Introduction
A review of the literature showed that there are disturbances of muscle activity and tongue function in patients with anterior open bite (AOB). However, most studies have had white subjects, even though open bite is more prevalent in black populations. In this pilot study, we examined jaw muscle activity and tongue pressure in East African black subjects.
Methods
Ten subjects (4 boys, 6 girls; ages, 10.1-13.2 years) were recruited. A cephalometric comparison of subjects with and without AOB was performed. Surface electrodes were placed on the bilateral anterior temporalis, superficial masseter, superior orbicularis oris, and anterior digastric muscles. Electromyograms were recorded at rest, maximal voluntary clenching, chewing, and swallowing. Tongue pressure during rest and swallowing, and maximal bite force were also measured.
Results
There was no statistically significant difference between the 2 groups in cephalometric measures (except in amount of open bite and overbite) and maximum bite force. During chewing, both groups showed similar coordinated activity patterns in the muscles, but the AOB subjects tended to show more electromyographic activity in the muscles of the balancing side. During clenching, the AOB subjects exhibited lower electromyographic activity compared with the controls. AOB subjects also had greater anterior tongue pressure during swallowing. Data from tongue pressure at rest were inconclusive.
Conclusions
East African blacks appear to have neuromuscular and skeletal predispositions to AOB, but their vertical incisor relationships are variable. Factors in addition to those investigated might be involved.
The role of the orofacial musculature in determining facial morphology and tooth position is unclear. Although facial morphology and bite force might be considered by some orthodontists to be more closely related than by others, it is unknown whether facial morphology determines the strength of the masticatory muscles or whether a strong musculature influences the form of the face. Patients with anterior open bite (AOB) have less strength in jaw elevator muscles, weaker occlusal force, abnormal deglutitive tongue action, and imbalanced coordination of muscle activity. Negative correlations have been established between the maximal activity of jaw elevator muscles and the vertical dimensions of the facial skeleton. Subjects with AOB have both reduced occlusal stability and increased vertical dimension. Since patients with AOB also have both long faces and weak masticatory muscles, some suggest that AOB is the result of an imbalance in jaw muscle activity, abnormal tongue pressure, or weak maximal bite force.
To understand the influence of craniofacial muscles, surface electromyography (EMG) is routinely used to characterize muscle functions. Time-domain EMG analyses can be used to identify muscle activity and action potential of motor units, whereas frequency-domain EMG analyses can aid in identification of muscle fatigue. Therefore, EMG was used to evaluate the function of the jaw and lip muscles in this study.
In addition to muscle activity, some researchers have focused on the tongue as the primary factor in the etiology of AOB. Proffit and Proffit et al measured force levels of the tongue against the maxillary incisors and palate during rest and normal swallowing, and concluded that the resting position of the tongue was more significant than the swallowing position in determining dental arch form. However, other investigators have shown that functional tongue movements during deglutition are significantly correlated with certain features of maxillofacial morphology such as AOB. Therefore, there is evidence in the literature that both resting and functional positions of the tongue are correlated with AOB.
Although some studies have described the dentoalveolar and skeletal characteristics of AOB and discussed various etiologic factors, such as tongue morphology and function and muscle activity, they often did not include black subjects, or, if other races were included, the samples were compared with white norms. There are significant differences in incisor relationships between the various ethnic groups. Also, there is much variation among races in the prevalence of AOB. American blacks have a prevalence of AOB 4 to 8 times higher than do their white counterparts. East African blacks also have a disproportionately high prevalence of AOB. The prevalence of AOB is significantly higher (56%) in East African blacks (Somalia and Ethiopia, 9-12 years old) compared with white, Hispanic, and Asian ethnic groups in an ongoing clinical trial (17%, P <0.01, unpublished data). Also, orthodontists anecdotally report that these patients are highly prone to develop AOB during treatment. Therefore, investigating AOB in this ethnic group is a unique opportunity to study this infrequent malocclusion.
This pilot study assessed craniofacial morphology, jaw and lip muscle activities, maximal bite force, and tongue pressure in East African blacks with and without AOB. The hypothesis was that ethnic risk factors related to a higher prevalence and increased severity of AOB in East African black children cause altered orofacial muscle activity and tongue pressure during function.
Material and methods
Ten East African children were recruited for the study: 4 boys and 6 girls, ages 10.1 to 13.2 years. All are the first generation of immigrants from Somalia (7 subjects), Ethiopia (2), and Eritrea (1) living in the Seattle urban area. Two subjects had AOB (lack of incisal overlap), and 3 had an AOB tendency (lack of incisal contact with minor incisal overlap <1.0 mm); 5 had incisal contact with incisal overlap (>1.0 mm) and served as the control group (3 boys, 2 girls). Incisal contact was determined cephalometrically and verified with the subjects’ preorthodontic casts. Subjects with AOB and AOB tendency were considered the AOB group (2 boys, 3 girls). There were similar sex distributions and age ranges for both groups. The parents and children were fully informed of the purpose of the study and gave their consent. The Angle classification of the patients included 6 Class II, end-on, 3 Class I, and 1 transient Class III molar relationship because of retention of the bilateral maxillary deciduous second molars. The most prevalent reported habit was mouth breathing, and all subjects denied a history of thumb or finger sucking. They had received no previous orthodontic treatment. All procedures were approved by the Institutional Review Board of University of Washington Regional Medical Center/Children’s Hospital.
Since the subjects were about to start orthodontic treatment, lateral cephalometric radiographs were available. The cephalometric landmarks were identified by the primary investigator (F.Y.), and the accuracy of landmark identification was confirmed by a secondary investigator (V.S.). If consensus was not reached, a third investigator (Z.-J.L.) was used. The cephalograms were digitized and redigitized 4 weeks later to evaluate intraexaminer error. These interreader and intrareader reliabilities were tested by using paired t tests for values measured by the 2 investigators using Dahlberg’s equation <SPAN role=presentation tabIndex=0 id=MathJax-Element-1-Frame class=MathJax style="POSITION: relative" data-mathml='(√Σd2/2n)’>(√Σd2/2n)(√Σd2/2n)
( √ Σ d 2 / 2 n )
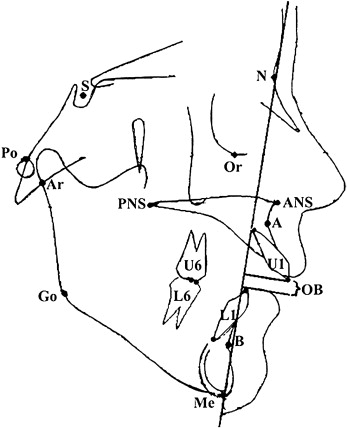
, respectively. No significant difference was identified from multiple t tests for each variable, and the errors were 0.7% to 6.0% for linear and 0.5% to 4.5% for angular measurements. AOB was measured as the distance between the incisal edge of the maxillary central incisor to the incisal edge of the mandibular central incisor measured parallel to the nasion-menton line. Landmarks used are illustrated in Figure 1 .
Maximal bite force was measured in the region of the right and left permanent first molars by using a portable occlusal force gauge (Model GM10, Nagano Keiki Co., Matusmoto, Japan). The device consists of a hydraulic pressure gauge that displays the resulting bite force (in kilograms) and a biting element (a disposable vinyl plastic tube). The subjects were positioned in the chair with the Frankfort horizontal plane parallel to the floor and were asked to execute maximal clenching on the device. The force was measured 1 side at a time and recorded 3 times in an interval of 3 minutes. The average of these values was used as the bite force at the location.
Eight pairs of miniature surface electrodes (1 cm sensor diameter, NeoTrace Kitty Cat Electrode, Kappa Medical, Prescott, Ariz) with an interelectrode distance of 5 to 10 mm were placed over the centers of the anterior temporalis (Tp), superficial masseter (Ma), superior orbicularis oris (Oo), and anterior digastric (Di) muscles bilaterally along the direction of the muscle fibers ascertained by palpation of the muscles. A ground electrode was secured on the forehead. The skin was cleaned with 99.5% alcohol swabs and a skin preparation gel (NuPrep, D. O. Weaver & Co, Aurora, Colo) to reduce skin impedance. An electrolyte gel (Beckman, Fullerton, Calif) was applied to the electrode to improve conduction. The EMG leads were connected to the amplifier probes (model P511, Grass Neurodata Systems, Warwick, RI). Each subject was seated on a chair with the head unsupported and oriented with the Frankfort horizontal plane parallel to the floor. The EMG activities for the following conditions were recorded at the sampling frequency at 4545.5 Hz: (1) at mandibular rest position for 30 seconds, (2) at maximal voluntary clenching (MVC) for 30 seconds, (3) during gum chewing, and (4) repeated water swallowing (20 mL, 3 times). The patients were instructed to practice these activities before the recording. For chewing, the recording was started after the gum became soft and could be shaped by the tongue.
After digital filtering (band pass of 60-1500 Hz), the following EMG parameters were analyzed.
- 1.
Integrated EMG activities (IEMG) at rest for 10 seconds.
- 2.
IEMG of all 4 muscle pairs and mean frequency (MF) of Ma and Tp pairs from the first and final 2.5-second period of the 15 seconds of MVC.
- 3.
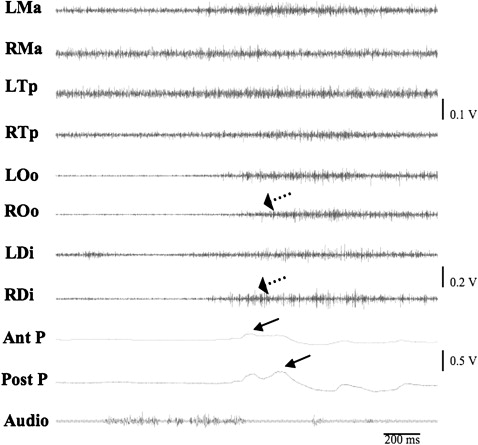
EMG timings and activities from 20 right-chewing cycles. Chewing side was identified by comparing bilateral Ma activity and choosing the one with higher amplitude ( Fig 2 ). The following EMG parameters were quantitatively assessed for each chewing cycle: onset of activity burst, burst duration measured from the onset to offset of activity burst, IEMG of activity burst, and chewing frequency measured from 1 onset to the next onset of the right Ma (ie, chewing cycle length) ( Fig 3 ). These parameter measurements were averaged for the 20 consecutive chewing cycles.

Fig 2 Raw EMG chewing recording. LMa, left masseter; RMa, right masseter; LTp, left temporalis; RTp, right temporalis; LOo, left orbicularis oris; ROo, right orbicularis oris; LDi, left digastric; RDi, right digastric. R and L at the top indicate chewing side.
Fig 3 Close-up view of chewing EMG recording. Solid arrows represent the activity burst, and dotted arrows indicate the onset of muscle activation. Two dashed vertical lines (from the onset of 1 burst of RMa to the next) define 1 chewing cycle length (100%). See Figure 2 for other abbreviations. - 4.
IEMG of each muscle during water swallowing.
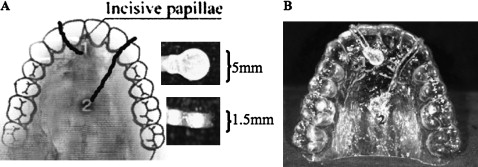
Tongue pressure on the hard palate of the maxilla was measured during rest and water swallowing with 2 titanium-bodied pressure transducers. Company specifications give a frequency response of 2.6 kHz, an operational pressure range of 13.3 to 153.3 kPa, and a capacity up to 400 kPa (Konigsberg Instruments, Pasadena, Calif). To install these transducers, an Essix-type appliance (1 mm thick) covering the anterior two thirds of the hard palate area was made for each subject. The 2 transducers were embedded in the appliance along the midline, one at the level of incisive papilla and the other at the level of the mesial surface of the first molars ( Fig 4 ). The cables of the transducers were led out of the front of the mouth and connected with the direct current amplifiers. The patients wore the appliance for a few minutes before the measurements to acclimatize themselves to it. The pressure recordings were taken simultaneously with EMG examination during rest position and 3 trials of water swallowing (20 mL) ( Fig 5 ).
Statistical analysis
Paired t tests were used to assess any side difference (right vs left) in maximal bite force and IEMG or MF during rest, clenching, and swallowing. Independent t tests were used to compare the means of maximal bite force, and cephalometric and EMG data between the AOB and control subjects. The results from nonpaired t tests were confirmed with a nonparametric test (permutation test). Since this was a pilot study with a limited sample size, no power estimate was performed, and probability levels of P <0.1 were considered statistically significant.
Results
Mean values for the cephalometric measurements were similar for both groups, with large variations in each group. The only statistically significant difference between the 2 groups was the amount of open bite ( P = 0.07, Table I ).



