Fig. 10.1
Osteoclasts tunneling. Osteoclasts differentiate at cracks in the bone and form a tunnel (lacuna) containing a central capillary surrounded by connective tissue. Fibroblast-like precursors of osteoblasts are activated to become osteoblasts and new bone is formed (Modified from Fig. 22.54 in The Molecular Biology of the Cell. B. Alberts et al., 4th Ed. 2002. Garland Science, Taylor & Francis Group, New York)
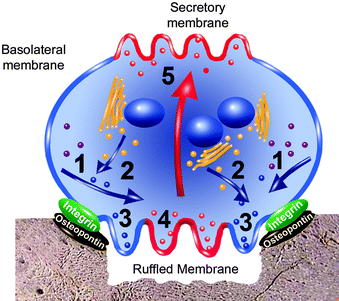
Fig. 10.2
Osteoclast membrane functions. Ruffled and secretory membranes differentiate from the outer osteoclast membrane at opposite ends (see text). Proteases, mostly cathepsin K and MMP-9 are synthesized in the endoplasmic reticulum (ER) (purple dots) and are diverted to lysosomes (blue dots – 1) because the proteases possess N-linked glycans that have attached mannose phosphate residues. Acid phosphatase (Acp5) is also synthesized in the ER and it passes into lysosomal trans-Golgi vesicles (yellow dots) along with the proteases – 2. The vesicles fuse to the cytosolic side of the ruffled membrane (near the integrin/osteopontin seal) and release their contents into the resorption compartment – 3. Protons and chloride ions are separately transported from the cytosol to the bone resorption compartment through this same region (Fig. 10.3). Calcium but not phosphate ions from demineralized bone, along with acid phosphatase and cathepsin K (red dots) enter acidic vesicles in the center of the ruffled membrane – 4. They pass through the cytosol (large red arrow) and are extruded into the extracellular fluid at the secretory membrane – 5 (Adapted from Fig. 1 in HK Väänänen, H Zhao, M Mulari and JM Halleen, “The Cell Biology of Osteoclast Function.” Journal of Cell Science. 113:377–381, 2000; Printed in Great Britain © The Company of Biologists Limited 2000 JCS1073: Reproduced with permission of the Company of Biologists ; Figure was modified by Dr. Wirsig-Weichmann)
The demineralizing compartment contains acidic, tissue-degrading enzymes (Sect. 3.3.2) that are secreted from lysosomes through the ruffled membrane. Demineralization and proteolysis in this compartment produce calcium ions and peptides that are endocytosed into acidic vesicles. These vesicles traverse the osteoclast ruffled membrane and cytosol and secrete their contents into the extracellular fluid where it can be picked up by blood capillaries (Fig. 10.2). Simultaneously (Fig. 10.3), transporter proteins move chloride ions from the extracellular fluid to the cytosol and from the cytosol into the demineralizing compartment. Other transporters move phosphate ions in the opposite direction. Protons from carbonic acid in the cytosol are exchanged for released sodium and dihydrogen phosphate. Increased cytosolic acidity likely restricts the osteoclast half-life to ∼1.3 days; new osteoclasts must be activated for demineralization to continue. Thus, once all the bone around a microcrack is removed, osteoclasts disappear and osteoblasts lay down new bone in response to the stresses, a process called remodeling (Sect. 9.2.1). Cementum and dentin behave similarly, but demineralized enamel cannot be remodeled because new ameloblasts cannot be generated.
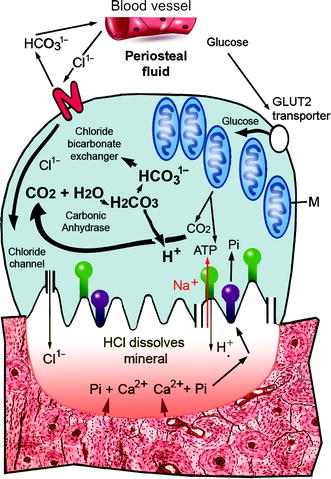

Fig. 10.3
Metabolite and ion exchange in osteoclasts. Glucose in extracellular fluid (top right) is transported into osteoclasts by a transporter protein (GLUT2). It is metabolized to pyruvate which enters mitochondria (M – center right) to provide ATP, CO2, and H2O. The ATP mediates a transfer of protons (H+) from carbonic acid (H2CO3) through the ruffled membrane in exchange for sodium (Na+) ions from demineralized bone (red). Sodium ions in the cytosol are transferred to the periosteum by the ATP-requiring Na+/K+ exchanger common to all cells (not shown). Carbonic anhydrase (CA) makes bicarbonate anions (HCO31−) from CO2 and H2O in the cytosol. The bicarbonate is exchanged for chloride ions from the extracellular fluid (Cl1−; top left). The chloride ions enter the sealed compartment through channel proteins in the ruffled membrane (bottom left and right). HCl accumulates in the demineralizing compartment and dissolves bone, releasing calcium (Ca2+) and dihydrogen phosphate ions and collagen polypeptides that are hydrolyzed by cathepsin K. The phosphate ions buffer the demineralizing compartment so that the pH remains at ∼4.5. A proton-dependent phosphate transporter moves sodium dihydrogen phosphate into the cytosol (purple) and it passes through the cell to the extracellular fluid by an unknown mechanism. A sodium ion-dependent type III phosphate transporter (Pit-2) in the basolateral membrane (Fig. 10.2) takes up sodium dihydrogen phosphate (Pi) from the extracellular fluid around and transfers it into the cytosol to increase pH. As also illustrated in Fig. 10.2, calcium ions are transported in acidic vesicles to the extracellular fluid (Modified from Figure 2 in Yamashita DS and Dodd RB (2000) “Cathepsin K and the Design of Inhibitors of Cathepsin K.” Curr. Pharmaceut. Des. 6:1–24). This figure was modified by Dr. Wirsig-Weichmann
10.1.1 Proteolysis in the Bone Resorbing Compartment
Hydrochloric acid in the demineralizing compartment dissolves the bone mineral and denatures exposed collagen fibers for hydrolysis by cathepsin K. This lysosomal endoprotease (Fig. 10.4a and b) has a cysteine thiol group at its catalytic center (Table 7.1; Fig. 10.4c). Cathepsin K is secreted in larger amounts than other lysosomal proteases and its specificity is due to unique amino acid residues around the peptide binding site. The enzyme hydrolyzes α1(I) and α2(I) polypeptides from incompletely denatured fibers in the demineralizing compartment, leaving the cross-linked, telopeptide ends (Sect. 4.2.2).
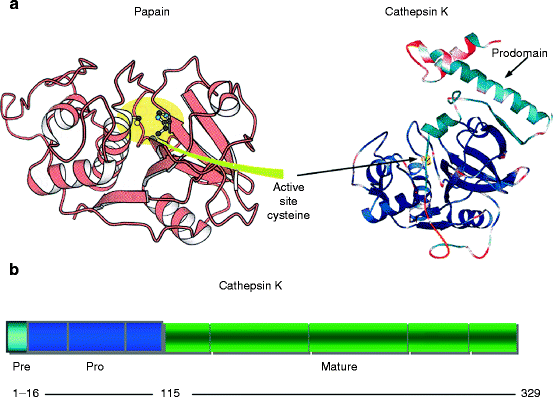
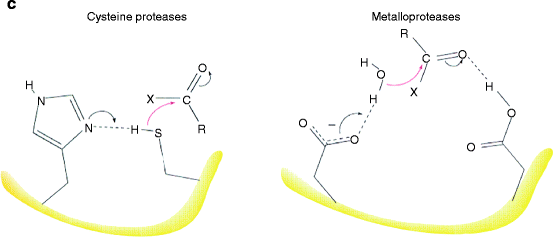


Fig. 10.4
Cathepsin K domain structure and mode of action. (a) Ribbon diagrams of papain and cathepsin K structures. Left – papain; right cathepsin K. Similarities of the respective structures and active site regions are obvious (Diagram of papain is part of Fig. 10.18 from Berg., et al., Biochemistry, 5th Ed. 2002, W.H. Freeman & Co., New York; diagram of cathepsin K is Fig. 1 from LaLonde (1999) “The Crystal Structure of Human Procathepsin K.” Biochemistry 38(3): 862–869). (b) Domain structure. Signal peptide for protein synthesis into the endoplasmic reticulum (ER) – light blue; propeptide inhibitor region – dark blue; mature protease – green. Gray bands indicate exon breaks. Numbers below indicate the number of amino acids in each domain (Modified from Fig. 1 in Donnarumma, M., et al. (2007) “Molecular analysis and characterization of nine novel CTSK mutations in twelve patients affected by pycnodysostosis. Mutation in brief #961.” Human Mutat. 28(5):524). (c) Catalytic mechanism of cysteine (papain family) proteases and metalloproteinases. The major classes of proteases are listed in Table 8.1. They use two fundamentally different catalytic mechanisms to stabilize a tetrahedral intermediate during polypep
Cathepsin K remains inactive in lysosomes because a propeptide occupies its active site, as in matrilysins (Sect. 8.1.3). Once secreted into the demineralizing compartment along with smaller amounts of other lysosomal proteases, the low pH loosens electrostatic and hydrogen bonds, promoting propeptide cleavage by autocatalysis or other protease action. Mutations that reduce or abolish cathepsin K activity are associated with pycnodysostosis, a rare genetic disease characterized in humans by a reduced stature and brittle (osteosclerotic) bones. MMP-9 (neutrophil gelatinase; Sect. 8.3.4) is another proteolytic enzyme in the demineralizing compartment of osteoclasts where it degrades type α1(II) chains left over from cartilage in endochondral-synthesized bones (Sect. 9.2.2).
10.1.2 10.1.3. Demineralization and remineralization
In addition to collagen peptides and calcium ions, demineralized bones release small amounts of transforming growth factor-beta, TGF-β (Sect. 3.2.2). TGF-β is not denatured by acid and not hydrolyzed by lysosomal proteases in the osteoclast demineralizing compartment. Its function is to be transferred transfer to the periosteum during demineralization where it stimulates new bone formation to complete the remodeling process. The TGF-β is endocytosed into acidic vacuoles along with peptides, calcium ions, and Acp5b. The acidic vacuoles pass through the center of the ruffled membrane and are exocytosed into the periosteum or bone marrow through the osteoclast secretory membrane (Fig. 10.2). A greater blood plasma content of Acp5b indicates increased osteoclast activity, but the best indicator of net bone loss is an increased level of type I collagen telopeptides containing pyridinoline (Sect. 4.2.2) A greater amount of telopeptide in blood plasma indicates a greater rate of bone demineralization, whereas a greater amount of osteocalcin indicates a greater rate of bone remineralization. A high telopeptide to osteocalcin ratio in blood plasma therefore indicates net bone demineralization in the body, whereas a low such ratio indicates net bone mineralization.
10.1.3 Osteoclast Ion and Proton Transport
The ruffled membrane is mostly composed of two chloride channel proteins: a chloride intracellular channel type 5 (ClIC-5) protein found in the endoplasmic reticulum (ER) and outer plasma membrane; and a chloride channel type 7 (ClC-7) protein homologous to voltage-gated chloride channels in the plasma membrane of nerves and muscles (Fig. 10.3). It also contains two other transporters: an ATPase that moves protons (H+) from the cytosol in exchange for Na ions (Na+), and a proton-dependent phosphate transporter that moves dihydrogen phosphate ions from the bone resorption compartment to the cytosol (Fig. 10.3). The calcium ions are moved out of the bone resorption compartment in acidic vesicles with digested collagen peptides and Acp5b. They are transported through the cytosol and secreted into the periosteum (or bone marrow).
The ATP produced by respiration is required to move the protons from the cytosol to the bone resorption compartment in exchange for sodium ions, fair amounts of which are released from demineralizing bone along with calcium and phosphate. The protons are produced with bicarbonate by carbonic anhydrase (CA), which utilizes the carbon dioxide and water from respiration. The CA catalytic site utilizes a zinc ion, like zincin proteases and alkaline phosphatase. The isozyme in osteoclast is identical to the carbonic anhydrase in red blood cells (CA-II). The sodium and bicarbonate ions in the cytosol are exchanged for extracellular chloride and potassium ions at the periosteal surface. Chloride ions flow from periosteum or bone marrow, through the cytosolic and ruffled membrane chloride transporters to the demineralizing compartment. Sodium ions flow in the opposite direction to the periosteum or bone marrow (Fig. 10.3). Protons and chloride ions accumulate within the bone resorption compartment, causing the pH to fall and the bone to demineralize. Phosphate and sodium ions from the bone are transported out from the ruffled membrane each at the expense of protons moving in and maintaining the demineralizing compartment’s acidity.
Despite the substantial amounts of Na+ ions released from demineralized bone and entering the cytosol in exchange for protons, excessive amounts of dihydrogen phosphate (H2PO4) also enter the cytosol. To compensate, there is a separate inward diffusion of disodium monohydrogen phosphate (Na2HPO4) from the periosteum/bone marrow through Pit-2 transporters (Sect. 9.3.5) in the osteoclast basolateral membrane (Fig. 10.2). Nevertheless, the pH eventually falls below levels compatible with mitochondrial function, perhaps explaining the osteoclast’s short half-life.
Summary
Stress-induced bony microcracks attract circulating white blood cells (monocytes) that differentiate into osteoclasts possessing an acidic compartment in contact with bone. This extracellular compartment is separated from the cytosol by a ruffled membrane attached by an integrin to osteopontin on the bone surface. Protons are produced by carbonic anhydrase acting on CO2 and H2O from respiration and exchanged through an ATP-dependent proton transporter for Na+ ions from the compartment. Bicarbonate ions in the cytosol are exchanged for periosteal chloride ions that pass on to the acidic compartment through chloride channel proteins in the ruffled membrane. HCl-demineralized collagen fibers are denatured and digested by cathepsin K, an acid-activated, papain-related gelatinase. Calcium ions, collagen peptides, and TGF-β from bone are taken up with cathepsin K-activated acid phosphatase into acid vesicles and exocytosed into the periosteum or bone marrow. Dihydrogen phosphate ions enter from bone through a proton-dependent phosphate transporter and pass through the cytosol separately to the periosteum or bone marrow. Once a microcrack is removed, TGF-β enhances osteoblast differentiation and a stronger layer of bone forms in response to the stress.
10.1.4 Osteoclast Differentiation
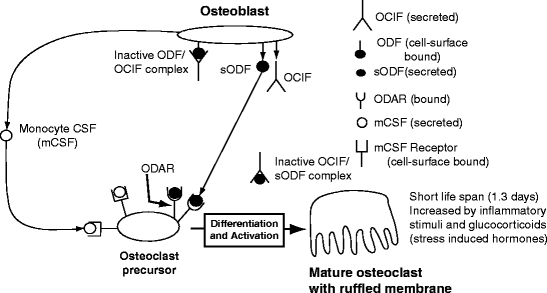
Osteoblasts around bony microcracks are induced to express two cytokines: monocyte Colony Stimulating Factor (mCSF) which is secreted and Osteoclast Differentiating Factor (ODF) which is mainly on the cell surface. The mCSF stimulates microcrack-adherent monocytes to proliferate and fuse into large multinucleated cells (preosteoclasts) that express Osteoclast Differentiation and Activation Receptor (ODAR).
When ODAR attaches to the osteoblast surface-bound ODF, the receptor/ligand complex activates a membrane-associated tyrosine-protein kinase to induce synthesis of the ruffled membrane. A tyrosine residue on ClIC-5 (Sect. 10.1.4) is phosphorylated by an activated protein kinase, called c-src, the normal cytosolic homologue of a viral tyrosine kinase which causes a sarcoma (transforms fibroblasts into cancer cells). The phosphorylated ClIC-5 interacts with phospholipids, a chloride channel protein (ClC-7) and two transporter proteins, the ATPase proton transporter, and the proton-dependent phosphate transporter. Mutations that suppress c-src or prevent expression or functioning of ClC-7 or ClIC-5 in mice or humans prevent osteoclast development and cause overly dense, brittle bones (osteopetrosis).
Bone resorption is a physiological process prevented by the osteoblast secreting osteoclast inhibition factor (OCIF), more commonly called osteoprotegerin (OPG). OCIF is a nonmembrane-bound decoy receptor that resembles ODAR and prevents ODF from binding to ODAR and therefore causing preosteoclasts to remain undifferentiated (Fig. 10.5). Bone resorption is related to the body’s response to injury or infection, a complex series of events called inflammation.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses