Introduction
The aim of this study was to analyze the current literature for the best evidence (randomized clinical trials) about the efficacy of functional appliances on mandibular growth in the short term.
Methods
A survey of articles published up to September 2009 was performed by using the following electronic databases: PubMed, Embase, Ovid Medline, Cochrane Central Register of Controlled Trials, Web of Science, LILACS, and Google Scholar. The reference lists of the retrieved articles were hand-searched for possible missing articles. No language restriction was applied during the identification of the published studies. A methodologic scoring process was developed to identify which randomized clinical trials were stronger methodologically. The selection process and the quality assessment were undertaken independently and in duplicate by 2 authors. A meta-analysis was attempted by using random-effects models. Clinical and statistical heterogeneity was examined, and a sensitivity analysis was performed.
Results
Electronic searches identified the following items: 146 articles were retrieved from PubMed, 45 from Cochrane Central Register of Controlled Trials, 29 from Ovid, 42 from LILACS, 628 from Web of Science, and 1000 from Google Scholar. Thirty-two articles fulfilled the specific inclusion criteria and were identified as potentially appropriate randomized clinical trials to be included in this meta-analysis. Only 4 articles, based on data from 338 patients (168 treated vs 170 controls) with Class II malocclusion in the mixed dentition, were selected for the final analysis. The quality analysis of these studies showed that the statistical methods were at the medium-high level. The outcome measurements chosen to evaluate the efficacy of the various functional appliances were Co-Pg, Pg/Olp + Co/Olp, and Co-Gn and the values were annualized and standardized to a uniform scale with the standardized mean differences (SMD). The results of the meta-analysis from the random-effects model showed a statistically significant difference of 1.79 mm in annual mandibular growth of the treatment group compared with the control group (SMD = 0.61, 95% CI, 0.30 to –0.93; chi-square test, 5.34; 3 df; P = 0.15; I 2 = 43.9%; test for overall effect, Z = 3.83 and P = 0.0001). The sensitivity analysis showed a substantially similar outcome of 1.91 mm (SMD = 0.65, 95% CI, 0.25 to 1.25; chi-square test, 4.96; 2 df; P = 0.08; I 2 = 59.7%; test for overall effect, Z = 3.19 and P = 0.001).
Conclusions
The analysis of the effect of treatment with functional appliances vs an untreated control group showed that skeletal changes were statistically significant, but unlikely to be clinically significant.
One of the most controversial topics in orthodontics relates to the effectiveness of functional appliances on mandibular growth. In skeletal Class II malocclusion, mandibular retrusion seems to be a major contributing factor; it occurs in about one third of the population. Functional appliances encompass a range of removable and fixed devices that are designed to alter the position of the mandible, both sagittally and vertically, to induce supplementary lengthening of the mandible by stimulating increased growth at the condylar cartilage.
Experiments have demonstrated that appliances that position the mandible anteriorly stimulate significant mandibular growth by condyle remodeling in animal models, but the effects produced in humans are not the same. Evidence shows that favorable growth responses are not always achieved with functional therapy; some authors reported increases in overall mandibular length and changes in the amount of condylar growth, but others believe that mandibular length cannot be altered by such therapy. It has been claimed that most of the correction of the malocclusion is due to dentoalveolar changes with a small but statistically significant amount of skeletal effects.
There are also controversies concerning the effects of functional appliances on the maxilla. Many studies indicate that forward growth of the maxilla might be inhibited, but other authors stated that there is no appreciable effect on the position of the maxilla.
All of these conflicting claims could be attributable to retrospective study designs or methodologic limitations such as small samples, inadequate or no control group, dishomogeneity of the groups for sex and age at the start of therapy, and different lengths of treatment.
In addition, there is a lack of long-term studies on the effect of functional appliances to evaluate the stability of skeletal changes because of the great difficulties in recruiting patients after treatment.
The aim of this study was to analyze results from randomized controlled trials (RCTs) in the literature concerning Class II functional therapy to evaluate the efficacy of functional appliances on mandibular growth.
Material and methods
A survey of articles published up to September 2009 about the effects of functional appliances on mandibular growth was performed by using several electronic databases: PubMed, Ovid (Ovid Medline and Embase), Cochrane Central Register of Controlled Trials, Web of Science, LILACS, and Google Scholar. All electronic searches were conducted on September 30, 2009. The search strategy for PubMed was conducted according to Cochrane Collaboration guidelines ( Table I ). The keywords used to identify the corresponding studies in the other databases were Class II malocclusion and orthodontic functional applianc∗ . A more selective search strategy for Google Scholar was performed with the following terms: Class II malocclusion , orthodontic functional appliances , and randomized clinical trial . Electronic search results are given in detail in Table II . The reference lists of the retrieved articles were also hand-searched to identify any articles to be included in this evaluation that might have been missed in the electronic bibliographic databases. No language restriction was applied during the identification process of published studies.
| Keywords | Results | |
|---|---|---|
| (1) | Randomized controlled trial | 335,072 |
| (2) | Randomized controlled trials | 338,843 |
| (3) | Random allocation | 66,497 |
| (4) | Double blind | 123,934 |
| (5) | Double blind method | 103,271 |
| (6) | Single blind | 29,535 |
| (7) | Single blind method | 24,124 |
| (8) | #1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 | 427,734 |
| (9) | Class II malocclusion | 4387 |
| (10) | Orthodontic functional appliances | 1599 |
| (11) | #9 OR #10 | 5386 |
| (12) | #8 AND #11 | 169 |
| (13) | #8 AND #11, limit: humans | 146 |
| Database | Keywords | Results | Not related to the topic or related with different aim | No RCTs | Selected |
|---|---|---|---|---|---|
| PubMed | Search history as described by Table I | 146 | 102 | 16 | 28 |
| Cochrane Central Register of Controlled Trials | (1) Orthodontic functional appliances; (2) Class II malocclusion; #1 AND #2; | 45 | 12 | 32 | 1 |
| Ovid | (1) Orthodontic functional appliances; (2) Class II malocclusion; #1 AND #2; | 29 | 11 | 17 | 1 |
| Web of Science | (1) Orthodontic functional applianc*; (2) Class II malocclusion; #1 AND #2; | 628 | 412 | 198 | 18 |
| Google Scholar | (1) Orthodontic functional applianc*; (2) Class II malocclusion; (3) randomized clinical trials; #1 AND #2 AND #3; | 1000 | 883 | 95 | 22 |
| LILACS | (1) Orthodontic functional applianc*; (2) Class II malocclusion; #1 AND #2; | 42 | 10 | 32 | 0 |
| Hand search | Bibliographies of RCTs, RCTs known to the authors before this study, and RCTs encountered during searches for other projects | 1 | 1 |
Selection criteria
To be included in our study, each article had to fulfil the following requirements: (1) related human clinical trials; (2) included a randomized selection of the samples; (3) concerned with functional appliances in the therapy of Class II malocclusion; (4) had a comparable untreated control group; (5) analyzed treatment effects not confounded by additional and concomitant treatments (headgear, extractions, or fixed appliances); (6) used cephalometric analysis at the start of treatment and just after removal of the functional appliances; and (7) concerned with mandibular anteroposterior changes measured by using the anatomic condylion.
Articles were not selected if they did not meet the inclusion criteria, if they did not relate to this topic, or if they related but had a different aim. Abstracts, laboratory studies, descriptive studies, individual case reports, series of cases, reviews, studies of adult patients, controlled clinical trials, retrospective longitudinal studies, and meta-analyses were excluded. Articles reporting interim outcomes or updates were considered only once because they related to the same samples of subjects. RCTs including patients who had received previous or concomitant treatment for their Class II malocclusion were also excluded.
Data abstraction
The selection process was done by 2 independent reviewers (E.M. and M.B.) using separate printed forms. The data extracted from each article were compared and discussed to resolve any discrepancies and to reach unanimous consensus. Cohen’s kappa score was calculated to determine interrater reliability.
Validity assessment
Because high-quality methodologic studies can provide more reliable conclusions, the current Cochrane guidelines for assessing quality were adopted to evaluate the risk of bias of the trials included. The quality assessment was undertaken independently and in duplicate by using separate printed forms by the 2 reviewers, and agreement was assessed with the kappa statistic.
Study characteristics
All selected RCTs related to orthodontic functional therapy in children during the transitional dentition stage of development with Class II malocclusion. From the selected articles, the following study characteristics were recorded: sample size, type of functional appliance, sex and age, time of treatment or observation, time of daily appliance wear, cephalometric measurements of outcomes, and follow-up. Clinical heterogeneity was assessed by examining the types of participants and the interventions for the outcome in each study.
Quantitative data synthesis
The effective increase of total mandibular length was the outcome chosen to evaluate the efficacy of the various functional appliances. Cephalometric reference points and linear measurements of overall mandibular length recorded from each selected study were condylion-pogonion (Co-Pg), condylion-gnathion (Co-Gn), and pogonion-occlusal line perpendiculare + condylion-occlusal line perpendiculare (Pg/Olp + Co/OLp).
Studies that used articulare were excluded because this end-point landmark does not measure the real mandibular increase. We did not consider angular measurements because they do not evaluate effective mandibular growth. The mean differences and standard deviations of the continuous outcomes were used to summarize the data for each group.
The outcome values were standardized to a uniform scale with standardized mean differences (SMD) and were combined by using random-effects models. When studies use different measurements to evaluate the same outcome, SMD (difference in mean outcome between groups or standard deviations of outcomes among participants) can be used in a meta-analysis for combining continuous data.
The significance of any discrepancies in the estimates of the treatment effects from the RCTs was assessed with the Cochrane test for heterogeneity and the I 2 statistic. We performed a sensitivity analysis to examine the effect of the study quality assessment on the overall estimates of effect.
Results
Electronic searches identified the following items: 146 articles from PubMed, 45 from Cochrane Central Register of Controlled Trials, 29 from Ovid, 42 from LILACS, 628 from Web of Science, and 1000 from Google Scholar. Articles that did not relate to our topic or related with a different aim, and those that were not RCTs, were excluded, as shown in Table II .
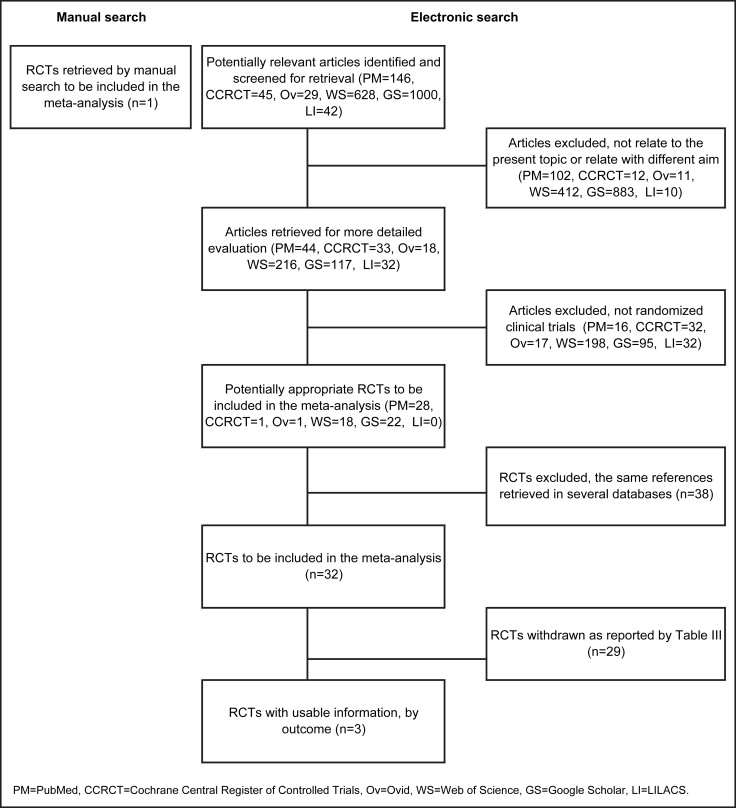
Of the remaining potentially appropriate RCTs, 38 were duplicates, so 32 were identified as eligible RCTs to be included in this study ( Fig 1 ). In detail, 6 studies were retrieved from PubMed; 4 from Google Scholar; 4 from both PubMed and Google Scholar; 4 from PubMed and Web of Science; 12 from PubMed, Google Scholar, and Web of Science; 1 from PubMed, Google Scholar, Web of Science, and Ovid; and 1 from PubMed, Google Scholar, Web of Science, and Cochrane Central Register of Controlled Trials ( Table III ).
| Reference | Databases | Study | Reason for exclusion |
|---|---|---|---|
| Web of Science, PubMed | Webster T et al. Associations between changes in selected facial dimensions and the outcome of orthodontic treatment. Am J Orthod Dentofacial Orthop 1996;110:46-53. | Reported interim outcomes or updates | |
| PubMed | Cura N, Saraç M. The effect of treatment with the Bass appliance on skeletal Class II malocclusions: a cephalometric investigation. Eur J Orthod 1997;19:691-702. | Concomitant use of headgear with Bass appliance; cephalometric analysis that did not use condylion | |
| PubMed | Mao J, Zhao H. The correction of Class II, division 1 malocclusion with bionator headgear combination appliance. J Tongji Med Univ 1997;17:254-6. | Concomitant use of headgear with bionator appliance | |
| Web of Science, PubMed | Tulloch JFC et al. Influences on the outcome of early treatment for Class II malocclusion. Am J Orthod Dentofacial Orthop 1997;111:533-42. | Related to the present topic but different aim | |
| Google Scholar, PubMed | Keeling SD et al. Anteroposterior skeletal and dental changes after early Class II treatment with bionators and headgear. Am J Orthod Dentofacial Orthop 1998;113:40-50. | Patients received previous treatment | |
| Google Scholar, Web of Science, PubMed | Ghafari J et al. Headgear versus function regulator in the early treatment of Class II division 1 malocclusion: a randomized clinical trial. Am J Orthod Dentofacial Orthop 1998;113:51-61. | No untreated control group | |
| Google Scholar, Web of Science, PubMed | Tulloch JFC et al. Benefit of early Class II treatment: progress report of a two-phase randomized clinical trial. Am J Orthod Dentofacial Orthop 1998;113:62-72. | Reported interim outcomes or updates | |
| Google scholar, Web of Science, PubMed | Illing HM et al. A prospective evaluation of Bass, bionator and twin block appliances. Part I—the hard tissues. Eur J Orthod 1998;20:501-16. | Lack of control group’s randomization | |
| Google scholar, Web of Science, PubMed, Cochrane | Ehmer U et al. An international comparison of early treatment of angle Class-II/1 cases. Skeletal effects of the first phase of a prospective clinical trial. J Orofac Orthop 1999;60:392-408. | Lack of control group’s randomization: the control sample is the same as in Tulloch et al | |
| PubMed | Wheeler TT et al. Effectiveness of early treatment of Class II malocclusion. Am J Orthod Dentofacial Orthop 2002;121:9-17. | Reported interim outcomes or updates | |
| Web of Science, PubMed | Cevidanes LH et al. Clinical outcomes of Fränkel appliance therapy assessed with a counterpart analysis.Am J Orthod Dentofacial Orthop 2003;123:379-87. | Cephalometric analysis did not use condylion | |
| PubMed | King GJ et al. Comparison of peer assessment ratings (PAR) from 1-phase and 2-phase treatment protocols for Class II malocclusions. Am J Orthod Dentofacial Orthop 2003;123:489-96. | No untreated control group | |
| Google Scholar, Web of Science, PubMed | O’Brien K et al. Effectiveness of treatment for Class II malocclusion with the Herbst or twin-block appliances: a randomized, controlled trial. Am J Orthod Dentofacial Orthop 2003;124:128-37. | No untreated control group | |
| Google Scholar, PubMed | Araujo AM et al. Adaptive condylar growth and mandibular remodelling changes with bionator therapy—an implant study. Eur J Orthod 2004;26:515-22. | Cephalometric analysis did not use condylion | |
| Google Scholar | Huang G. Twin-block appliance is effective for the correction of Class II Division I malocclusion during mixed dentition. J Evid Based Dent Pract 2004;4:222-3. | Summary trial of O’Brien et al | |
| PubMed | Banks P et al. Incremental versus maximum bite advancement during twin-block therapy: a randomized controlled clinical trial. Am J Orthod Dentofacial Orthop 2004;126:583-8. | No untreated control group | |
| Google Scholar, Web of Science, PubMed | Tulloch JF et al. Outcomes in a 2-phase randomized clinical trial of early Class II treatment. Am J Orthod Dentofacial Orthop 2004;125:657-67. | Reported interim outcomes or updates | |
| Google Scholar | Kalha A. Early treatment with the twin-block appliance is effective in reducing overjet and severity of malocclusion. Is the twin-block orthodontic appliance effective in early treatment of developing class II division 1 malocclusion? Evid Based Dent 2004;5:102-3. | Summary trial of O’ Brien et al | |
| Web of Science, PubMed | Gill DS et al. Prospective clinical trial comparing the effects of conventional Twin-block and mini-block appliances: part 1. Hard-tissue changes. Am J Orthod Dentofacial Orthop 2005;127:465-72. | No untreated control group | |
| Google scholar, Web of Science, PubMed. | Cevidanes LH et al. Assessment of mandibular growth and response to orthopedic treatment with 3-dimensional magnetic resonance images. Am J Orthod Dentofacial Orthop 2005;128:16-26. | Magnetic resonance imaging study, no cephalometric analysis | |
| Google Scholar, Web of Science, PubMed | Cevidanes LH et al. Comparison of relative mandibular growth vectors with high-resolution 3-dimensional imaging. Am J Orthod Dentofacial Orthop 2005;128:27-34. | Magnetic resonance imaging study, no cephalometric analysis | |
| Google Scholar, Web of Science, PubMed | Efstratiadis S et al. Evaluation of Class II treatment by cephalometric regional superpositions versus conventional measurements. Am J Orthod Dentofacial Orthop 2005;128:607-18. | No untreated control group | |
| Google Scholar | Liu Ji-hui. A comparative study of clinical effects of Twin-Block and strigth wire appliance combined with cervical headgear and class II elastic on the treatment of class II, division I malocclusion. Chinese Journal of Traditional & Western Medicine 2006;7:1925-9. | No untreated control group | |
| PubMed | Karacay S et al. Forsus nitinol flat spring and Jasper jumper corrections of Class II division 1 malocclusions. Angle Orthod. 2006;76:666-72. | Concomitant use of fixed edgwise appliances | |
| Google Scholar, PubMed | Lee RT et al. A controlled clinical trial of the effects of the Twin Block and Dynamax appliances on the hard and soft tissues. Eur J Orthod 2007;29:272-82. | No untreated control group | |
| Google scholar, Web of Science, PubMed, Ovid Medline | Dolce C et al. Timing of Class II treatment: skeletal changes comparing 1-phase and 2-phase treatment. Am J Orthod Dentofacial Orthop 2007;132:481-9; erratum in Am J Orthod Dentofacial Orthop 2007;132:727. | Reported interim outcomes or updates | |
| Google Scholar | Ren Y. Very few indications justify early treatment for severe Class II malocclusions. Evid Based Dent 2004;5:100-1. | Summary trial of Tulloch et al | |
| Google scholar, Web of Science, PubMed | O’Brien K et al. Early treatment for Class II Division 1 malocclusion with the Twin-block appliance: a multi-center, randomized, controlled trial. Am J Orthod Dentofacial Orthop 2009;135:573-9. | Reported interim outcomes or updates | |
| Google Scholar, PubMed | Baccetti T et al. Comparison of 2 comprehensive Class II treatment protocols including the bonded Herbst and headgear appliances: a double-blind study of consecutively treated patients at puberty. Am J Orthod Dentofacial Orthop 2009;135:698.e1-10. | No untreated control group |
Twenty-nine of the 32 RCTs were withdrawn according to the following exclusion reasons: (1) lack of an untreated control group ; (2) lack of random allocation of the untreated control group ; (3) lack of cephalometric analysis ; (4) cephalometric analysis that did not use anatomic condylion ; (5) simultaneous use of additional treatments ; (6) progress reports ; (7) summary trials ; or (8) related to this topic but with a different aim. The reasons for excluding these studies are given in detail in Table III . Only 3 articles met all eligibility criteria and were selected for the final analysis. One more RCT with available outcomes, identified by hand-searching, was included.
Study characteristics
We collected data from 338 patients (168 treated vs 170 controls) with Class II malocclusion in the mixed dentition. Two trials included only subjects with severe Class II malocclusion and without clinically obvious facial asymmetry. The samples were heterogeneous for the numbers and the ages of participants.
The numbers of treated subjects and controls ranged from 17 to 73 and from 17 to 74, respectively; the mean ages of 2 studies was homogeneous at 9.57 years, whereas the others reported mean ages of 8.5 and 11.6 years. Only O’Brien et al and Tulloch et al detected an effective level of skeletal maturity through the evaluation of cervical spine and hand-wrist radiographs, respectively.
It was not possible to assess clinical sex heterogeneity because, in 2 trials, data were not available about the sexes of the samples. Strong heterogeneity was found in time of daily appliance wear, probably due to the different functional appliances tested. Every selected RCT analyzed a different type of functional appliance (activator, bionator, Twin-block, Frankel 2, Harvold activator), but they had similar mechanisms of action. Treatment or observation times ranged from 15 months to 18 months. No article reported long-term results.
Considerable variability in the outcome measurements (Co-Pg, Co-Gn, and Pg/Olp + Co/OLp ) was seen because of the many different types of cephalometric analyses adopted by the investigators. The data of the selected articles are summarized in Table IV .
| Selected references | Sample and appliances | Sex | Age (y) | Setting | Treatment or observation time | Time of daily appliance wear | Methods of meassurement | Follow-up |
|---|---|---|---|---|---|---|---|---|
| Jakobsson | 17 act | NA | 8.5 | Karolinska Institutet | 18 mo | 11.5 h/d | Co-Pg | No |
| 1967 | 18 contr | 8.5 | (Sweden) | 18 mo | ||||
| Nelson et al | 12 HA | 7 M, 5 F | 10-12.9 | University of Otago | 18 mo | Minimum of 14 h | Cephalometric analysis, | No |
| 1993 | 13 FR-2 | 7 M, 6 F | Mean, 11.6 | (New Zealand) | 18 mo | Co-Go, Go-Pg, Co-Pg | ||
| 17 contr | 11 M, 6 F | 18 mo | ||||||
| Tulloch et al | 53 bio | 30 M, 23 F | 9.4 | University of | 15 mo | Not | Cephalometric analysis, | No |
| 1997 | 61 contr | 35 M, 26 F | 9.4 | North Carolina | 15 mo | declared | Co-Gn, hand-wrist radiographs | |
| O’ Brien et al | 73 TB | NA | Mean, 9.7 | National Health Service | 15 mo | 24 h/d | Cephalometric analysis, | No |
| 2003 | 74 contr | Mean, 9.8 | (United Kingdom) | 15 mo | excluding contact sports and swimming | Pg/Olp + Co/OLp, stage of maturation of cervical spine analysis |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


