Introduction
Our aim was to compare the dentoalveolar development in beagle dogs after palatal repair according to the Von Langenbeck technique with and without implantation of a dermal substitute.
Methods
Nineteen beagles (age, 12 weeks) were assigned to 2 experimental groups and an untreated control group. Palatal surgery was performed with the Von Langenbeck technique in the 2 experimental groups. The dermal substitute Integra (Plainsboro, NJ, USA) was implanted in 1 experimental group, and the other served as sham group. Dental casts were made before surgery and at several times in all groups to measure dentoalveolar development. Transversal distances, arch depth, tipping, and rotation were determined. Histologic evaluations were performed at 3, 7, and 15 weeks after surgery. The degrees of reepithelialization and tissue organization were evaluated microscopically.
Results
All wounds healed without complications. Scar tissue attached to the bone was found in both experimental groups. Deposition of bone in the Integra occurred after implantation, indicating its osteoconductivity. Transversal dentoalveolar development was similar in both experimental groups, but it was significantly less than in the control group.
Conclusions
Implantation of Integra after the Von Langenbeck procedure for palatal repair does not improve dentoalveolar development.
In cleft palate patients, maxillary growth and dentoalveolar development are often impaired after surgical closure of the palate. This is caused by 3 main factors: intrinsic developmental deficiencies, functional distortions, and the surgery. The last factor is considered the main one for midfacial hypoplasia in these patients. A strong indication for the involvement of surgery is the largely undisturbed growth in untreated cleft palate patients. Specific techniques, timing, sequence of surgical interventions, use of presurgical orthopedic appliances, and the surgeon’s skills might all influence the extent of growth impairment.
A widely accepted method for the primary closure of palatal clefts is palatoplasty with the Von Langenbeck technique or its modifications. Extensive research in animal models has been carried out to investigate the impairment of maxillary growth and dentoalveolar development after palatal surgery. These studies show that growth impairment is mainly due to manipulation of the mucoperiosteum. Both wound contraction and scar tissue formation in the lateral wound areas seem to be responsible for the iatrogenic effects. Wijdeveld et al used dogs in which a midpalatal soft-tissue cleft was closed by the Von Langenbeck technique. Their results showed similar growth disturbances as in the clinical situations. Furthermore, they found that the extent of the disturbances was related to age at the surgery. Especially when surgery was performed before or during the transition of the posterior teeth, the deviations in maxillary arch dimensions were most prominent. This was caused by decreased sutural growth and palatal tipping of the teeth in the lateral areas. Scar fibers were continuous with the cervical periodontal ligament, thus forming a mechanical connection between the teeth and the palatal bone. This presumably causes traction on the erupting permanent teeth.
Studies have been performed to prevent the attachment of scar tissue to the palatal bone. Surgical techniques that reduce the denudation of the palatal bone by means of local turnover flaps, buccal fat pad grafting, and free flaps showed some beneficial effects, but other attempts with low-level laser therapy, biodegradable poly-(L-lactic) acid membranes, or tissue expansion show no significant long-term improvements. In the partially split flap technique described by Leenstra et al, the palatal mucoperiosteum is split parallel to the bony surface. This technique causes less scar formation, and the growth pattern of the maxilla is more normal. However, theoretically, many of these techniques are hampered by the risk of avulsion of the major palatine artery, leading to necrosis of the palatal flaps. In spite of all efforts, most authors agree that scar-tissue formation after palatal repair is still a main cause of maxillary growth inhibition. Another approach is to supplement the tissue shortage after palatal repair with a dermal substitute.
Biodegradable collagen membranes, sometimes supplemented with other extracellular matrix components such as glycosaminoglycans, have been used for reconstruction of large burn wounds with promising results. Animal studies indicate that these materials might also be suitable for intraoral surgery. It has been reported that they reduce wound contraction and subsequent scarring. This study was designed to evaluate dentoalveolar development in beagle dogs after palatal repair according to the Von Langenbeck technique combined with implantation of the dermal substitute Integra (Plainsboro, NJ, USA). This study is one in a larger series that aims at the development of a substitute for oral mucosa that minimizes the iatrogenic effects of cleft palate closure.
Material and methods
After approval by the Board for Animal Experiments of the Radboud University Nijmegen Medical Centre in The Netherlands, the experiments were performed with 19 male beagles. The animals were kept under normal laboratory conditions and fed standard dog chow and water ad libitum. The dogs were divided randomly into 3 groups: an unoperated control group (group 1, n = 5) and 2 experimental groups (group 2, Von Langenbeck; and group 3, Integra; both, n = 7). Palatal surgery according to Von Langenbeck was performed in both experimental groups at 12 weeks of age. At the start of study, all dogs had complete deciduous dentitions.
Before surgery, the animals were injected intramuscularly with 0.5 mL Haloperidol (0.05 mg/mL fentanyl and 2.5 mg/mL droperidol; Janssen Pharmaceutica, Beerse, Belgium) and 0.5 mL atropine (0.5 mg/mL atropine sulphate). Subsequently, they were anesthetized with an intravenous injection of 30 mg per kilogram of Narcovet (60 mg/mL sodium pentobarbital; Apharmo, Arnhem, The Netherlands). After intubation, anesthesia was maintained with Ethrane (15 mg/mL enflurane; Abott, Amstelveen, The Netherlands). The palatal mucosa was cleaned with Betadine solution (povidoniodine; Dagra-Pharma, Diemen, The Netherlands). In addition, approximately 2.0 mL of Xylocaine (0.4 mg/mL lidocaine hydrochloride and 0.0125 mg/mL adrenaline; Astra Chemicals, Rijswijk, The Netherlands) was injected into the palatal mucosa to prevent excessive bleeding during surgery.
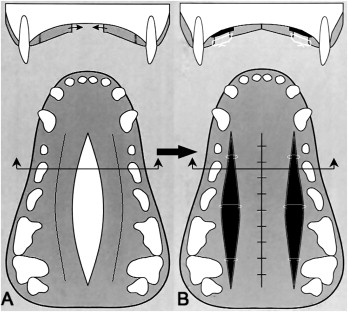
In both experimental groups, a standardized soft-tissue defect was created in the medial region of the palate by incising, elevating, and removing an elliptical mucoperiostal flap about 3.5 cm long. This flap extended distally from the canines to the region distal to the deciduous third molars. The maximum width of the flap was about 6 mm, which was about one-third of the transverse distance between the deciduous first molars. Full-thickness relaxation incisions were made in the mucoperiosteum on both sides of the palate close to the posterior teeth. The remaining palatal mucoperiosteum was elevated from the underlying bone with a small raspatory. Special attention was taken not to avulse the major palatine neurovascular bundle during this procedure. The midpalatal soft-tissue defect was closed and sutured (4-0 Vycril, Ethicon; Johnson & Johnson, Amersfoort, The Netherlands), leaving 2 areas of denuded bone adjacent to the dentition. In experimental group 2 (Von Langenbeck), the denuded bone areas were left untreated. The denuded bone areas in experimental group 3 were filled with Integra. Before use, the Integra was rinsed for 30 seconds in chlorhexidine digluconate 1% in water for disinfection, followed by 30 minutes in sterile physiologic saline solution. Then the top silicon cover layer of the Integra matrix was gently removed with tweezers. Subsequently, the Integra was placed directly on the denuded bone and placed under the wound margins for a few millimeters ( Fig 1 , B ). The matrices were carefully fixed in place with tensionless sutures crossing the defect (4-0 Vycril, Ethicon). Additional protection of the wounds was omitted, because this might have caused persistent inflammation and cytotoxic effects that could have delayed wound healing. The animals in group 1 served as unoperated controls. All experimental animals of groups 2 and 3 were medicated preoperatively with 1 mL of Albipen 15% (150 mg/mL ampicillin anhydrate; Mycofarm, de Bilt, The Netherlands). All animals received a normal diet after surgery.
Alginate impressions (CA 37; Cavex, Haarlem, The Netherlands) and standardized intraoral slides of the palate were taken in all animals at the start of the study (group 1) or before surgery (groups 2 and 3) at 12 weeks of age, and at 15, 17, 19, 22, 25, and 27 weeks of age. For this purpose, the animals were premedicated with 0.5 ml Haloperidol (0.05 mg/mL fentanyl and 2.5 mg/mL droperidol) and 0.5 mL atropine (0.5 mg/mL atropine sulplhate). Subsequently, they were anesthetized with an intravenous injection of 15 mg per kilogram of Nesdonal (50 mg/mL thiopental sodium). The alginate impressions were poured within 2 hours.
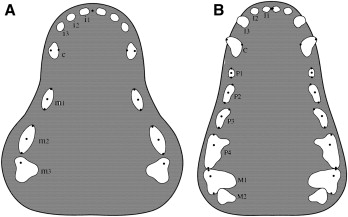
The following measuring points for the deciduous and permanent dentitions were defined on the dental casts ( Fig 2 , A and B ): (1) midpoint, the point between the central incisors; (2) tip points, the tips of the right and left canines, deciduous first, second, and third molars, permanent first, second, third, and fourth premolars, and permanent first molars; and (3) crest points, mesial and distal crests at the gingival margin of the right and left deciduous first and second molars, permanent first, second, third, and fourth premolars, and permanent first molars.
Standardized photographs of the dental casts were taken on the same day. A digital camera (D1x body, Nikon, Tokyo, Japan) and an AF Micro Nikkor fixed-focus lens (1:2.8, 105 mm) (Nikon) with an integrated Macro light-speed SB-295 ring flash (Nikon) were used. Measuring points were placed on the printed photographs with a pencil. The measurements were made only on fully emerged teeth. Therefore, it was inevitable that some data were missing at certain ages. A digital caliper (MarCal 16 EX, Carl Mahr Holding, Göttingen, Germany) and a protractor were used. Two independent observers measured all dental casts (R.O., L.V.). One observer made all measurements twice. The measuring points in the deciduous and permanent dentitions were used to calculate the following variables.
The transversal distances were the distances between the tips of the corresponding left and right teeth, if present, calculated for canines, deciduous first, second, and third molars, permanent first, second, third, and fourth premolars, and permanent first molars.
Arch depth was the distance between the midpoint and the midpoint of the line between the tips of the left and right deciduous third molars or, if available, between the left and right permanent first molars.
Tipping was calculated as the distance of the projection of the tip of a certain tooth to the line between the mesial and distal crests of the same tooth. It was also recorded whether the teeth were tipped palatally or buccally. Tipping was calculated for the deciduous first and second molars, permanent first, second, third, and fourth premolars, and permanent first molars.
Rotation was defined as the angle between the line used for the determination of arch depth and the line through the mesial and distal crests of individual teeth. Rotation was calculated for the same teeth as for tipping.
For the intraoral photos, an F body camera and a medical fixed-focus lens (1:5.6, f = 200 mm) with close-up lens (all, Nikon) and integrated ring flash and Kodak Ektachrome film (200 ASA; Eastman Kodak, Rochester, NY) were used. The reproduction ratio was 2:3. A plain intraoral mirror was placed behind the tuberosities of the maxilla at an angle of 45° to the palate. Optimal sharpness was obtained by carefully positioning the camera; 2 photos were taken at each occasion.
For the histologic evaluations, 2 dogs from each group were killed at 15 weeks of age (deciduous dentition), 19 weeks of age (after eruption of premolars), and 27 weeks of age. At those times, the animals were brought under general anesthesia by using 30 mg per kilogram of Narcovet (60 mg/mL sodium pentobarbital). After some minutes, a lethal dose of Narcovet was injected intravenously. Subsequently, the maxillae were dissected and immersed in 4% formaldehyde in 0.1 mol/L phosphate buffer at room temperature. After fixation, they were sawn into 5 smaller blocks. Two blocks contained the left and right second premolars and the adjacent lateral palatal tissue. Two other blocks contained the third premolars and the palatal tissue. The fifth block contained the midpalatal area between the fourth premolars and the first molar. The tissue blocks were decalcified in 20% formic acid and 5% sodium citrate. Decalcification was checked radiographically. Then the blocks were dehydrated through a graded series of ethanol and embedded in paraffin. Serial paraffin sections of 7 μm were made throughout the entire wound. Each tenth section was mounted onto a superfrost slide (Menzel-Gläser, Braunschweig, Germany) and stained with hematoxylin and eosin for general tissue survey. Polarized light microscopy was used to evaluate the collagen fiber orientation.
Statistical analysis
Before surgery, the anteroposterior distance and the 4 transversal distances in the deciduous dentition of all dogs were used for standardization to compensate for size and shape differences between individual dogs. The mean value of each distance of all dogs was calculated. Subsequently, a standardization factor was determined for each dog. This was calculated as the quotient of the mean and the initial values. This individual standardization factor was used during the entire experimental period for each distance. To assess the interobserver error of the method, all dental casts were measured by 2 independent observers (R.O., L.V.). Furthermore, to analyze intraobserver agreement, 1 observer (R.O.) made all measurements twice. Paired t tests were used to assess differences between the 2 observers for each variable. The reliability coefficients between the measurements of the 2 observers were calculated for each variable as Pearson correlation coefficients. For the assessment of intraobserver agreement, the duplicate measurement error was calculated by using Dahlberg’s formula.
Results
Wound closure was completed without complications after 2 weeks in both experimental groups. No wound showed signs of inflammation after 5 weeks. Palatal rugae had not regenerated in the wound area in either group at 15 weeks after surgery ( Fig 3 ).
The mean difference between the 2 observers for most variables was smaller than the duplicate measurement error. However, the duplicate measurement errors for arch depth in the deciduous dentition and rotation of the deciduous second molars on both sides were greater than the standard deviations of these variables. Statistically significant differences ( P <0.05) between the 2 observers were found for the following variables: arch depth in the deciduous dentition, rotation of the deciduous second molars on both sides, and rotation of the permanent fourth premolars on the left side. The overall differences for these variables between the observers were 0.41 mm, 1.32°, 1.12°, and 1.15°, respectively. The accuracy of the method was considered acceptable.
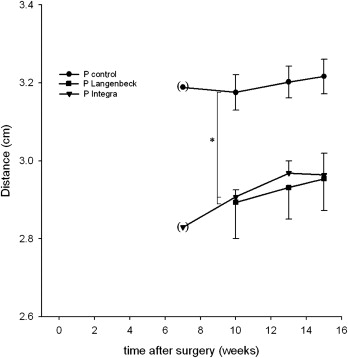
At the start of the study (12 weeks of age), there were no statistically significant differences between the groups. All transverse and anteroposterior distances increased in time with all groups. At 3, 5, 7, 10, and 13 weeks after the start, no statistically significant differences in these distances were found in the 3 groups, except for the transversal tip distances at the P1 (which has no predecessor), m1 (P2), and m2 (P3). All groups were comparable for the parameters of tipping and rotation.
The transversal distance P1 tip width of the 3 groups increased with time. The only significant differences between the groups was found for the transversal distance P1 tip at 10 weeks after surgery (control group >experimental groups 2 and 3). The transversal distance P1 tip widths were 3.18 ± 0.08 cm in the control group, 2.89 ± 0.16 cm in the Von Langenbeck group, and 2.91 ± 0.03 cm in the Integra group ( Fig 4 ).