SEM
Solution
Presence of bacteria
Biofilm status
Culture growth (%)
6 % NaOCl
−
Absent
0
3 % NaOCl
−
Absent
20
1 % NaOCl
+
Disrupted
90
1 % NaOCl/MTAD
+
Disrupted
0
2 % CHX
+
Intact
0
+ Control
+
Intact
100
− Control
−
Absent
0
. Although chlorhexidine effectively kills biofilm, it lacked the ability to hydrolyze it, thus failing to achieve one of the basic objectives of endodontic treatment – debridement. By default, 6 % NaOCl is the only known endodontic irrigant, to date, capable of addressing the problems associated with endodontic biofilm; therefore, this chapter will only address complications associated with using NaOCl during endodontic treatment.
NaOCl: Cytotoxicity
Unfortunately, the chemical characteristic responsible for complete hydrolysis of biofilm produces devastating effects on living tissue. In a classical 1985 study, Pashley et al. [9] investigated the effect on red blood cells (RBC) and found that 5.25 % NaOCl, when diluted with saline at a ratio of 1:1,000, produced 96.3 % hemolysis of an RBC sample. The study also included the intradermal injection effect using a rat model and 5.25 % NaOCl. The intradermal injections resulted in immediate hemorrhage within the entire area of solution contact, and the affected areas ulcerated after 24 h. Pashley et al. warned: “NaOCl, while a very effective proteolytic solvent, is extremely cytotoxic and should be used judiciously and with caution in endodontic treatment. Even the suggestion that NaOCl, at some dilution, will only affect necrotic tissue should be abandoned.” Pashley et al. further noted that one of the serious clinical consequences of using NaOCl is the passage of some of the solution through the foramina, which sometimes occurs when the needle is momentarily wedged tightly into the canal during irrigation. Twenty-eight years after this warning, Pashley coauthored another publication that identified a far simpler and more dangerous cause of the NaOCl extrusion incident – direct intravenous injection via intraosseous infusion [10].
NaOCl: Complications
Complications from NaOCl extrusion includes (1) maxillary sinus incidents [11], (2) severe pain [12], (3) cellulitis [13], (4) life-threatening events [14], (5) permanent facial disfigurement [15], (6) permanent nerve damage [16], (7) secondary infection [17], and (8) acute kidney injury [18]. At the root of the problem is a broad misunderstanding of the reasons NaOCl is extruded from the apical foramen. It is generally believed that apical extrusion of NaOCl happens, as Pashley described, when an irrigation needle is wedged into a canal during irrigation; however, two studies disagree with this belief. First, in a survey of the diplomats of the American Board of Endodontics, only 20 % of the responding diplomats reported they felt the needle was wedged in the canal [19]. Second, in a one-of-a-kind clinical study, Hypaque (a radiopaque dye) was used as an irrigating solution [20]. It is important to note that the Hypaque investigators were aware of the possibility of forceful apical extrusion and reported that care was taken to insure that no irrigation needle was wedged into the walls, yet in both vital and nonvital teeth, apical extrusion of Hypaque was noted (Fig. 6.1). Considering the tissue toxicity of even the smallest amount of NaOCl, it seems reasonable that many patients would feel some degree of postoperative pain or discomfort following traditional endodontic irrigation. Gondim et al. proved this to be a statistically significant fact [21].
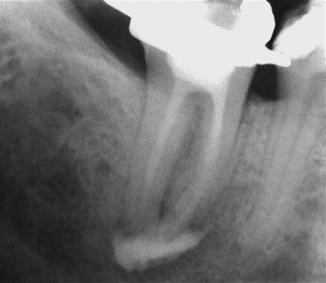

Fig. 6.1
In a unique clinical study, Salzgeber used a radiopaque dye (Hypaque) as an endodontic irrigant delivered judicially and cautiously via a non-binding needle during canal preparation and final irrigation. Despite careful delivery, sometimes regardless of the pulp’s vitality, the dye extended into the apical tissues. Regarding nonvital teeth: “When the Hypaque did extend into the periapical tissues in teeth with necrotic pulps, it seemed to respect no boundaries and occupied a random portion of the rarefied area.” In a situation as shown here, due to tissue reaction with NaOCl, post-op pain is likely a consequence, a finding supported by Gondim et al. [21] (From Salzgeber and Brilliant [20])
NaOCl: Reviewing the Extrusion Incident
A typical NaOCl extrusion is characterized in Fig. 6.2, but if an irrigant can escape the apical foramen as easily as demonstrated in Fig. 6.1, then why are these characteristic signs and symptoms of the NaOCl incident so rare? Furthermore, why isn’t the facial area directly superficial to the involved root apex virtually ever affected; while very specific other parts of the face in Fig. 6.2 are usually always affected, and other areas like the cheek are never affected. This is apparent again with the patient shown in Fig. 6.3a. Curiously, almost no ecchymosis is apparent at or near the right alar lobule, and this is the area directly superficial to the apex of the treated tooth (upper right lateral incisor), yet ecchymosis is apparent up to the super palpebral vein (arrow in “A”) and down to the angle of the mouth [22]. In July 2013, Boutsioukis et al. [23] published an extensive 16-page review paper that included 105 references and examined 40 case histories and stated: “There is a lack of clinical studies focusing on irrigant extrusion during root canal irrigation. Currently available case reports provide limited data on the possible factors that may influence irrigant extrusion.” It is important to note that the Boutsioukis’ et al. review was published in July 2013, four months before Pashley et al. described their novel theory of direct intravenous injection via intraosseous infusion.
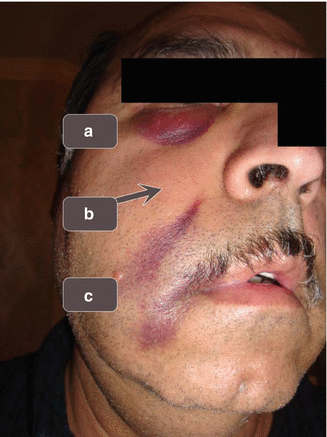
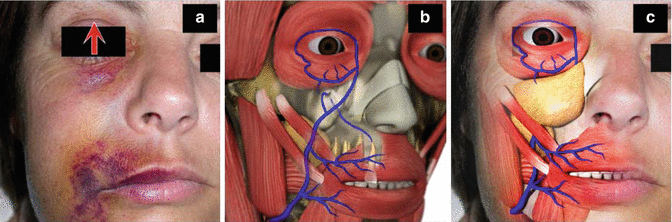

Fig. 6.2
The pathognomonic appearance of a NaOCl extrusion incident typically includes hemifacial edema and ecchymosis involving (A) one or both eyelids and (B) upper and lower lips beginning at the angle of the mouth but (C) never includes the cheek area [18] (With permission from Saudi Journal of Kidney Diseases and Transplantation)

Fig. 6.3
(a) The classical pathognomonic facial appearance of NaOCl infusion resulting from the treatment of the maxillary right lateral incisor. Interestingly, although the right superior palpebral vein (red arrow) shows the hemorrhagic effect of NaOCl infusion, the midface area just below the eyelids and upper lip is virtually unaffected (From Witton and Brennan [22]). (b) The course of the anterior facial vein and its tributaries including the palpebral veins of the eyelids, the superior and inferior labia veins, and, most importantly – an uncommon connection [40] – with the superior alveolar vein(s) that normally drains blood from the teeth to the pterygoid plexus of the veins in the infratemporal fossa. (c) The area between the eyelids and the angle of the mouth is unaffected because the malar fat pad and the zygomatic muscles cover the anterior facial vein, thus hiding any hemorrhagic effect (Figs. b & c with Permission from SybronEndo)
The article immediately following Boutsioukis’ July 2013 review was one of the three ex vivo studies published between April 2013 and May 2014 [24–26]. These ex vivo studies contained a method flaw obviously due to the investigator’s lack of knowledge regarding the more recent findings of Pashley et al. The principle investigator with Pashley was Zhu [10], and their work was not referenced in any of Boutsioukis’ ex vivo studies [24–26]. Additionally, two extremely important case histories [27, 28] were not included in the Boutsioukis’ review; therefore, the method flaw and the case histories will be examined in detail. Finally, the review criticizes the ex vivo study by Desai and Himel [29] as not specifying a research hypothesis or aiming regarding “irrigan[t] extrusion,” while in fact Desai stated: “The specific aim of this in vitro study was to compare the relative safety of various intracanal irrigation systems.” Furthermore, in Desai’s discussion, he stated the following: “The protocol for this study was designed to maximize the possibility of irrigant extrusion through an unrestricted, yet normal apex. It is understood that in clinical situations several factors might decrease the extent to which these systems extrude solutions. Periapical tissues and bone provide resistance to apical extrusion as well as non-patent canals. If quantities of periapical extrusion occurred clinically such as reported in this article, greater adverse treatment reactions associated with full-strength sodium hypochlorite would most likely occur. The model used most likely correlates, by design, to a canal that is open to atmospheric pressure, such as occurs when the apex of a tooth is extruding into the maxillary sinus with no apical covering or restriction.”
Maxillary Sinus Considerations
The maxillary sinus is uniquely located in the immediate vicinity to the apices of maxillary teeth. With age, the alveolar bone surrounding these apices becomes thinner to the point where the root tips may project into the maxillary sinus and may not be covered with bony lamina dura or even the schneiderian membrane [30]. Furthermore, the ostium maxillae communicate directly with the nasal cavity and consequently normal atmospheric pressure. Provided the root canal is fully patent during treatment, this unique root canal system and maxillary sinus anatomical relationship offers no resistance to fluid extrusion during endodontic irrigation. Two previously cited studies [25, 29] used similar methods and materials. Each experiment used single straight-rooted teeth with open apical foramen exposed to normal atmospheric pressure. In the Boutsioukis’ experiment, the canals were shaped to a #35/.06 and irrigated with open-ended (NaviTip) needles placed at WL – 1 mm with a delivery rate of 15.6 mL/min. Desai’s canals were shaped larger to a #50/.04 and also irrigated with an open-ended (NaviTip) needles placed at WL – 1 mm but at a slower delivery rate of 7 mL/min. The percent extrusion was very similar: Boutsioukis ≈60 % and Desai (larger apical size) recorded ≈70 %. In summary, both studies found that an unrestricted apical foramen permits a very high irrigant extrusion escape from the root canal system as in the case of the maxillary sinus situation described earlier [30].
Maxillary Sinus: NaOCl Incident – Case Reports
One of the earliest case histories of NaOCl extruded into the maxillary sinus reported a relatively benign reaction; the authors stated: “The expected deleterious sequelae were not seen” [31]. The authors described a routine endodontic treatment that resulted in the extrusion event and did not report any needle binding nor any dramatic physiological response, just that the patient indicated the taste of NaOCl in his throat during treatment. Treating the extrusion event consisted of flushing sterile water through the palatal canal of the maxillary first molar and out the maxillary sinus via the ostium. Amoxicillin, a decongestant, and Motrin were prescribed for seven days. Except for a mild soreness associated with the tooth and congestion of the associated maxillary sinus and a brownish material expressed when blowing his nose, the patient made a full recovery. Other case reports were not so favorable; Kavanagh and Taylor [32] reported a similar case with a different outcome. During routine treatment of an upper right second bicuspid, NaOCl was inadvertently injected into the maxillary sinus resulting in acute severe facial pain and swelling. A futile attempt was made to aspirate the extruded NaOCl via the endodontic access opening, resulting in the need to admit the patient for a Caldwell-Luc procedure under general anesthesia. The tooth was eventually extracted three months after the hospital procedure. Recently, a never before described sequelae resulting from the extrusion of NaOCl into the maxillary sinus has been reported [33]. Sleiman, who maintains a practice limited to endodontics, was referred to a patient with a chief complaint concerning an uncomfortable feeling relative to her right maxillary molar region where she had received endodontic treatment several months earlier. The clinical examination was normal, and while the radiographic appearance of the molar region revealed that the maxillary first molar had been treated endodontically, the treatment appeared unremarkable having been properly prepared and obturated; the only exception noted radiographically was a vague appearance of something unusual within the maxillary sinus. This vague appearance resulted in a CBCT scan. The panoramic view (Fig. 6.4a) revealed that tissue filled half the volume of the affected maxillary sinus. A close examination of the posterior maxillary sinus wall (Fig. 6.4b) revealed areas of bone loss. Referring to Fig. 6.5 (red arrow), it must be noted that the posterior wall of the maxillary sinus forms part of the anterior boarder of the infratemporal fossa, an area rich with several complex nerves leaving the cranium, and that exposure to NaOCl has been reported to cause permanent nerve damage [16]. Sleiman postulated that “Potentially, it could be the position of the patient during the root-canal procedure that made NaOCl stagnate on the posterior wall and aggravate[d] the damage.” When the patient was questioned about the procedure, she reported that during the treatment, she “had a chlorine taste in her throat arising from her nose as a liquid was dripping internally,” and on her way home from the endodontic treatment, the strange chlorine type liquid began to drip from her nose. Nothing of further consequence was reported. After viewing the CBCT results, the patient was referred to an otorhinolaryngologist for additional examination and treatment.
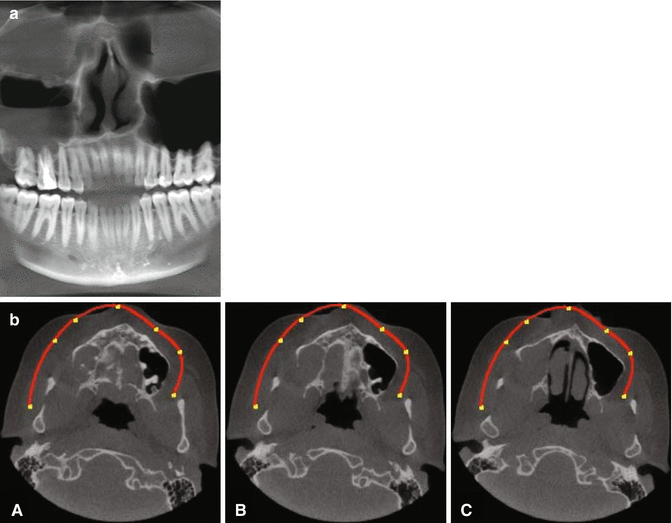
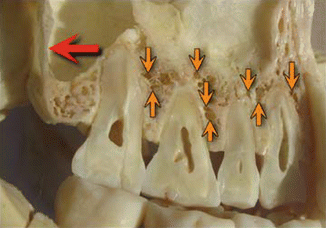

Fig. 6.4
(a) Panoramic CBCT scan demonstrates half of the maxillary sinus associated with endodontically treated tooth which is filled with inflammatory tissue (Courtesy of Dr. Philippe Sleiman, Beirut, Lebanon). (b) Sectional CBCT scan of same maxillary sinus demonstrates areas of the posterior wall that are nonexistent (Courtesy of Dr. Philippe Sleiman, Beirut, Lebanon)

Fig. 6.5
(Red arrow) The posterior maxillary sinus wall is a critical anatomical feature because it forms a significant portion of the anterior boarder protecting the infratemporal fossa. In addition to the pterygoid venous plexus, the infratemporal fossa contains the following nerves: mandibular (inferior alveolar, lingual, buccal), plus the otic ganglion and chorda tympani (Yellow Arrows). A few of the many abundant sinusoid spaces are identified throughout the maxilla
Maxillary Sinus: NaOCl Incidents – Treatment and Prevention
These three cited cases represent the spectrum of morbidity associated with extruding NaOCl into the maxillary sinus. Today’s imaging technology can, and has revealed heretofore, unknown consequences of extruding NaOCl into the maxillary sinus; therefore, even the most seemingly inconsequential incidents involving extrusion of NaOCl into the maxillary sinus must be approached with caution. The literature universally suggested antibiotic and anti-inflammatory therapeutic treatment in the case of most NaOCl incidents [34, 35]; however, from the CBCT images presented in Fig. 6.4b, it is apparent that in some cases, a consultation with an otorhinolaryngologist may be appropriate in cases involving the maxillary sinus. Regarding prevention, as previously mentioned, the study conducted by Desai was modeled to simulate a root without any resistance to apical extrusion, and balanced to atmospheric as may occur in the maxillary sinus. Desai concluded: “This study concluded that the EndoVac did not extrude irrigant after deep intracanal delivery and suctioning the irrigant from the chamber to full working length.” In concluding his case study, Sleiman opined: “One of the safest options that we currently have at our disposal is the EndoVac [Apical Negative Pressure] system, which is designed specifically to deliver fresh irrigant all along the root-canal system and, most importantly, to clean the last 3 mm of the root-canal system using the microcannula. It allows us to be certain that no chemicals can go beyond the limits of the root-canal space, nor cause any serious or even minor damage.”
Pathognomonic Appearance of NaOCl Extrusion: A Problem
The facial appearance resulting from injecting NaOCl beyond the apical termination of the root canal does not agree with the 1985 Pashley intradermal injection findings. Consider a hypothetical situation whereby excessive amounts of NaOCl exceed the Hypaque extrusion in Fig. 6.1 [20]. If that was the root cause of the NaOCl incident, then according to Pashley in 1985, all of the superficial tissue should be ecchymotic and eventually ulcerate. That doesn’t happen. Very specific parts of the face and neck are profoundly affected by ecchymosis: (1) the upper and lower eyelids on one (Fig. 6.6a) [27] or both sides of the face (Fig. 6.6b) [28], (2) the angle of the mouth only on the affected side (Fig. 6.6a, b), (3) sometimes the inferior boarder of the mandible only on the affected side (Figs. 6.6b and 6.7) [36], and (4) while other parts of the face, specifically the cheek, are never affected (Fig. 6.7).
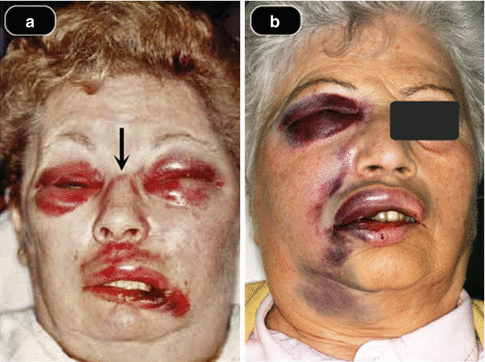
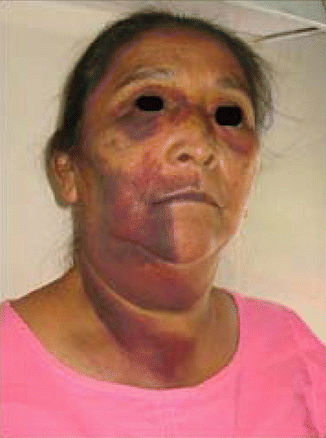

Fig. 6.6
(a) Upper left cuspid from Mehra et al. [27]. Most edematous and hemorrhagic effects of published NaOCl incidents are hemifacial. Although bilateral circumorbital ecchymosis is not uncommon, this case clearly demonstrates a vascular connection via superficial nasal veins (arrow) between both left and right circumorbital venous complexes suggesting that the NaOCl followed the venous connection across the bridge of the nose. (b) Upper right cuspid from Hülsmann [28]. There is no apparent venous connection between the orbits as shown in (a); thus, only the right side is affected. However, in this unique case, the anterior facial vein is positioned more toward the medial area of the face. Accordingly, since part of it is not hidden under the malar fat pad, the entire course of the anterior facial vein from the circumorbital veins to where it courses under the mandible joins the common facial vein which is apparent (Reproduced with permission of Elsevier)

Fig. 6.7
Upper right cuspid from de Sermeño et al. [36]. The ecchymotic pattern of this severe NaOCl incident is classically hemifacial, except where it crosses the bridge of the nose to include left circumorbital area as in Fig. 6.4b. Note the classical absence of ecchymosis in the area of the malar fat pad despite almost all of the middle, lower face and neck being affected
Pathognomonic Appearance of NaOCl Extrusion: A New Theory
In 2013 [10], a new theory hypothesized that the NaOCl extrusion incident is not the result of injecting excessive NaOCl into the periapical tissue alone, but rather its direct injection into the venous system, specifically (in most cases) the anterior facial vein and its associated complex of the veins (Fig. 6.3b). This theory evolved from the 1963 study by Rickles et al. [37] initiated by a fatal case history caused by air entering the circulatory system through the root canal space. Their study determined that when the air pressure inside the root canals was increased by using intracanal needles, air would enter the venous system, which would result in a fatal cardiac embolism. Twenty-seven years later, Davies and Campbell [38] reported three fatalities resulting from air entering the vascular system during implant surgery. They specifically stated that “For air embolism to occur there must be an open vessel, a gradient between extravascular and intravascular pressure, and a source of air. Bone tissue is very vascular….” A year prior to the Davies and Campbell paper, Manisali [39] reported an unusual case of canal overfilling (Fig. 6.8). In this unique case report, a radiopaque substance (iodoform paste) was injected into a lower second premolar and was forced out the apical foramen. At first it formed into a disorganized periapical mass similar to the appearance reported by Salzgber, but then within a few millimeters, it formed a second irregular mass that produced a well-defined wavy line extending distally. Manisali opined that the paste could have entered and coursed its way through a vein, not the inferior alveolar canal clearly shown positioned below the wavy line.
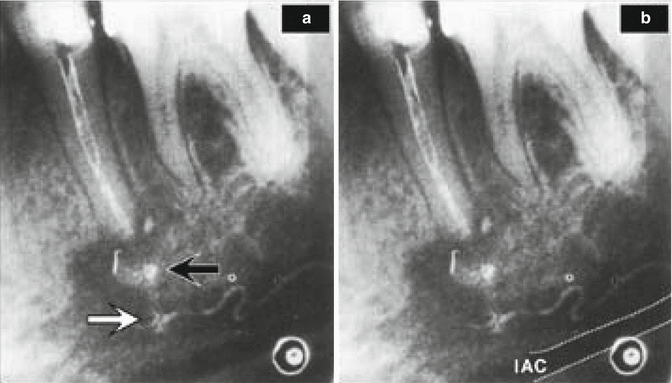

Fig. 6.8
From Manisali et al. [39], this figure shows overfill of iodoform paste which exhibits several unusual features. (a) The apical overfill of paste (black arrow) initially resembles the disorganized extrusion of Hypaque in Fig. 6.1, but a few millimeters below the initial overfill, a second mass (white arrow), appears again as another random mass then forms into a well-organized wavy line. (b) This line is not within the inferior alveolar canal (dotted line). Also note worthy is the faint radiopacity connecting the two masses. When viewed in its entirety, the paste overfill initially respects no boundaries upon leaving the apical foramen, then it becomes well organized as if running inside a blood vessel as it extends distally above the inferior alveolar canal
A careful examination of Fig. 6.3a shows an ecchymotic threadlike line extending across the upper eyelid. It is clearly the superior palpebral vein that is part of the anterior facial venous network (Fig. 6.3b). A direct vascular connection between the anterior facial vein and the maxillary teeth does, albeit rare, occur [40]. If NaOCl is extruded above a specific pressure gradient through patient’s maxillary right lateral incisor, it could enter a vein connected directly to the anterior facial vein and then spread through the venous complex affecting all areas extending from the upper eyelid to the angle of the mouth and beyond to the heart and the entire vascular system. The cheek would not be affected because the cheek fat pad (malar fat pad) and some of the zygomatic muscular fibers cover the anterior facial vein (Fig. 6.3c), therefore masking the hemorrhage. The case shown in Fig. 6.6b is very unique because it exhibits the full course of the anterior facial venous complex from the eyelids to the area where it courses under the mandible and joins the jugular vein; in this case, the anterior facial vein is positioned more laterally than usual and thus not hidden by the malar fat pad. Except for the inclusion of the other eye, Figs. 6.6a and 6.7 both share the pathognomonic characteristics of Fig. 6.3a including the absence of ecchymosis in the cheek area. Both circumorbital areas apparent in Figs. 6.6a and 6.7 are connected via a complex of superficial veins across the bridge of the nose (black arrow Fig. 6.6a). One apparent flaw in the theory is the fact that veins lack the elasticity of arteries and collapse easily, thus possibly nullifying the theory – but medullary sinusoids do not collapse, and they connect directly to veins. Schoeffel encountered another problem while investigating the uptake of ambient air by a healthy periodontal ligament [41]. He used the lower first premolar of young healthy dogs (Fig. 6.9a) and bonded a 21-gauge needle into a root canal space with an apical foramen prepared to 0.80 mm (Fig. 6.9b), and although the root canal space was pressurized to 175 mmHg above atmospheric pressure, no uptake was measured over 30 min.
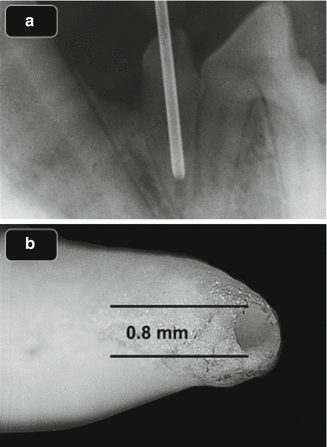

Fig. 6.9
(a) Radiograph of 21-gauge needle bonded into the root canal of live dog (From Schoeffel [41]). (b) Tooth after extraction demonstrating apical termination opened to 0.8 mm
Intraosseous Injection
In 1928, Drinker proposed that the intraosseous space be considered a non-collapsible vein [42]. Medullary bone contains thousands of small non-collapsible sinusoids that drain into larger veins [43–45]. The blood pressure in these spaces is approximately 30 mmHg, also known as the ¼ rule or 25 % of normal mammalian blood pressure [46, 47]. Since 1934, the interosseous (IO) space has been used to provide a reliable and safe method for allowing the introduction into systemic circulation [48–56]. Figure 6.10 shows a commercially available device used by the military and civilian medical personnel to establish rapid access to the venous system. Quoting from the FAST patent application 5,360,711: “It has long been recognized that access to the vascular system is available via bone marrow sinuses. See, e.g., Tocantins et al. [55], Turkel and Bethell, A New and Simple Instrument for Administration of Fluids Through Bone Marrow, War Medicine, pp. 222–25 (1944). Infusion of drugs or other fluids into the marrow (intraosseous infusion) results in rapid transmission of such fluids into the vascular system. This method of infusion can be quite important when the patient has very low blood pressure or collapsed veins.” Additionally, the IO route is used routinely in dentistry to effect profound anesthesia [57]. Accordingly, constructing an ex vivo model as either “open” or “closed” ignores the very well-established medullary bone space anatomy and physiology relative to the circulatory system. Under the correct conditions, intraosseous injection can occur when the pressure gradient exceeds approximately 30 mmHg [10]. Schoeffel’s observations in 1980 seem in conflict with Rickles findings, since he was using a model using a pressure gradient (170 mmHg); however, his observations were correct because the pressure approximated a healthy and intact periodontal ligament, not medullary sinusoids.


Fig. 6.10
FAST device for interosseous infusion
Pathognomonic Appearance of NaOCl Extrusion: A New Theory – Support
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


