Introduction
Primary failure of eruption (PFE) is characterized by nonsyndromic eruption failure of permanent teeth in the absence of mechanical obstruction. Recent studies support that this dental phenotype is inherited and that mutations in PTH1R genes explain several familial cases of PFE. The objective of our study was to investigate how genetic analysis can be used with clinical diagnostic information for improved orthodontic management of PFE.
Methods
We evaluated a family (n = 12) that segregated an autosomal dominant form of PFE with 5 affected and 7 unaffected persons. Nine available family members (5 male, 4 female) were enrolled and subsequently characterized clinically and genetically.
Results
In this family, PFE segregated with a novel mutation in the PTH1R gene. A heterozygous c.1353-1 G>A sequence alteration caused a putative splice-site mutation and skipping of exon 15 that segregated with the PFE phenotype in all affected family members.
Conclusions
A PTH1R mutation is strongly associated with failure of orthodontically assisted eruption or tooth movement and should therefore alert clinicians to treat PFE and ankylosed teeth with similar caution—ie, avoid orthodontic treatment with a continuous archwire.
Primary failure of eruption (PFE), originally described by Proffit and Vig, is characterized by nonsyndromic eruption failure of permanent teeth in the absence of mechanical obstruction. The hallmark features of this condition are (1) infraocclusion of affected teeth, (2) increasing significant posterior open-bite malocclusion accompanying normal vertical facial growth, and (3) inability to move affected teeth orthodontically.
Many studies have noted the heritable basis of this dental phenotype, and recently mutations in parathyroid hormone receptor 1 ( PTH1R ) have been identified in several familial cases of PFE. A detailed clinical analysis showed several types of nonsyndromic PFE, including type I and type II, with both types primarily affecting the posterior segments, either unilaterally or bilaterally. Type II is further characterized by greater eruption potential of the most distal tooth affected with PFE. It is not clear whether specific phenotypes (clinical variations described as type I vs type II) are associated with distinct genetic mutations or whether the clinical variation represents the broad spectrum associated with, for instance, PTH1R . It is known that, despite clinical severity or type, PFE does not respond to orthodontic force. Previous studies in our laboratory ruled out AMELX , RUNX2 , POSTN , and AMBN as candidate genes for PFE in families and individuals. However, recent advances in gene discovery, particularly with PFE, will most likely provide a novel diagnostic tool to aid in the clinical management of orthodontic patients when failure of eruption is suspected.
Eruption failure can be a part of a syndrome or occur as an isolated condition, such as PFE. In either case, it is critically important to distinguish between eruption failure due to local or mechanical causes (cysts, interference of adjacent tooth, lateral pressure from the tongue, or secondary to a syndrome such as cleidocranial dysplasia) and failure of the eruption mechanism completely. The orthodontic management of mechanical failure of eruption (MFE) is quite different from that of PFE. In the case of syndrome involvement, the orthodontic diagnosis triage should include whether the syndrome results in MFE, including but not limited to osteopetrotic bone, fibrous gingival tissue, or impacted teeth, or whether it is due to delayed eruption. The genetic alteration of at least 1 gene, PTH1R , is evidence that there is also a biologic distinction between PFE and other eruption disorders such as MFE. Hence, once MFE is ruled out, genetic analysis of PTH1R should be a critical part of the orthodontic diagnostic regimen.
PTH1R resides on the small arm of chromosome 3 (3p22-21.1) and encodes a G-coupled protein receptor for both parathyroid hormone (PTH) and parathyroid hormone-related protein (PTHrP). A key function of PTH is regulation of calcium metabolism, whereas PTHrP plays a major role in skeletal development during early bone growth by regulating chondrocyte proliferation and differentiation. Recessive mutations in PTH1R have been identified in people with Blomstrand chondrodysplasia (BOCD; OMIM: 215045), a genetic disorder characterized by advanced endochondral bone maturation, increased bone density, and short stature. Those affected with BOCD also had abnormalities in breast development and several impacted teeth. In BOCD, autosomal recessive mutations have led to mutational inactivation of PTH receptors. Other conditions associated with mutations in PTH1R include Jansen metaphyseal chondrodysplasia (OMIM: 156400), enchondromatosis, and Eiken syndrome (OMIM:600002), all of which include cartilage or skeletal dysplasia. Despite the many disorders associated with mutations in PTH1R , the recent report of PTH1R mutations associated with PFE makes this a high-priority candidate gene for confirming diagnosis of a nonsyndromic PFE phenotype. The objective of our study was therefore to investigate how genetic analysis can be used with clinical diagnostic information for improved orthodontic management of PFE.
Material and methods
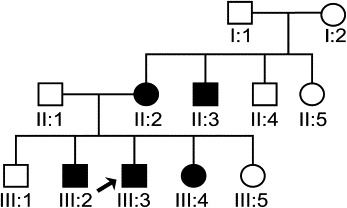
Enrollment for this study was based on PFE in at least 2 family members. All subjects (adults and minors) consented to participate (including a release for dental records). Approval for this study was granted by the Biomedical Institutional Review Board at the University of North Carolina at Chapel Hill. Through detailed interviews, a pedigree ( Fig 1 ) was extended in 1 family. This family consisted of 12 people from 11 to 72 years of age. Five male and 4 female members were available for clinical and molecular testing. A comprehensive clinical evaluation was performed to determine (1) a positive diagnosis of PFE with no mechanical or secondary barrier and also based on at least 1 unerupted tooth after unsuccessful orthodontic treatment for the proband; (2) the skeletal relationship including vertical (overbite or open bite) and sagittal (Angle Class I, Class II, or Class III molar relationship) based on clinical examinations and cephalometric radiographs; and (3) anomalies in growth or stature either absent or present. The clinical evaluation included clinical photographs; panoramic and cephalometric radiographs were obtained for participating family members.
Mutational analysis was performed after extraction and purification of DNA from buccal cells (PureGene kit, Gentra Systems, Minneapolis, Minn) or saliva (Oragene, DNA Genotek, Toronto, Ontario, Canada). We amplified and sequenced all coding exons of PTH1R (exons 3-16) for the 9 subjects using primer sets as described previously. To include splice junctions in our analysis, primer sets were designed to delineate regions that included a minimum of 25 bases of intron sequence in addition to the exon sequence. Amplification was performed by using Accuprime polymerase chain reaction buffer and enzyme mix (Life Technologies/Invitrogen, Bethesda, Md) under the following conditions: 10 minutes at 95°C activation/premelt, followed by 35 cycles of 30 seconds at 94°C melt, 30 seconds at 60°C anneal, and 3 minutes of 72°C extension. The polymerase chain reaction products were purified by using ExoSaplt (USB, Cleveland, Ohio) and sequenced at the University of North Carolina at Chapel Hill’s Genome Analysis Core facility.
Sequences were compared to wild type PTH1R (accession NM_000316.2 ) from GenBank release GRCh37 by using the BLAST algorithm. For our study, previously reported single nucleotide polymorphisms and synonymous substitutions in the coding region were ignored.
Results
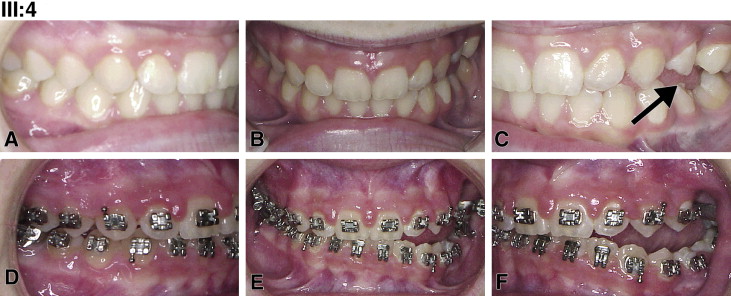
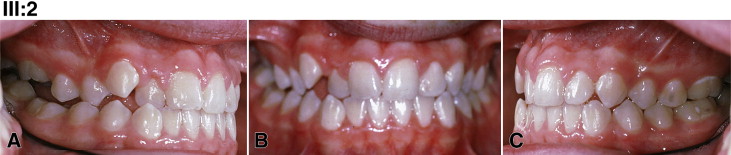
Pedigree analysis by visual inspection ( Fig 1 ) suggested an autosomal dominant inheritance with variable expressivity of PFE. For all examined persons, there was a complete absence of systemic disorders, and stature was within normal limits but varied as expected according to population norms. Two affected family members were classified as PFE type I (subjects III:3 and II:2); 2 were type II (subjects III:2 and III:4). In the family, the severity varied greatly. For example, the pretreatment records of subject III:4 showed a much less severe manifestation of PFE with only unilateral involvement ( Fig 2 , A-C ). Subjects III:4 and III:2 ( Fig 3 ) had previously been diagnosed with ankylosis rather than PFE, since the more severe classic appearance of PFE was lacking (only 2 or 3 affected teeth in 1 quadrant). This is in contrast to their full sibling, subject III:3 ( Fig 4 ), whose pretreatment photos showed a severe bilateral open bite that included the canines through the second molars. Subject II:2 ( Fig 5 , A-C ) was similar in severity to her son (subject III:3). In this family, an identical genetic diagnosis (see next section) was an equalizing element; although the clinical diagnoses were disparate between affected members, the resultant orthodontic failure was the same.

Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


