Fig. 10.1
The sinus cavity is an aerial cavity of a triangular pyramid shape whose medial base corresponds to the lateral vertical wall of the nose and whose apex extends within the zygomatic process of the maxillary bone
The maxillary sinus bony cavity is internally lined by a mucous membrane, named Schneiderian membrane, which is covered by a pseudostratified columnar ciliated epithelium. The sinus epithelium has similar characteristics to the respiratory epithelium, but with less vascularization than the nasal epithelium. The antral mucosa is thinner and less vascular than nasal mucosa [1]. The membrane is of variable thickness, ranging between 0.13 and 0.5 mm.
A natural opening from the maxillary sinus (maxillary ostium) is located anterosuperiorly on the medial wall of the sinus. It is the only sinus drainage pathway, and it drains into the middle meatus. The ostium has a mean diameter of 2.4 mm [3] (Fig. 10.2).
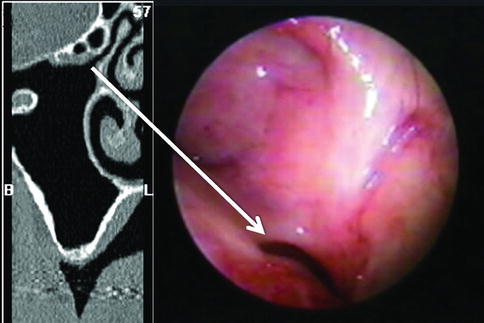

Fig. 10.2
Computed tomography scan (left side) and an endoscopic view (right side) of the maxillary sinus ostium (arrow)
The ciliated cells of the maxillary mucosa are able to beat about 8–20 times per second, in a specific direction, resulting in a pattern of mucus flows, which often moves material against gravity to the sinus’ exit. Thus, mucus may travel around the entire sinus cavity prior to exiting through the ostium and draining out.
Non-ciliated cells of the maxillary mucosa are characterized by microvilli that cover the apical aspect of the cell and serve to increase the surface area of the epithelium in order to improve air humidification and warming.
Serous and mucinous glands are located under the basal membrane and produce thick or thin mucus in response to the autonomic nervous system (parasympathetic system) [4]. The sinus’ mucus is composed mostly of water (96 %), but also contains immunoglobulins and additional factors. It constitutes the most important mechanism of protection of the nasal-sinus apparatus.
The maxillary vascular network is extensive, guaranteeing a high amount of blood flow, which have important surgical implications (Fig. 10.3).
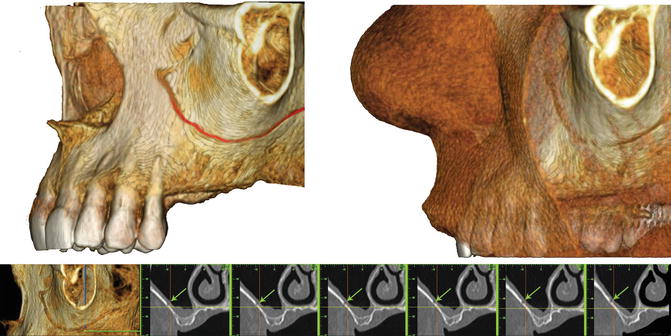

Fig. 10.3
Computed tomography scan 3D view of the lateral wall of the maxillary sinus which shows the point of emergence of the infraorbital artery (IOA) and the point of anastomosis between the IOA and the alveolar antral artery (AAA) as well as the route of the AAA. The arrow identifies the CT image of AAA
The venous drainage of the maxillary sinus occurs via the facial vein, the sphenopalatine vein, and the pterygoid plexus. The fact that the venous drainage of the sinus may occur both via typical pathways as the jugular veins and via the ethmoidal and frontal sinuses up to the cavernous sinus implies a risk of cerebral infection following maxillary sinus contamination. Spread of infection via this route is a serious complication of maxillary sinus infections [5].
The maxillary sinus nerve supply is via the maxillary branch of the trigeminal nerve, coming directly from the posterior middle superior nerves (in the posterior wall) and anterior superior alveolar nerves (anterior wall), the infraorbital nerve (superior or orbital wall), the anterior palatine nerve, and the lateral branches of the sphenopalatine nerve (medial or nasal wall) [6].
The alveolar nerves travel within the anterior wall of the sinus and innervate the related teeth [7]. Thus, it may be difficult to distinguish between pain of a dental origin and pain originating from the maxillary sinus.
The maxillary sinus plays several roles in the human body, such as reducing the relative weight of the cranium, insulating sensitive structures, providing immunological defense, and more. Still, the function of the paranasal sinuses remains partially unknown. It is suggested that the main functional structure of the nasal fossa and paranasal sinuses is the mucosal lining, which helps the removal of inhaled particles [8].
Morphological Features of Upper Maxillary Roots in Relation to the Maxillary Sinus, Periapical Infections, and Sinusitis
Many bacterial and fungal species, such as Streptococcus, Candida (especially C. albicans), and Propionibacterium may be involved in the etiology of persistent periapical infections [9]. Cultural studies showed that one of the most common bacterial species found in this type of lesions is Enterococcus faecalis [10–12].
In patients with chronic rhinosinusitis with nasal polyposis (CRS with NP), the presence of a complex bacterial contamination has been demonstrated. This contamination may cause damage to the mucosa, resulting in an inflammatory process, which may lead to a hyperplastic process. Staphylococcus species, Streptococcus viridans, Pseudomonas aeruginosa, Enterococcus faecalis, and Corynebacterium [13], and even Fungi, may be associated with cases of chronic hyperplastic sinusitis (CHS).
It is important to note that many microorganisms were found both in the maxillary sinus and in endodontic infections, for example, Staphylococcus epidermidis, Staphylococcus aureus, Klebsiella spp., and Streptococci [14].
Microorganisms that may contaminate the extra-radicular tissues are the few bacterial species able to survive outside the root canals in an immune-hostile environment. For example, Actinomyces (A. israelii and A. radicidentis) that elude the host immune system response by avoiding the phagocytic mechanism through bacterial aggregation [14]. Infection of the sinus caused by Actinomyces may therefore be difficult to treat because of the characteristics of resistance of these microorganisms [15, 16].
Intimate anatomic relationship may exist between the roots of the first and second maxillary molars and the maxillary sinus, sometimes with only a thin layer of cancellous bone separating the sockets of these maxillary molars and the floor of the sinus cavity. In many cases the molar roots may even protrude into the sinus cavity [8]. The root tip may touch or overlap the sinus floor, and the root apices may project into the floor of the sinus causing small elevations or prominences [17]. The mean distance between the maxillary posterior teeth and the floor of the maxillary sinus is about 2 mm.
Periapical periodontitis may result in maxillary sinusitis of dental origin with resultant inflammation and thickening of the mucosal lining of the sinus in areas adjacent to the involved teeth (Fig. 10.4a, b). Melen et al. reported that the frequency of sinusitis of dental origin varied considerably, ranging between 4.6 and 47 % of all sinusitis cases [18]. Studies showed that sinus mucosal hyperplasia is present in approximately 80 % of maxillary teeth with periapical pathology [19]. The closer the apex of a pulpally involved tooth to the floor of the sinus, the more likely and the greater the impact on the sinus tissues [20].
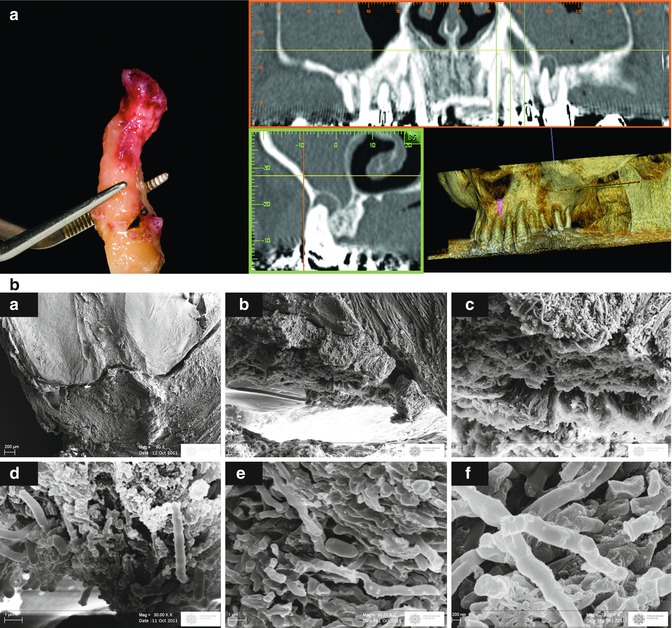

Fig. 10.4
(a) Computed tomography views of a maxillary premolar tooth with persistent periapical endodontic lesion and the tooth soon after extraction showing the apical lesion attached to the root apex. (b) A sequence of SEM images at different magnification of a section passing through the apical foramen of treated tooth. (a: Apical third of the root canal system showing a pathological tissue attached to the apex, b, c, e: Bacterial biofilm in contact with the apical wall of the root surface extending to the external root surface, d: bacterial biofilm at higher magnification showing intertwining bacterial filaments appears to be mainly constituted by E. Coli). A pathologic tissue is attached to the apex. Bacterial biofilm protrudes through the apical foramen
In a study performed on cadavers in 1943, Bauer [21] reported a direct extension of dental sepsis into the sinus and presented examples of pulpally involved teeth with histologically evident extension of the disease into the maxillary sinus. The same study also revealed microscopically the “diseased areas,” showing the destruction of the bone separating the sinus from the teeth, with particular loss of the cortical bone normally found in the sinus floor. Other changes were found in the sinus mucosa, such as swelling with inflammation, cyst formation, hypertrophy, and even transformation of the mucosa to granulation tissue, hyalinization, or complete necrosis [21].
Sinusitis may follow endodontic surgery in the absence of previous inflammation of the sinus mucosa. The resected apex, the root canal filler (most frequently gutta-percha), and/or the retrograde filler may migrate into the maxillary sinus cavity, inducing a foreign body reaction by the respiratory mucosa, leading to a sinusitis. Moreover, oral pathogens may directly infect the maxillary mucosa by migrating from the oral environment or from the periapical lesion area into the sinus. Another mechanism of sinusitis following periapical surgery is the obstruction of the ostium due to postoperative swelling of the maxillary sinus mucosa with complete impairment of the function of the mucociliary cleansing system, even in the absence of membrane perforation and direct communication between the sinus and oral cavities [22].
Sinusitis (or rhinosinusitis) is defined as an inflammation of the mucous membrane that lines the paranasal sinuses and is classified to several categories based on chronology of the disease:
1.
Acute: a new infection that may last up to four weeks and can be subdivided symptomatically into severe and non-severe.
2.
Recurrent acute: four or more separate episodes of acute sinusitis that occur within one year.
3.
Subacute: an infection that lasts between four and 12 weeks and represents a transition between acute and chronic infection.
4.
Chronic rhinosinusitis: the signs and symptoms last for more than 12 weeks.
5.
Acute exacerbation of chronic sinusitis: the signs and symptoms of chronic rhinosinusitis exacerbate but return to baseline after treatment.
The typical signs and symptoms of sinusitis are headache and facial pain, or a constant dull pressure (dull pain), or ache over the affected sinuses [4]. This pain is typically localized in the involved sinus and may change when the affected person bends over or lies down or during mastication. A feeling of “fullness” around the first molar-second premolar area is often present [23].
Sinusitis may be accompanied by thick nasal discharge and may contain pus (purulent) and/or blood. Nasal discharge is considered a significant sign of sinus infection.
Severe acute or subacute sinusitis rarely produces fever. However, as said before, severe fulminant sinusitis may produce high fever.
Some authors correlated the time course of disease development to the degree of infection [5, 24–26].
In periapical chronic inflammation, the disease develops slowly, with no significant infection, and its spreading within the sinus may be slow and with minimal impact. On the contrary, acute infectious pulpal disease is much more rapidly spreading and destructive, involving the adjacent sinus within a short time. There have been reported cases of rapid spread of dental infections through the maxillary sinus leading to subsequent periorbital cellulitis, blindness, and even life-threatening cavernous sinus thrombosis, exemplifying the serious potential complications of endo-antral syndrome (EAS) [5, 24–26].
The clinical examination of a patient with suspected maxillary sinus disease should include extraoral tapping on the anterior and lateral walls of the sinus over the prominence of the cheekbones and/or palpation intraorally of the lateral surface of the maxilla between the canine fossa and the zygomatic buttress. If there is dental involvement, the teeth will be moderately or extremely sensitive to palpation and/or percussion. The pain typically radiates to all posterior teeth in the quadrant, so that all these teeth usually become tender to percussion [23].
Selden termed endo-antral syndrome (EAS) as the spread of pulpal disease beyond the confines of the dental supporting tissues into the maxillary sinus [27–29] and underlined five radiographic features characterizing EAS [29]:
1.
Pulpal disease in a tooth whose apex approximates the floor of the maxillary sinus
2.
Periapical radiolucencies on pulpally involved teeth
3.
Radiographic loss of the lamina dura defining the inferior border of the maxillary sinus over the pulpally involved tooth
4.
A faintly radiopaque mass bulging into the sinus space above the apex of the involved tooth, connected neither to the tooth nor to the lamina dura of the tooth socket (representing localized swelling and thickening of the sinus mucosa)
5.
Variable degrees of radiopacity of the surrounding sinus space (comparison to the contralateral sinus is often helpful)
Clinical cases do not always present with all five features; thus, diagnostic and therapeutic decisions may be challenging.
In cases with maxillary sinus pathologies with doubtful differential diagnosis or with severe signs and symptoms of sinusitis, consultation with an ear, nose, and throat (ENT) specialist is strictly recommended.
Therapeutic Decision Making
In cases of maxillary sinusitis following endodontic surgery, a pharmacological approach may be considered in the short term, but only in cases with clean serum exudate and without presence or migration of a bone graft in the sinus cavity [30]. The drug therapy consists of amoxicillin and clavulanic acid 1 gr TID and metronidazole 500 mg TID per os for 7–10 days or levofloxacin 400 mg BID per os for 7–10 days in patients allergic to penicillin. In cases of a massive sinus infection, with or without migration of a bone graft, after ENT consultation a surgical approach should be considered [30].
Endodontic Surgery and Sinus-Related Complications
Oberli et al. [17] evaluated a possible correlation between radiographic findings and presence of an oroantral communication (OAC). They analyzed the correlation between the positions of the root tip and the periapical lesion versus the sinus floor and the presence of an oroantral communication (OAC). While the perforation of the maxillary sinus during periapical surgery could not be predicted from the periapical radiographs, the following radiographic classification was proposed: (a) class I apices, where there is a distance between the root tip and the sinus floor; (b) class II apices, where the root tip touches the sinus floor; and (c) class III apices, where the root tip overlaps the sinus floor. The same classification was applied for the periapical lesions: (a) class I lesions, where there is a distance between the lesion and the sinus floor; (b) class II lesions, where the lesion touches the sinus floor; and (c) class III lesions, where the lesion overlaps the sinus floor.
In terms of clinical relevance, class I lesions show a high probability of leaving the sinus unharmed after periapical surgery, while for class III lesions (lesions seen inside the sinus cavity) perforation of the membrane may be unavoidable [17].
The outline of the lesions was described as either well defined or blurred. A well-defined outline means a sharp traceable radiopaque line. A blurred outline of the lesion means an unclear radiographic border of the lesion, which makes it impossible to trace the lesion accurately or to classify it.
The thin layer of bone covering the root is seen as a fusion of the lamina dura and the floor of the sinus [33]. In some cases with chronic periapical periodontitis, this line can be well defined and easy to trace, although in others the radiopaque line appears less defined or blurred.
A periapical radiograph may fail to show the lamina dura covering the root apex in areas with defective bony covering and it is not adequate in cases of large lesions. However, panoramic radiography provides an extensive overview of the sinus floor and its relationship with the teeth roots. It allows determination of the size of periapical lesions and cysts as well as radiodense foreign bodies [23]. But, both methods may not provide a clear visualization of the real dimensions and position of the lesion, due to intrinsic limitations.
Eberhardt et al. [20] stated that “standard dental radiographs, including panoramic radiograph and pluridirectional tomography, present a 2-dimensional image and as such are inadequate and/or impractical for precise morphometric assessment of osseous relationships. Because of the projection angle between the x-ray and the periapical film, it is difficult to detect whether a root tip overlapping the floor of the maxillary sinus is anatomically protruding into the maxillary sinus or is just being projected into it.”
The use of 3D radiographic techniques as computerized tomography (CT) and cone-beam CT should be considered in order to adequately diagnose the anatomical relationships between the lesion and the maxillary sinus, to prevent invasion into the sinus lumen during surgery, and to plan the surgical procedure and timing [17
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


