of the adjacent teeth and, frequently, ankylosis of the affected teeth. This is in contrast to ankylosis, which can be successfully treated by extraction of the ankylosed tooth and subsequent orthodontic movement of all other teeth. Thus, misdiagnosis of PFE and treatment with a continuous archwire can actually lead to an inferior occlusal result, providing a significant disservice to the patient.
It is expected that the uncertainty surrounding an eruption-disorder diagnosis will diminish with the increasing application of genetic analysis in this field. Unlike the limited clinical indicators discussed above, genetic analysis of specific genes offers an objective measure of the presence of pathology. Recent studies have shown that a genetic mutation in the PTH1R gene (associated with bone homeostasis) is also associated with PFE. The mutation is present in multiple members of some families who exhibit PFE. It has previously been reported that 10% to 40% of PFE cases are familial, and we expect that this estimate will increase as more is learned about the genetic makeup of patients diagnosed with PFE. The potential for a genetic diagnosis of PFE or other dental disorder is a huge step forward in establishing a definitive and objective diagnosis of PFE in patients with clinical characteristics of the disorder. Although genetic “testing” is not currently available for use in clinical practice for most dental disorders, it is in the early phases of development in the diagnosis of PFE and could be a chairside diagnostic test in the future. Logical first steps in developing this diagnostic rubric are to document and then associate the clinical features of PFE with the associated genetic mutations.
In this study, we sought to take advantage of a unique data set to establish the clinical diagnostic criteria that distinguish PFE from other eruption disorders, particularly ankylosis. The combination of objective genetic information and clinical data from affected persons can be used to establish a genotype-phenotype correlation for PFE and, by extension, an objective diagnosis: ie, determined by associating clinical (phenotypic) features with genetic (genotypic) analysis. We therefore compared clinical features identified in the genetically characterized sample to a broader data set of patients diagnosed with PFE based on clinical parameters only. The resultant developmental and morphologic features identified in patients with obvious clinical characteristics of PFE and harboring a genetic mutation in PTH1R will represent a hallmark of the condition, giving clinicians greater diagnostic certainty and subsequent improved clinical management.
Material and methods
A data set of 64 patients with eruption disorders was collected at the University of North Carolina over several years. This group consisted of patients of the Graduate Orthodontic Clinic, Faculty Practice, and various private practices (sent for consultation and recommendations from faculty at the University of North Carolina). After phenotypic review using radiographs or clinical photos, we placed these patients into 3 categories: those definitively diagnosed with PFE through genetic analysis that showed a mutation in PTH1R (n = 11; genetic PFE cohort), patients diagnosed with PFE based on clinical records alone (n = 47; clinical PFE cohort), and patients diagnosed with ankylosis based on clinical criteria (n = 6; clinical ankylosis cohort). All records were evaluated by the 3 authors, and agreement in diagnosis and feature identification was confirmed for all subjects. Those in the ankylosis cohort had a confirmed history of trauma or were treated with extraction of the affected tooth or teeth and had successful orthodontic treatment of the remaining teeth. All other subjects were diagnosed with PFE based on clinician acumen, history of unsuccessful orthodontic treatment, or genetic analysis.
Of the 58 subjects diagnosed with PFE, 27 underwent genetic (mutational) analysis (previously described); a mutation or polymorphism in PTH1R was identified in 11 patients, and an unclassified nonfunctional single nucleotide polymorphism in PTH1R was identified in the remaining 16. These 11 subjects comprised the genetic PFE cohort, and those with single nucleotide polymorphisms were grouped into the clinical PFE cohort. Mutational analysis was performed as follows: DNA was extracted and purified from salivary samples (Oragene; DNA Genotek, Toronto, Ontario, Canada). All coding regions of PTH1R (exons 3-16) were amplified and sequenced using previously described primer sets. Splice junctions were included in the sequencing results using primer sets designed to delineate regions that included a minimum of 25 bases on the intron sequence, in addition to the exon sequences. The amplification of sequences was performed with HotStart polymerase chain reaction MasterMix (GE Healthcare Life Sciences, Piscataway, NJ) under the following conditions: 10 minutes at 95°C activation or premelt, followed by 35 cycles of 30 seconds at 94°C melt, 30 seconds at 60°C anneal, and 3 minutes at 72°C extension. The polymerase chain reaction products were purified with Exosapit (Affymetrix, Santa Clara, Calif) and sequenced at the University of North Carolina’s genome analysis core facility. All sequences were compared with a wild type of PTH1R (accession NM_000316.2 ) from GenBank release GRCh37 using the BLAST algorithm.
Clinical (phenotypic) information was reviewed for all 3 cohorts to assess the subjects’ clinical features. The records assessed included a minimum of a panoramic radiograph for every patient, and cephalometric radiographs, intraoral periapical radiographs, and clinical photographs were used when available. The following information was gathered for the 3 cohorts: (1) unilateral or bilateral presentation of infraoccluded teeth; (2) arch involved (affected teeth in the maxilla, mandible, or both); (3) teeth with infraocclusion (at least 1 premolar, first molar, and second molar); (4) location of affected teeth in the alveolar ridge (supracrestal or infracrestal), and third molars were excluded from evaluation, as were second molars in young patients who would not be expected to have second molars erupted according to normal dental eruption timing; (5) presence or absence of root anomalies, including descriptions; (6) presence or absence of any other abnormal or noteworthy findings, including specific descriptions; and (7) record types provided.
Additionally, the following information was included when available and applicable: (1) PFE type I or II (determined by the degree of eruption of the second molars, as discussed below); (2) age; and (3) Class III dental or skeletal relationship (determined by high-quality clinical photos or cephalometric radiographs that clearly showed the skeletal Class III relationship; patients lacking these records were classified as indeterminable).
For the clinical and genetic PFE cohorts, the classification of PFE was recorded as type I or II as previously described in the literature. These types are distinguished based on the timing of onset and the presentation. Briefly, type I PFE is characterized by a progressive posterior open bite, in which all teeth distal to the most mesial infraoccluded tooth are affected and do not erupt into occlusion. Those with type II PFE exhibit greater eruption potential, although still inadequate, for the more distal teeth, such as second molars. Comparison of the eruption disorders based on the 3 cohorts was completed to identify similar and distinguishing characteristics. The genetic PFE cohort provides an objective basis to classify the associated clinical features. Therefore, the following comparisons were made: clinical features of the genetic PFE cohort with the clinical PFE and clinical ankylosis cohorts based on the attributes listed above.
Results
Twenty-four of the 58 PFE patients had age information available. The average age of the patients in this data set for which age was recorded was 12 years 9 months (range, 6 years 2 months-18 years 4 months).
Previously identified mutations in the PTH1R gene in addition to a novel mutation in PTH1R (1092delG, which results in a frameshift and premature termination of the PTH1R protein) formed the basis of the genetic cohort. This novel mutation, associated with the clinical finding of infraoccluded deciduous teeth, was discovered in a small nuclear family (n = 2). The index case was a boy, 7 years 9 months of age, with PFE affection of the permanent and deciduous teeth in the form of a right lateral posterior open bite and a mild Class III skeletal malocclusion ( Fig 1 ).
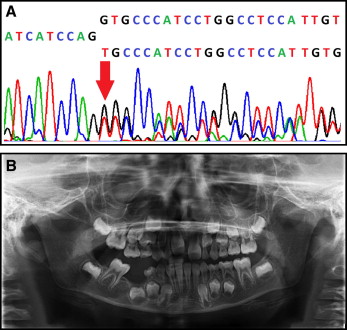
We evaluated a subset of patients with eruption failure that included both clinical and genetic data to establish a genotype-phenotype correlation. The Table summarizes the numbers and percentages of patients in our entire sample (n = 64) who had various clinical features. Specifically, the genetic cohort (n = 11) was examined for clinical characteristics of PFE. Since the subjects in the genetic cohort had a confirmed mutation in PTH1R , the hallmark features consistent with PFE are outlined based on this data set. Our phenotypic analysis primarily using radiographs and clinical photographs showed the following.
| PFE (genetic) (n = 11) | PFE (clinical) (n = 47) | Ankylosis (n = 6) | ||||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Symmetry | ||||||
| Unilateral | 6 | 54.5 | 21 | 44.7 | 5 | 83.3 |
| Bilateral | 5 | 45.5 | 26 | 55.3 | 1 | 16.7 |
| Arch involved | ||||||
| Maxilla | 0 | 0.0 | 9 | 19.1 | 2 | 33.3 |
| Mandible | 1 | 9.1 | 5 | 10.6 | 4 | 66.7 |
| Both | 10 | 90.9 | 33 | 70.2 | 0 | 0.0 |
| Teeth involved | ||||||
| At least 1 premolar | 8 | 72.7 | 30 | 63.8 | 0 | 0.0 |
| First permanent molar | 11 | 100.0 | 43 | 91.5 | 6 | 100.0 |
| Second permanent molar | 7 | 63.6 | 31 | 66.0 | 0 | 0.0 |
| Location in alveolar ridge | ||||||
| Supracrestal | 11 | 100.0 | 40 | 85.1 | 5 | 83.3 |
| Infracrestal | 0 | 0.0 | 2 | 4.3 | 1 | 16.7 |
| Both | 0 | 0.0 | 5 | 10.6 | 0 | 0.0 |
| PFE classification | ||||||
| Type I | 4 | 36.4 | 25 | 53.2 | NA | NA |
| Type II | 6 | 54.5 | 13 | 27.7 | NA | NA |
| Indeterminable | 1 | 9.1 | 9 | 19.1 | NA | NA |
| Mutation type | ||||||
| Intronic—substitution | 4 | 36.3 | NA | NA | NA | NA |
| Coding—substitution | 2 | 18.2 | NA | NA | NA | NA |
| Coding—insertion (frameshift) | 3 | 27.3 | NA | NA | NA | NA |
| Coding—deletion (frameshift) | 2 | 18.2 | NA | NA | NA | NA |
| Coding—nonfunctional SNP | 0 | 0.0 | 16 | 34.0 | NA | NA |
| Angle Class III | 7 | 63.6 | 11 | 23.4 | 3 | 50.0 |
| Dilacerated or blunted roots | 1 | 9.1 | 10 | 21.3 | 2 | 33.3 |
| Other dental anomaly | 4 | 36.4 | 16 | 34.0 | 2 | 33.3 |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


