Introduction
In this in-vitro study, we aimed to compare the residual monomers in composites beneath brackets bonded to enamel, using a light-emitting diode (LED) or a halogen unit, and to compare the residual monomers in the central to the peripheral areas of the composite.
Methods
Twenty bovine teeth preserved in 0.1% thymol were used in this study. Ten teeth were used to standardize the thickness of the composite film, since different thicknesses would cause different absorbance of light. Brackets were bonded to 10 bovine incisors, with the halogen light (n = 5) and the LED (n = 5). The brackets were debonded, and the remaining composite on the enamel surface was sectioned in 2 regions: peripheral (0.8 mm) and central, resulting in 2 subgroups per group: central halogen (n = 5), peripheral halogen (n = 5), central LED (n = 5), and peripheral LED (n = 5). The spectrometric analysis in the infrared region was used to measure the free monomers with the attenuated total reflectance method.
Results
Normal distribution was tested by using the Kolmogorov-Smirnov test. Data were compared by 2-way analysis of variance (ANOVA) at P <0.05. The LED group showed fewer residual monomers than did the halogen group ( P = 0.014). No differences were found among the regions ( P = 0.354), and there were no interactions between light type and region ( P = 0.368).
Conclusions
LED leaves less residual monomer than does the halogen light, even with half of the irradiation time; there were no differences between the central and peripheral regions, and no interaction between light type and region.
The halogen light is the most commonly used source of polymerization of dental materials, with known efficiency. Halogen bulbs produce light when electric energy heats a small tungsten filament to a high temperature.
Mills proposed the use of light-emitting diode (LED) technology for the polymerization of light-initiated dental materials to overcome the shortcomings of halogen light-curing units. LEDs use junctions of doped semiconductors to generate light instead of the hot filaments in halogen bulbs. LEDs have a lifetime over 10,000 hours and undergoes little degradation of output over this time. LEDs require no filters to produce blue light, resists shock and vibration, and saves energy to operate. The longer lifespan of LEDs and more consistent light output compared with halogen bulb technology indicate that it should have a great use in dentistry.
For these reasons, LED technology is closer to the ideal of a light-curing unit, which in theory should have the following characteristics: light power of at least 300 mW per square centimeter, a narrow spectrum of light emitted (around the wavelength that camphorquinone reacts, with peak absorption at 468 nm), low heat generation, sterilized active point, and silent cooling fan or no fan.
The efficiency of a light-curing unit is closely related to its lighting potency: the higher the luminous density irradiated, the more free radicals involved in the light-curing process, and the more effective the cure of the composite. The increase in luminous intensity is the key to diminish irradiation time.
Although the shear bond strength test is more widely used and practical, evaluation of residual monomers through infrared spectroscopy is important in the results of physical test referents to dental materials. It is a sensitive method that identifies monomers in cured adhesive; monomers are responsible for increased bonding failures and can also cause adverse biologic effects, showing estrogenic characteristics.
The aims of this study were to quantify the residual monomers in the composite films beneath the brackets photo-initiated by LED and halogen lights, and to verify any difference in the polymerization of the central and the peripheral areas according to the light source used.
Material and methods
Twenty bovine teeth preserved in 0.1% thymol were used after approval by the Ethics Board of State University of Rio de Janeiro. Ten teeth were used to standardize the thickness of the composite film, since different thicknesses would allow different absorbance of light. The brackets were bonded by the same operator (F.A.R.C) without previous enamel conditioning to facilitate removal of the composite film. For debonding, the brackets were bent with pliers, and the films were removed from the dental surface with a surgical blade.
The thickness of each film was measured 3 times with digital calipers. The data were compared statistically by analysis of variance (ANOVA), and there were no statistically significant differences ( P = 0.702). Thus, it can be assumed that the thicknesses of the films were standardized.
The other 10 teeth were cleaned with a mixture of water and pumice in a rubber-polishing cup for 15 seconds, thoroughly rinsed with water for 15 seconds, and dried with an oil- and moisture-free airstream. Each tooth was etched with 37% phosphoric acid gel for 15 seconds, rinsed, and dried again.
Transbond XT (3M Unitek, Monrovia, Calif) primer was applied to each tooth, thinned with a gentle stream of air, and cured according to the manufacturer’s recommendations. All brackets were central incisor metal brackets, Edgewise Standard (Morelli, Sorocaba, São Paulo, Brazil) because they have plan bases and are larger than the others, resulting in a longer path for the light to the center of the base, simulating a critical clinic situation. The brackets were bonded to the enamel by using Transbond XT adhesive paste (3M Unitek).
The sample was divided into 2 groups of 5 teeth according to the type of light used to photo-initiate the polymerization process. The procedures for the halogen group were 20 seconds of light exposure 10 seconds at each proximal surface with the Ortholux XT Visible Light Curing Unit (3M Unitek) with light intensity of 400 mW per square centimeter. The procedures for the LED group were 10 seconds of light exposure, 5 seconds in each proximal surface, with the Ortholux LED Curing Light (3M Unitek) with light intensity of 800 mW per square centimeter.
The bonding procedures were performed in the same environment to prevent different luminous intensities that could interfere with the results. The distance of the curing light to the bracket was minimal; once the tooth was isolated, it allowed close positioning of the active point to the bracket-tooth interface. A surge protector was used to prevent input energy variations. The light intensity was measured before and after the experiments to determine whether there was any change, by using an Optilux Radiometer (SDS, Kerr, Danbury, Conn) that measures the energy emitted between 400 and 500 nm.
The samples were identified by group and numbered in the sequence of bonding. They were debonded in the same sequence, minimizing the discrepancy between the bonding and debonding times to standardize the residual polymerization time of the samples. After bonding, the samples were stored in black plastic containers to prevent light and additional irradiation on the composite films. The containers were stored for 24 hours in an incubator at 37°C to simulate the temperature of the human mouth.
Polymerized composite films were obtained by removing the brackets by their deformation with pliers and preserving the whole film on the dental surface ( Fig 1 ). Digital calipers were used to mark margins of 0.8 mm in the composite, scratching the film with 1 active point and maintaining the other in the external margin of the composite.
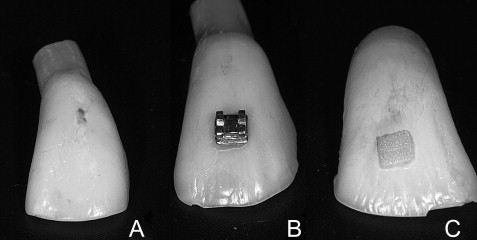
Segmentation of the composite was performed through the marked margins with a low-speed diamond bur, making 2 subgroups from each group: central region of the photo-initiated films with the halogen light (CH), peripheral region of the photo-initiated films with the halogen light (PH), central region of the photo-initiated films with the LED (CL), and peripheral region of the photo-initiated films with the LED (PL).
The composite films of each subgroup were mashed into a fine powder, which was weighted by a precision scale to separate an amount between 0.0011 and 0.0012 g from each subgroup.
Infrared spectroscopy was used with the attenuated total reflectance technique. The obtained dust was analyzed in the center of the crystal of the universal accessory for attenuated total reflectance samples, which is part of the Spectrum appliance (PerkinElmer, Salem, Mass). The samples were evaluated in the region of 650 to 4000 cm −1 for 32 scans, with a resolution of 4 cm −1 and pressed by a pistol with a constant tension.
The monomers of Transbond XT are the bisphenol-A-glycidyl methacrylate (Bis-GMA) and the triethylene glycol dimethacrylate (TEGDMA). Both show double bonds in the extremes of the monomer molecule. The unsaturation generates an absorbance of energy from the infrared beam in the region of wave number 1640 cm −1 , so that with greater band intensity, more unsaturation and more monomers are present. In this study, the spectra were generated according to the transmittance (%T), so that with higher values, fewer monomers were present.
Statistical analysis
The Kolmogorov-Smirnov test was used to verify the normal distribution of the sample. To check for statistical differences among the results, 2-way ANOVA was used, with region as the dependent factor and light type as the independent factor. A significance level of P ≤0.05 was considered.
Results
The mean values of band intensity in the wave number 1640 cm −1 were 98.76 ± 0.89 for the CH group, 97.92 ± 0.54 for the PH group, 99.23 ± 0.99 for the CL group, and 99.22 ± 0.49 for the PL group. The statistical analysis showed no significant difference among the central and peripheral regions ( P = 0.354). There was a significant difference between light types; the LED group had less residual monomer than did the halogen group ( P = 0.014). The interaction between light type and region was not statistically significant ( P = 0.368). Mean values and standard deviations are shown in Table I . Table II shows the 2-way ANOVA values, and Figure 2 shows data dispersion.
| CH | PH | CL | PL | |
|---|---|---|---|---|
| Mean value | 98.76 | 97.92 | 99.23 | 99.22 |
| SD | 0.89 | 0.54 | 0.99 | 0.49 |
| Source | Type III sum of squares | df | Mean square | F | P |
|---|---|---|---|---|---|
| Tests of within-subjects effects | |||||
| Region | 0.761 | 1 | 0.761 | 0.969 | 0.354 |
| Region∗ group | 0.714 | 1 | 0.714 | 0.910 | 0.368 |
| Error (region) | 6.279 | 8 | 0.785 | ||
| Tests of between-subjects effects | |||||
| Intercept | 195244.608 | 1 | 195244.608 | 527527.734 | 0.000 |
| Group | 3.664 | 1 | 3.664 | 9.899 | 0.014 |
| Error | 2.961 | 8 | 0.370 | ||
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


