Introduction
The purposes of this study were to detect, locate, and examine the changes in transverse nasal width, area, and volume from bone-borne, surgically assisted rapid maxillary expansion (SARME) with the Dresden distractor by using computer tomography (CT).
Methods
Sixteen patients (average age, 28.7 years) underwent axial CT scanning before and 6 months after SARME. They also underwent CT fusion on specific bony structures. The nasal bone width was examined in the coronal plane. The cross-sectional images of the nasal cavity were taken of the area surrounding the apertura piriformis, the choanae, and in between. We calculated cross-sectional areas and nasal volume according to these data.
Results
All but 2 patients had an increase in nasal volume of at least 5.1% (SD, 4.6%). The largest value of 35.3% (SD, 45.8%) was measured anteriorly on the nasal floor, decreasing cranially and posteriorly. This correlated with the V-shaped opening of the sutura palatina. There was no significant correlation between increase in nasal volume and transversal expansion.
Conclusions
Because most of the air we breathe passes over the lower nasal floor, SARME is likely to improve nasal breathing.
Rapid maxillary expansion (RME) is indicated in treating transverse maxillary deficiency orthopedically. RME, with a history of more than 145 years, was introduced by Angell. After initially falling into disrepute, it was reintroduced in the middle of the last century by Haas.
Its primary goal is to maximize orthopedic and to minimize orthodontic tooth movements. Tooth-borne expansion appliances were used initially; they were banded or bonded at the maxillary first premolars and molars.
RME exerts high forces that can easily split the midpalatal suture in young patients, forcing the maxillary halves apart. Separation becomes difficult after the midpalatal suture interlocks in late adolescence and even more difficult after fusion in adults because synchondrosis does not occur. But the greatest resistance associated with palatal expansion is because of the progressive ossification and thus increased rigidity of the entire viscerocranium. Consequently, in adults, preference is given to presurgical bilateral osteogenesis and fracture of the midpalatal suture. In these patients, the expansion procedure is based on distraction osteogenesis after surgical assistance.
There are reports of loss in vitality, extrusion, root resorption, buccal attachment loss, and serious tipping of the anchor teeth associated with tooth-borne RME. Presurgical osteotomy cannot completely eliminate these negative side effects. An alternative to the tooth-borne procedure is bone-borne fixation of the hyrax screw to the palatal bone with no interference of the teeth.
The bone-borne Dresden distractor (DD) has proved to be an effective device that prevents the negative side effects associated with tooth-borne RME.
Several experimental and clinical studies show no orthodontic advantages of RME, such as correction of dental crossbites only. RME also eliminates the effects of nasal obstruction on facial form, reduces the susceptibility to infections, and often leads to improved nasal breathing.
RME and surgically assisted RME (SARME) cause not only dentofacial but also craniofacial structural changes such as enlargement of the nasal cavity width and nasal volume.
The traditional explanation for the influence of RME and SARME on the nasal cavity is based on the separation of the nasal cavity’s lateral walls. The increase in the distance between the nasal cavity’s lateral walls enlarges the cross-sectional area and increases nasal volume, facilitating breathing. Transverse maxillary deficiency can reduce nasal dimensions and cause mouth breathing.
Documented evidence is still lacking of the effects of surgically assisted bone-borne dental arch expansion and associated transversal nasal airway dimensions on the number and location of the changes. Intercanine and intermolar width changes due to orthodontic treatment that would be relatively easy to measure cannot be extrapolated to changes in human nasal airway dimensions.
Evaluation of the nasal cavity became possible with lateral and posteroanterior cephalometric radiography. Although these methods were useful in determining obstructions in the nasal and pharyngeal areas, they are inadequate for measuring nasal resistance, airflow, and nasal area dimensions.
Rhinomanometry is another method to characterize nasal respiration objectively. From the applied data, we can derive an airway-resistance value from the airflow and the minimum cross-sectional area of the nasal airway—ie, the nasal valve.
Acoustic rhinometry (AR) was introduced by Hilberg in 1989 as a simple, painless, noninvasive, and reliable method for measuring the cross-sectional area and nasal cavity volume.
In dentistry, computed tomography (CT) has stood the tests for localizing displaced teeth, implant planning, difficult endodontic and complex surgical issues, and various applications in orthodontics. Timms et al first used CT to assess bone changes associated with RME.
The purposes of this study were to examine and detect the increases in transversal nasal width and the changes in nasal area and volume from bone-borne SARME with the DD by using CT.
Material and methods
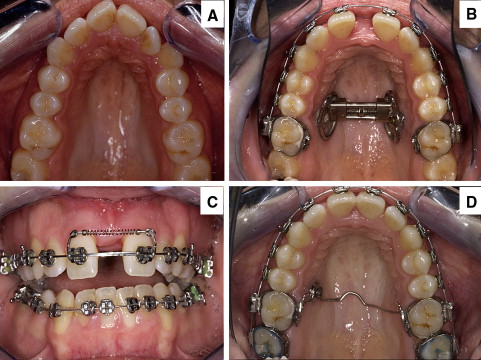
Sixteen patients (6 male, 10 female) from 17 to 36 years of age (mean, 28 years 8 months) underwent SARME with the DD. There were no dropouts or appliance failures. Initial diagnostic findings in all patients showed maxillary transverse constriction combined with Class II or Class III malocclusion or open bite, and no previous history of nasal disease ( Fig 1 , A ).
All patients underwent an axial spiral CT scan with the Somatom Sensation 16 (Siemens, Forchheim, Germany).
The scans were taken immediately before (T1) and an average of 6 months (SD, 2.5) after (T2) a bone-borne implant-supported RME device with the DD was placed.
The patients were positioned so that the occlusal plane was perpendicular to the horizontal plane. The area imaged was between margo infraorbitalis cranially and gnathion caudally. The image data were then converted into axial layers 0.5 to 0.8 mm thick. Three-dimensional (3D) reconstructions, image fusions, and measurements were done on workstations by using a software program (Syngo VX49B image fusion, Leonardo workstation VD10B, Siemens).
The CT scans were originally produced for the 3D evaluation of the DD’s effects on skeletal structures and teeth.
The surgical assistance according to Glassman et al and expansion-appliance placement were done during 1 operation with the patients under general anesthesia.
According to the method of Glassman et al, a bilateral osteotomy of the lateral walls of the maxillary sinus was performed 5 mm from above the apices of the apertura piriformis toward the pterygomaxillary fissure to break the resistance of the maxillary tuberosity and the contact between the maxilla and the zygomatic bones. To prevent irregular fractures of the alveolar ridge of the maxillary central incisors, we “preformed” the premaxilla above the central incisors with a chisel.
It was unnecessary to split the midpalatal suture surgically. The hyrax screw was activated intraoperatively to monitor the amount of surgical assistance required.
The expansion appliance consisted of a hyrax screw (Forestadent, Pforzheim, Germany) directly attached to the hard palate on 1 side with an implant (EO implant, Straumann, Freiburg, Germany; length, 4.0 mm; diameter, 3.5 mm; diameter of abutment, 5 mm) and with a self-drilling osteosynthesis miniscrew (Martin, Tuttlingen, Germany; length, 9-13 mm) on the other side. It was positioned between the roots of the second premolar and the first molar ( Fig 1 , B ).
The distraction device was attached only on 2 points, although a physical, parallel movement of the maxillary halves took place. Segmented archwires with a tension coil spring in the anterior part of the vestibule were used to open space in the incisor region with a 3-point support ( Fig 1 , C ). We thus incorporated the multi-bracket appliance for preparation, just before the SARME with the DD.
Three days after surgery, the patients were told to activate the screw 4 times a day (twice in the morning, twice in the evening) for 8 days (± 2 days) with 29 quarter rotations (24-36) of 0.25 mm each, for an average expansion of 7.25 mm.
The planned expansion was achieved with some overcompensation (0.5-1 mm) to neutralize the tooth-tipping effect and prevent relapse. The appliance was kept in place for 3 to 6 months to permit bone mineralization. After removing the expansion device, the implant was left in place and used for further retention and anchorage ( Fig 1 , D ).
The transverse dimension measurements and the CT-Osteo-3D-Fusions of the CT scans were taken by 1 person. The CT images and their evaluation were standardized.
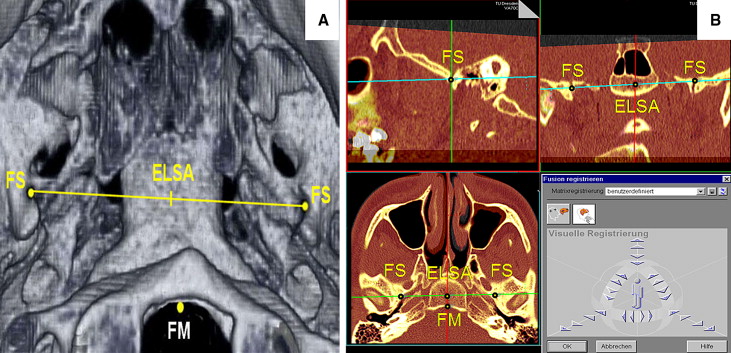
Each patient’s T1 and T2 CT scans were superimposed by using specific anatomic superimposition points: the foramen spinosum left and right (circumference), the anterior margin of the foramen magnum, and ELSA (the intersecting point of the line connecting the 2 foramina spinosa) ( Fig 2 , A ). We located and superimposed them using sagittal, coronal, and transversal reference levels ( Fig 2 , B ).
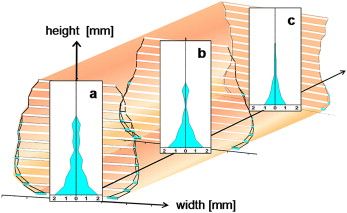
T1 and T2 bony nasal widths between the lateral walls of the nasal cavity were examined in the coronal plane starting from the nasal floor upward in 3-mm steps (W 1 … W k ). The height ranged from nasal floor (height 0) to the highest measurable nasal width.
The cross-sectional images of the nasal cavity were taken in the anterior vicinity of the apertura piriformis, behind the choanae region, and in between ( Fig 3 ).
To calculate nasal volume, we took transverse measurements as described above. Using this data, we calculated the 3 cross-sectional areas of the front (A ant ), middle (A mid ), and back (A post ) (A = 0.5 ∗ [W 1 … W k ] ∗ height).
The distance between these figures (X 1 , X 2 ) was used as a third dimension to determine the skeletal nasal volume (V): V = 0.5 ∗ ([A ant + A mid ] ∗ X 1 + [A mid + A post ] ∗ X 2 ). All cross-sectional areas and volumes of each patient were calculated before and after expansion with the DD (Excel, Microsoft, Redmond, Wash).
The data were analyzed with Excel. We measured each patient’s 3 cross-sectional areas and nasal volume and then calculated the mean values and standard deviations in our sample to identify differences in areas and volumes between T1 and T2.
Stochastic error was monitored, since 1 investigator (W.D.) repeated a patient’s CT-Osteo-3D-Fusion and retook the measurements of the T1 and T2 CT scans 15 times to determine the reproducibility of the linear nasal transversal measurements. The extension of the stochastic error of 1 linear measurement into the calculated volumes was considered to estimate their statistical significance.
The nonparametric paired Wilcoxon signed rank test was used to assess the statistical significance of width and cross-sectional areas before and after expansion. The level of significance was set at P <0.05.
The t test (α <0.05) was used to determine the correlation between transversal expansion of the DD and transversal dimension changes in the nose.
Results
The stochastic error of 1 transversal measurement was 0.06 cm ( P <0.05). All T1 vs T2 transverse dimensions and cross-sectional area measurements showed high significance ( P <0.05) according to the paired Wilcoxon signed rank test.
Because of the mathematical calculation of the cross-sectional areas and nasal volume of the transverse nasal cavity dimension, there was a direct correlation between them. Consequently, the change of cross-sectional areas and nasal volumes correlated directly or indirectly with changes of transverse cavity dimensions.
The transverse nasal cavity dimension changes and thus also the nasal volume enlargement decreased from the nasal floor in the cranial and dorsal directions ( Fig 3 ).
The largest expansion, 35.3% (SD, 45.8%), was observed on the anterior nasal floor ( Table I ). We observed relative and absolute differences in width, particularly in the transversal plane.
| Mean width (cm) | T2-T1 | ||||||
|---|---|---|---|---|---|---|---|
| Height (mm) | T1 | T2 | cm | SD (cm) | % | SD (%) | |
| Anterior | 39 | 1.53 ∗ | 1.53 | 0 | 0 | 0 | 0 |
| 36 | 1.78 ∗ | 1.78 ∗ | 0 | 0 | 0 | 0 | |
| 33 | 1.91 ∗ | 1.91 ∗ | 0 | 0 | 0 | 0 | |
| 30 | 1.92 ∗ | 1.92 ∗ | 0 | 0 | 0 | 0 | |
| 27 | 2.17 ∗ | 2.17 ∗ | 0 | 0 | 0 | 0 | |
| 24 | 2.18 ∗ | 2.26 ∗ | 0.08 ∗ | 0.20 ∗ | 2.99 ∗ | 7.92 ∗ | |
| 21 | 2.29 ∗ | 2.34 ∗ | 0.05 ∗ | 0.15 ∗ | 1.84 ∗ | 5.94 ∗ | |
| 18 | 2.26 ∗ | 2.32 ∗ | 0.07 ∗ | 0.14 ∗ | 2.70 ∗ | 5.50 ∗ | |
| 15 | 2.34 ∗ | 2.38 ∗ | 0.04 ∗ | 0.13 ∗ | 1.72 ∗ | 5.30 ∗ | |
| 12 | 2.34 ∗ | 2.46 ∗ | 0.12 ∗ | 0.35 ∗ | 9.46 ∗ | 30.14 ∗ | |
| 9 | 2.38 ∗ | 2.48 ∗ | 0.10 ∗ | 0.24 ∗ | 5.27 ∗ | 12.84 ∗ | |
| 6 | 2.25 ∗ | 2.44 ∗ | 0.19 ∗ | 0.22 ∗ | 8.29 ∗ | 9.04 ∗ | |
| 3 | 1.90 ∗ | 2.13 ∗ | 0.23 ∗ | 0.30 ∗ | 12.97 ∗ | 17.12 ∗ | |
| 0 | 1.23 ∗ | 1.64 ∗ | 0.41 ∗ | 0.47 ∗ | 35.29 ∗ | 45.79 ∗ | |
| Middle | 39 | 1.99 ∗ | 1.99 ∗ | 0 | 0 | 0 | 0 |
| 36 | 2.13 ∗ | 2.13 ∗ | 0 | 0 | 0 | 0 | |
| 33 | 2.35 ∗ | 2.35 ∗ | 0 | 0 | 0 | 0 | |
| 30 | 2.36 ∗ | 2.36 ∗ | 0 | 0 | 0 | 0 | |
| 27 | 2.39 ∗ | 2.39 ∗ | 0 | 0 | 0 | 0 | |
| 24 | 2.53 ∗ | 2.53 ∗ | 0 | 0 | 0 | 0 | |
| 21 | 2.72 ∗ | 2.79 ∗ | 0.07 ∗ | 0.09 ∗ | 2.46 ∗ | 2.90 ∗ | |
| 18 | 2.85 ∗ | 2.88 ∗ | 0.03 ∗ | 0.08 ∗ | 0.88 ∗ | 2.75 ∗ | |
| 15 | 3.04 ∗ | 3.04 ∗ | 0.01 ∗ | 0.02 ∗ | 0.30 ∗ | 0.81 ∗ | |
| 12 | 3.16 ∗ | 3.21 ∗ | 0.05 ∗ | 0.06 ∗ | 1.45 ∗ | 2.06 ∗ | |
| 9 | 3.17 ∗ | 3.24 ∗ | 0.07 ∗ | 0.10 ∗ | 2.13 ∗ | 2.93 ∗ | |
| 6 | 3.06 ∗ | 3.21 ∗ | 0.15 ∗ | 0.18 ∗ | 4.62 ∗ | 5.44 ∗ | |
| 3 | 2.80 ∗ | 3.01 ∗ | 0.20 ∗ | 0.31 ∗ | 6.79 ∗ | 11.21 ∗ | |
| 0 | 2.21 ∗ | 2.57 ∗ | 0.35 ∗ | 0.35 ∗ | 15.18 ∗ | 15.64 ∗ | |
| Posterior | 36 | 3.00 ∗ | 3.00 ∗ | 0 | 0 | 0 | 0 |
| 33 | 2.83 ∗ | 2.83 ∗ | 0 | 0 | 0 | 0 | |
| 30 | 2.84 ∗ | 2.84 ∗ | 0 | 0 | 0 | 0 | |
| 27 | 2.60 ∗ | 2.60 ∗ | 0 | 0 | 0 | 0 | |
| 24 | 2.57 ∗ | 2.58 ∗ | 0.01 ∗ | 0.02 ∗ | 0.42 ∗ | 1.02 ∗ | |
| 21 | 2.47 ∗ | 2.48 ∗ | 0.01 ∗ | 0.01 ∗ | 0.26 ∗ | 0.61 ∗ | |
| 18 | 2.49 ∗ | 2.51 ∗ | 0.02 ∗ | 0.08 ∗ | 0.49 ∗ | 2.53 ∗ | |
| 15 | 2.49 ∗ | 2.52 ∗ | 0.02 ∗ | 0.04 ∗ | 0.86 ∗ | 1.57 ∗ | |
| 12 | 2.59 ∗ | 2.61 ∗ | 0.02 ∗ | 0.06 ∗ | 0.78 ∗ | 2.41 ∗ | |
| 9 | 2.81 ∗ | 2.87 ∗ | 0.06 ∗ | 0.07 ∗ | 2.11 ∗ | 2.56 ∗ | |
| 6 | 2.98 ∗ | 3.06 ∗ | 0.08 ∗ | 0.07 ∗ | 2.72 ∗ | 2.33 ∗ | |
| 3 | 2.98 ∗ | 3.09 ∗ | 0.12 ∗ | 0.11 ∗ | 3.83 ∗ | 3.50 ∗ | |
| 0 | 2.61 ∗ | 2.82 ∗ | 0.21 ∗ | 0.23 ∗ | 8.04 ∗ | 9.41 ∗ | |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


