Introduction
The purpose of this research was to study the influence of soft laser treatment on the process of bone repair after expansion of the midpalatal suture.
Methods
The sample for this case-control experimental study was 11 dogs. They were randomly divided into 2 groups, both of which underwent rapid maxillary expansion with a hyrax appliance. The animals in group 1 were also treated with laser therapy. They were killed, and histologic specimens of the palatal suture were prepared. The Student t test was applied for independent data, and the Mann-Whitney test was used for nonparametric data.
Results
A significant difference was observed in the quality of the palatal sutures between the animals in groups 1 and 2. The connective tissues of the sutures in the group 1 animals were similar to the original configurations, with more advanced osteogenesis and fibrogenesis, compared with those of group 2.
Conclusions
Soft laser appears to influence the behavior of the repair process, contributing to suture reorganization and palatal bone osteogenesis during and after expansion.
Palatal expansion, or rapid maxillary expansion, is a technique adopted by orthodontists to treat malocclusions with real or relative transverse maxillary deficiency. The technique is performed under clinical conditions by using expansion appliances or by surgical intervention. The procedure includes an active phase with expansion appliances to disarticulate the jaw bones by widening the palatal suture, and a passive phase, in which a series of procedures leads to bone remodeling and restoration of the suture, with a clinical increase in the width of the palate. The use of expanders requires retention for 3 to 4 months to allow adequate reossification, so that the effects are stabilized. The palatal expander is indicated for these situations: real and relative maxillary deficiency; severe nasal stenosis; Class III surgical, nonsurgical, and pseudo Class III; cleft palates in adults; certain patients with problems of arch length, seeking to avoid treatment protocols involving extractions that could affect the facial profile; and situations in which the beneficial side effects of displacement of the jaw are desirable to increase the vertical dimensions in cases of exaggerated overbite.
Soft laser is a form of highly concentrated nonionizing radiation that, when in contact with different tissues and depending on the type, results in thermal, photochemical, and nonlinear effects. As a nonionizing form of energy, laser radiation is noninvasive and well tolerated by the tissues; it does not have mutagenic effects, and it can be applied repeatedly, without risks for the patient. Low-level lasers have predominantly analgesic, anti-inflammatory, and biomodulatory effects and promote an increase in local microcirculation and speed of wound healing.
In the orthodontic literature, a few reports have shown the application of soft lasers in the midpalatal suture in connection with expansion. Therefore, in this study, we sought to assess whether low-level lasers promote changes in the repair after the orthopedic separation of the midpalatal suture.
Material and methods
For this experimental study, small dogs ( Canis lupus familiaris ) of various breeds were used that met the following inclusion criteria: clinically healthy, 6 months of age, and in the permanent dentition phase at the start of the experiment. The animals were obtained from a local veterinary center, according to local availability, comprising a convenience sample. The principles of animal experimentation were applied, in accordance with the guidelines of the Brazilian College of Animal Experimentation, and the study was approved by the research ethics committee of the University of Marília in Brazil.
During the study period, 11 dogs (9 males, 2 females) were studied and divided into 3 groups: 2 experimental groups and 1 animal as the control. Experimental group 1 consisted of 5 animals that received 20 applications of laser therapy every 48 hours, during and after expansion. Thus, the laser applications amounted to a period of 39 days. On day 40, the dogs were killed. Experimental group 2 consisted of 5 animals that underwent maxillary expansion without laser treatment. In both groups, the expansion was applied for 7 days. One animal was used as control, to determine the histologic aspects of the unmodified suture. It did not undergo any procedure but was kept under the same conditions as the other animals. They were kept in private kennels, they were free to roam and interact with each other, they were separated only from other dogs that were not part of the study, and they all received the same feed.
The clinical procedures performed were painless, but the animals were sedated with 1% acepromazine intradermally, at a dose of 0.1 mg per kilogram, and ketamine intramuscularly, at a dose of 2 mg per kilogram, to ensure better immobilization.
To make the mold, first, an individual tray for each dog was produced; it was then filled with irreversible hydrocolloid and placed in the animal’s mouth, adjusting it to the edges of the maxillary dental arch. Once the material had solidified, the mold was removed and then cast in plaster to obtain a working model.
The trimmed plaster model was then sent to the prosthetist to make the metal crowns that would fit in the canines and premolars of the animals in the experimental groups. Next, the tooth-borne hyrax-type (Dentaurum, Ispringen, Germany) expansion device was created and cemented by using zinc phosphate cement (ZCI zinc cement; SS White, Gloucester, United Kingdom). Glacier photopolymerized resin (SDI, Victoria, Australia) reinforcement was then added to the first, second, and third premolars, providing greater fixation of the device ( Fig 1 ).
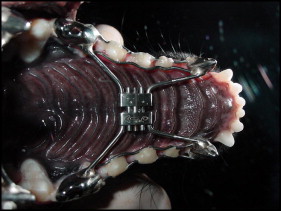
After fixation, the expansion appliance was activated. The activation of the screw expander followed the protocol adopted by the specialization course in orthodontics at the University of Marília, where the study was carried out. Over the first 3 days, 2 quarter turns were made in the morning and 2 quarter turns in the afternoon. Over the next 4 days, this was reduced to 1 quarter turn in the morning and 1 in the afternoon. The activation procedures were the same for both groups 1 and 2, differing only in the application of laser therapy in group 1.
For the laser application in group 1, the Photon Lase III device (DMC Equipamentos, São Carlos, Brazil) was used, with energy density of 90 to 120 J per square centimeter and a wavelength of 790 to 904 nm. The application sites were 4 points, distributed bilaterally and parallel to the midpalatal suture. Initially, the laser was applied concomitantly with the expansion activation, and then the laser was applied at intervals of 48 hours over 39 days. There was no need to sedate the animals during these procedures.
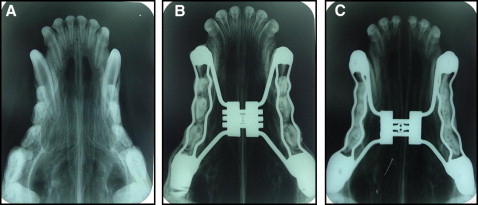
All animals underwent x-rays in the occlusal view before and after the placement of the device and also in the postexpansion period ( Fig 2 ). In the experimental groups, widening of the suture was observed, confirmed by occlusal x-rays after installation of the device ( Fig 2 , C ). A transverse maxillary increase was observed clinically in the experimental animals, as well as a diastema between the 2 central incisors.
Histologic sections
The animals were killed by the same veterinarian, after the administration of preanesthesia medication of acepromazine 1% intramuscularly at a dose of 0.5 mL per kilogram of the animal’s weight. Subsequently, dissociative anesthesia with 1 mL per kilogram of 10% ketamine was administered intramuscularly and finally 5 mL per kilogram of 19.1% potassium chloride intravenously or intracardially. The whole procedure was conducted without causing any suffering to the animals.
After death, the head and trunk were separated by using a band saw. Subsequently, the jaw and the maxilla were separated by cutting the skull in half. A manual scalpel, surgical scissors, and rat-tooth forceps were used to remove the soft tissues (muscles, tendons, eyes, tongue, ligaments), preserving the palate, gums, bones, and teeth. The anatomic piece obtained was cleaned by rinsing under running water. After this process, the piece was immersed in hydrogen peroxide (10 volumes) for about 5 minutes and again rinsed with water. The piece was then placed in a 10% formaldehyde solution. The anatomic pieces were dissected, and the maxilla was delimited by cuts, including the anterior palate, close to the palatal suture, where the laser was applied. The anatomic piece collected was placed in a decalcifying solution containing the following compounds: 0.7 mL nitric acid, 0.5 mL formaldehyde, and 60 mL water. The sections were embedded in paraffin, sectioned and stained with Masson’s trichrome, hematoxylin and eosin, and toluidine blue, and examined under a light microscope.
Histologic evaluation
Osteoblasts were counted in both groups by an author (V.C.C.E.S.), along the transition line between the suture and the newly formed bone, and immediately recounted by another author (A.P.), blind to the first evaluation. For these evaluations, the researchers used a microscope (BX50; Olympus, Center Valley, Pa) with magnification of 400 times, coupled with a 400-dot reticle eyepiece with 62,500-μm squares. The counts were compared by using the Student t test for independent data.
Histologic evaluations were performed by using a semiquantitative method based on knowledge of the aspects related to the normal characteristics of the palatal suture and the findings of embryologic and experimental studies. A system for scoring the degree of suture and palatine bone reorganization was proposed by using 4 histologic variables. The first variable scored between 1 and 4, and the others scored between 1 and 3, as shown in Table I . The sums of each scored histologic variable allow a number from 4 to 13, which provides a gradation of the histologic maturation process, with 4 corresponding to the initial phase of the tissue reorganization of the suture and the adjacent palatine bone after expansion, and 13 characterizing a completely restructured suture, resembling the original configuration. The slides were analyzed by comparing the control animal with those of the study groups.
| Variable | Score | Description |
|---|---|---|
| Connective tissue of the suture | 1 | Loose |
| 2 | Dense with perpendicular fibers | |
| 3 | Dense with fibers in various directions | |
| 4 | Dense with fibers arranged in bundles | |
| Blood vessels of the suture | 1 | Large disorganized vessels |
| 2 | Small vessels, irregularly distributed | |
| 3 | Small vessels, evenly distributed | |
| Cells in the suture | 1 | Many osteoblasts and fibroblasts |
| 2 | Few osteoblasts and fibroblasts | |
| 3 | Mature-looking cells | |
| Palatal bone tissues at edges of the suture | 1 | Bone tissue in formation, with irregular trabeculae bordering large medullary spaces |
| 2 | Bone tissue in formation, with regular trabeculae bordering small medullary spaces | |
| 3 | Mature-looking bone |
Step sections of the material stained by Masson’s trichrome, hematoxylin and eosin, and toluidine blue were reviewed by 3 evaluators (V.C.C.E.S., A.P., and a pathologist), who agreed on terms and definitions. The same raters reviewed and scored the slides again later. All raters were blinded to the study groups, each other’s results, and the previous round of scoring.
The agreement within each rater (interrater reliability) for the 4 histologic variables was calculated by using the Cohen kappa and the related Fleiss kappa. Raters’ error analysis was determined through the construction of median confidence intervals by using the nonparametric Wilcoxon signed rank test. The groups were compared with the Mann-Whitney test for nonparametric data by using a 95% confidence level. The statistical software used was Minitab (version 14; Minitab Inc, State College, Pa).
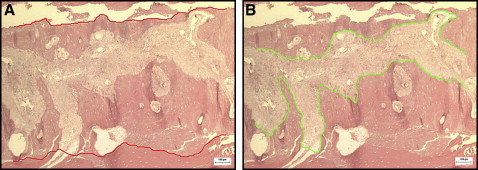
Digital photographs of the slides were obtained with 40-times magnification, and 2 lines were drawn on each image following the contours of the newly formed bone and the palatal suture. The widths of the suture and the newly formed bone were measured to assess whether the laser influenced the actual expansion of the suture. For this purpose, the “thickness” measurement tool of the application Image-Pro Plus (version 4.5.0.29; Media Cybernetics Inc, Rockville, Md) was used to measure the thickness between the 2 previously drawn lines, automatically calculating the maximum, minimum, and average distances ( Fig 3 ).
Statistical analysis
Statistical analysis comparing the average scores for the width of the suture and the newly formed bone was performed by using the Student t test for independent data with a significance level of P <0.05.
Results
The histologic study of the palatal sutures of the animals in groups 1 and 2, when compared with the control animal, showed widening of the suture, and the connective tissue had become looser than its original configuration. A rich vascularization was observed in the area of the suture ( Fig 4 ). The connective tissues surrounding the suture in the group 1 animals were more similar to the configuration of the unexpanded suture, with fibroblasts and collagen fibers that tended to form bundles in various directions, although this tissue was less dense than in the original suture. The vessels were more evenly distributed and more regular in size, similar to the vessels of the unexpanded suture ( Fig 5 ). In the connective tissue of the suture, a predominance of dense connective tissue was observed in group 1, with fibers in various directions; in group 2, dense connective tissues were observed with perpendicular, and therefore less mature, fibers ( Figs 5 and 6 ).
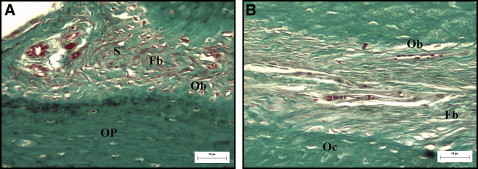
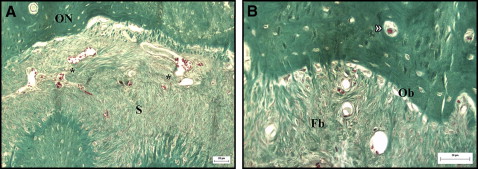
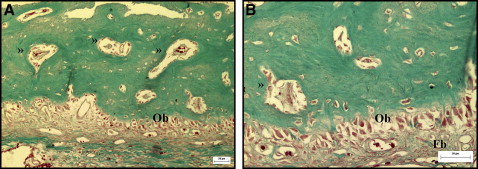
A large number of osteoblasts was observed along the edges of the suture in the newly formed bone tissue in all animals subjected to suture expansion; however, no significant difference was observed in the osteoblast counts between groups 1 and 2 ( Table II ). The medullary spaces in the newly formed bone were smaller and more regular in group 1. No pathologic changes were observed at the sites of the laser application in group 1.
| Experimental group | Mean number of osteoblasts | P |
|---|---|---|
| Group 1 (with laser) | 15.52 + 2.05 | 0.1447 |
| Group 2 (without laser) | 19.78 + 2.05 |
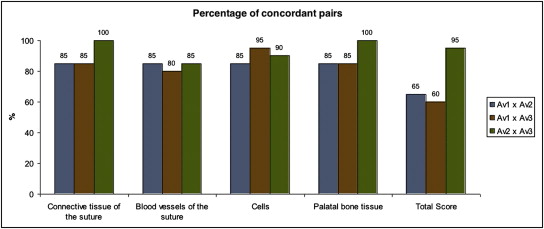
Tables III and IV show the agreement between the evaluators according to the scores for midpalatal suture maturation (the pairs were assessed by the Cohen kappa method). In Table III , a better agreement between evaluators 2 and 3 can be seen; they agreed substantially. The agreement between evaluators 1 and 3 was the lowest: the lowest percentage of agreeing pairs for the total score (60%) was between evaluators 1 and 3 ( Fig 7 ). Considering the 3 evaluators at once, Fleiss kappa coefficients of 0.536 for the total score and 0.798 for the bone tissue by the suture were found ( Table IV ).
| Variable | Evaluator 1 × evaluator 2 | Evaluator 1 × evaluator 3 | Evaluator 2 × evaluator 3 |
|---|---|---|---|
| Conjunctive tissue | 0.602 | 0.451 | 0.839 |
| Blood vessels | 0.681 | 0.583 | 0.681 |
| Cellularity | 0.625 | 0.875 | 0.733 |
| Bone tissue around suture | 0.706 | 0.706 | 1.000 |
| Total score | 0.434 | 0.381 | 0.808 |
| Variable | Kappa |
|---|---|
| Conjunctive tissue | 0.627 |
| Blood vessels | 0.647 |
| Cellularity | 0.744 |
| Bone tissue around suture | 0.798 |
| Total score | 0.536 |

Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


