Implant dentistry has become an excellent treatment modality since its inception in the modern era of dentistry. It not only allows a fairly conservative and esthetic alternative for treating partial edentulism but also provides a stable functional foundation for treating complete edentulism. Dental implants are a viable treatment option when there is sufficient quantity and quality of bone to achieve the desired functional and esthetic results.
The reduction in bone volume may have occurred as a result of periodontal disease before tooth loss, pneumatization of the maxillary sinus, long-term wearing of ill-fitting dentures, osteoporosis, and the physiologic bone remodeling that occurs after tooth extraction. In the past, the available bone was not modified and dictated the implant position, size, and number. Fewer and short implants were used in areas of deficient bone volume, thereby compromising the long-term treatment outcome. Today, the treatment plan first considers the final prosthesis options, followed by performing the necessary modifications to achieve the ideal environment needed for optimal implant placement.
Several surgical techniques can be used for correction of atrophied alveolar bone to establish the structural base of osseous tissue for supporting dental implants. Such techniques include guided bone regeneration, onlay bone grafting, interpositional bone grafting, distraction osteogenesis, ridge split, and sinus augmentation. The three-dimensional pattern of bone loss dictates selection of the optimum technique or techniques necessary to reestablish adequate bone volume and intermaxillary relationships to provide the most favorable biomechanical and esthetic conditions for implant placement.
Bone Graft Materials and Mechanism of Bone Regeneration
Different bone augmentation materials are used for alveolar reconstructive therapy, including autografts, allografts, alloplasts, and xenografts. Bone grafts of any type can regenerate bone through three possible mechanisms: osteogenesis , osteoinduction , and osteoconduction . Grafts may develop bone from one, two, or all three of these mechanisms to varying degrees.
Osteogenesis is new bone formation induced by osteoprogenitor cells that are present in the graft, survive the transplant, proliferate, and differentiate to osteoblasts; this process is called phase I osteogenesis . Autogenous bone is the only graft material with osteogenic properties.
Osteoinduction involves new bone formation by stimulation and recruitment of osteoprogenitor cells derived from undifferentiated mesenchymal stem cells at the graft site; this process is called phase II osteogenesis . Recruitment and differentiation are accomplished through a cascade of events triggered by graft-derived inducing factors called bone morphogenic proteins (BMPs), which are members of the transforming growth factor-β (TGF-β) superfamily. These BMPs are present in the matrix of the graft and are accessed after the mineral content of the bone graft has been removed by osteoclastic activity. It has been shown that osteoinductive materials can induce bone formation even in ectopic sites (subcutaneous tissue).
Osteoconduction is ingrowth of vascular tissue and mesenchymal stem cells into the scaffold structure presented by the graft material. Bone growth occurs by resorption or apposition from the existing surrounding bone in a process called creeping substitution , or phase III osteogenesis . Therefore, this process must occur in the presence of bone or undifferentiated mesenchymal cells. Osteoconductive materials do not grow bone when placed in soft tissue. Instead, the material remains relatively unchanged or is resorbed.
Types of Bone Grafts
An autograft , in which the graft is harvested from the same individual, is considered the gold standard of all bone-grafting materials because it is the only bone graft material that provides the three mechanisms of osteogenesis, osteoinduction, and osteoconduction required for bone regeneration. Autogenous grafts are nonimmunogenic, and their superiority comes from the transfer of osteocompetent cells to provide the osteogenic potential.
Autogenous bone can be harvested from intraoral sites such as the symphysis, maxillary tuberosity, ramus, coronoid process, and debris from osteotomies performed for insertion of the implant. The advantage of harvesting the graft intraorally is the ease of harvesting the bone, which may be present in the same surgical field and thus avoid exposure of another surgical site. Conversely, disadvantages include limitation of the amount of harvested bone. Extraoral bone graft harvesting is used to provide a greater volume of material for patients undergoing major augmentation procedures. Iliac crest, tibia, fibula, and cranial bone are common sites for graft harvesting.
An allograft is a graft that is taken from the same species as the host but is genetically dissimilar. The grafts are prepared as fresh, frozen, freeze-dried, mineralized, and demineralized ( Table 19-1 ). Numerous configurations of allograft bone are available, including powder, cortical chips, cancellous cubes, cortical struts, and others. Once the grafts are harvested, they are processed through different methods, including physical débridement, ultrasonic washing, treatment with ethylene oxide, antibiotic washing, gamma irradiation for spore elimination, and freeze-drying. The goal of these steps is to remove the antigenic component and reduce the host’s immune response while retaining the biologic characteristics of the graft. However, the mechanical properties of the graft are weakened.
| MATERIAL | COMMERCIAL SOURCE | COMPOSITION | BONE GROWTH METHOD | RESORPTION TIME |
|---|---|---|---|---|
| DFDB (demineralized) | Pacific Tissue Bank Grafton MTF DynaGraft |
Collagen + growth factors | Mainly osteoinduction, varies according to processing method | ±6 mo |
| FDB (mineralized) | MinerOss Puross |
Minerals + collagen | Mainly osteoconduction | 1 yr + |
Allogeneic bone is principally osteoconductive, although it may have some osteoinductive capability, depending on how it is processed. In 1965, Marshall Urist described the process of acid demineralization of bone before its implantation by using 0.5 to 0.6 mol/L hydrochloric acid. The organic bone matrix includes BMPs, which are factors responsible for the de novo bone formation. BMP is not acid-soluble, but the calcium and phosphate salts of hydroxyapatite (HA) are removed from the bone in the acid-reducing process, and as a result, demineralization of the freeze-dried bone (FDB) more readily exposes the BMPs with their osteopromotive effect. Therefore, FDB is primarily osteoconductive, whereas demineralized freeze-dried bone (DFDB) is believed to be osteoinductive.
The results of studies performed with DFDB are conflicting. Controversy still exists about the osteopromotive effects of DFDB. Some reports raise the question of variability in the concentration of BMPs in the commercially available grafts and conclude that the osteoinductive properties of DFDB are variable from one cadaver to another; in addition, the product fabrication process may have some effect on the osteoinductivity of the allograft if the demineralization process is very sensitive. It has been shown that the osteoinductive properties of the graft are eliminated if the calcium content is less than 2% by weight. Moreover, controversy exists about the use of ethylene oxide for sterilization of the graft because it may be destructive to BMPs.
Demineralized cortical bone was found to have higher concentration of BMPs than trabecular bone does. In addition, membranous cortical bone exhibits greater concentrations of BMPs than endochondral cortical bone does, so the skull represents a better source for inductive proteins than the rest of the skeleton.
Several studies have been conducted to evaluate the safety of allografts. According to the American Association of Tissue Banks, the probability of DFDB containing human immunodeficiency virus is 1 in 2.8 billion, as compared with a risk of 1 in 450,000 with blood transfusions. Rigorous background checks are performed on the donor and family before the donor is accepted into the program. Clinically, biopsy specimens of sites containing allograft from human patients sometimes show chronic inflammatory cells; however, the histologic appearance is nonspecific, and the inflammation cannot be attributed to an immune reaction with certainty.
Xenografts are derived from the inorganic portion of bone of a genetically different species than the host. One of the most popularly used xenografts is bovine bone. It is reported to be good bone bank material, provided that it is completely deproteinated by high temperature processing (1100° C) and all the residual organic material that might provoke an immune response is totally removed.
Concern has been raised over the risk for transmission of disease from cattle to humans through the bone graft material derived from the bovine bone used for dental implants. The recent incidents of bovine spongiform encephalopathy in humans have underscored this likelihood. Results from an analysis conducted by the German Federal Ministry of Health and the Pharmaceutical Research and Manufacturers Association of America showed that the risk for disease transmission was negligible and could be attributed to the stringent protocols followed in sourcing and processing the raw bovine bone used in the commercial products.
One of the best-known xenografts is Bio-Oss (Osteohealth, Shirley, NY) ( Table 19-2 ). It is processed by having all its organic material removed. This leaves a crystalline structure that practically matches human cancellous bone in structure. In 1992, Klinge and colleagues noted total resorption of Bio-Oss granules at 14 weeks after placement in rabbit skulls. However, Skoglund and co-authors reported that granules were present even after 44 months.
| MATERIAL | BRAND NAME | STRUCTURE |
|---|---|---|
| Deproteinized bovine bone mineral | Bio-Oss | Cancellous or cortical |
| Inorganic ovine HA + cell-binding peptide | PepGen P-15 | Peptide + microporous HA |
| Osteograft N | Microporous + macroporous | |
| Coral (Ca carbonate) | Biocoral Interpore 200 (Coralline) | Natural coral |
Another popular alternative xenograft is coralline hydroxyapatite , which is made from ocean coral. This material was created with the intention of producing a graft material with a more consistent pore size. Coral, which is composed mainly of calcium carbonate, is processed to remove most of the organic content. It is then subjected to high pressure and heat in the presence of an aqueous phosphate solution. When this process is completed, the calcium carbonate skeleton is totally replaced with a calcium phosphate skeleton (hydrothermal exchange). The material is concurrently sterilized in this process.
The generation of biomimetic microenvironments by using scaffolds containing cell recognition sequences in combination with bone cells offers tremendous potential for skeletal tissue regeneration. PepGen P-15 (DENTSPLY Friadent CeraMed, Lakewood, Colo.) is the first engineered collagen I binding domain for potential osteoblasts and is able to multiply the complete regeneration cascade. It is a combination bone replacement graft material composed of natural inorganic bovine-derived HA matrix coupled with a synthetic cell-binding peptide (P-15) ( Figs. 19-1 and 19-2 ).
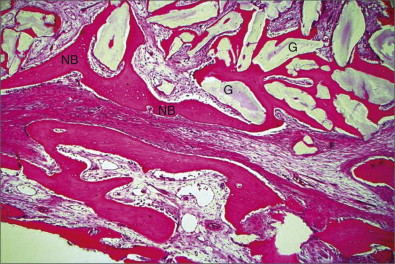
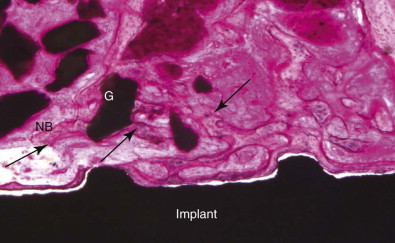
Alloplasts are synthetic bone substitutes that possess osteoconductive potential ( Box 19-1 ). The ideal synthetic graft material should be biocompatible and elicit minimal fibrotic changes. The graft should support new bone growth and undergo remodeling. Other features include similar toughness, modulus of elasticity, and compressive strength as the host’s cortical or cancellous bone. Many synthetic materials are available, including bioactive glass, glass ionomers, aluminum oxide, calcium sulfate, calcium phosphates as α- and β-tricalcium phosphate (TCP), synthetic HA, and synthetic absorbable polymers.
Among the main advantages of synthetic bone substitutes are unlimited supply and avoidance of a secondary surgical procedure for harvesting autogenous bone with its associated morbidity, such as pain, bleeding, paresthesia, infection, dysfunction, and other complications. However, the main disadvantage is lack of the osteoinductive power offered by autogenous grafts, which lead to direct osteogenesis from the implanted viable osteoblasts.
Clinicians may prefer performing grafting procedures with combination grafts . This will combine the osteogenic potential of autogenous bone and the unlimited supply offered by bone substitutes, which act as expanders or fillers . Combination grafts therefore decrease donor site morbidity caused by harvesting a large volume of autogenous bone for extensive reconstructive procedures.
Types of Bone Grafts
An autograft , in which the graft is harvested from the same individual, is considered the gold standard of all bone-grafting materials because it is the only bone graft material that provides the three mechanisms of osteogenesis, osteoinduction, and osteoconduction required for bone regeneration. Autogenous grafts are nonimmunogenic, and their superiority comes from the transfer of osteocompetent cells to provide the osteogenic potential.
Autogenous bone can be harvested from intraoral sites such as the symphysis, maxillary tuberosity, ramus, coronoid process, and debris from osteotomies performed for insertion of the implant. The advantage of harvesting the graft intraorally is the ease of harvesting the bone, which may be present in the same surgical field and thus avoid exposure of another surgical site. Conversely, disadvantages include limitation of the amount of harvested bone. Extraoral bone graft harvesting is used to provide a greater volume of material for patients undergoing major augmentation procedures. Iliac crest, tibia, fibula, and cranial bone are common sites for graft harvesting.
An allograft is a graft that is taken from the same species as the host but is genetically dissimilar. The grafts are prepared as fresh, frozen, freeze-dried, mineralized, and demineralized ( Table 19-1 ). Numerous configurations of allograft bone are available, including powder, cortical chips, cancellous cubes, cortical struts, and others. Once the grafts are harvested, they are processed through different methods, including physical débridement, ultrasonic washing, treatment with ethylene oxide, antibiotic washing, gamma irradiation for spore elimination, and freeze-drying. The goal of these steps is to remove the antigenic component and reduce the host’s immune response while retaining the biologic characteristics of the graft. However, the mechanical properties of the graft are weakened.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


