Fig. 5.1
Fluorescent-labeled risedronates were injected into 9-week-old rats and 24 h later, the tibia was harvested. Thin layers of the fluorescent activity were observed at the bone resorption lacunae (arrows) (From Roelofs et al. (2012))
The common structure of BPs contains two phosphates atoms connected by a hydroxycarbon backbone, which can form a structure responsible for the strong affinity to calcium ions. One remaining side chain (R2 chain) represents the molecular and functional variations of BPs. The current generation of BPs contains a nitrogen atom in the R2 chain and thus is called a nitrogen-containing BP or aminobisphosphonate (Fig. 5.2).
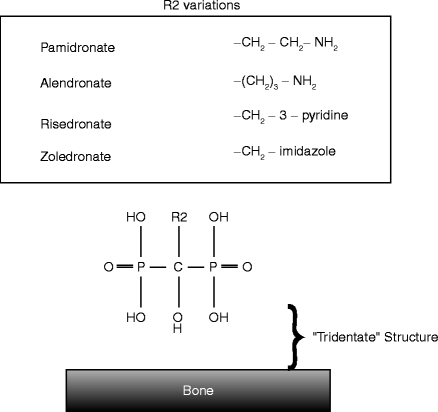

Fig. 5.2
The common structure of nitrogen-containing BPs. The strong affinity to hydroxyapatite is accommodated by two phosphates and hydroxyl R1 chain. The R2 chain varies among different BPs; however, the current generation of BPs contains a nitrogen molecule
The first experimental use of BP in humans was to treat a fibrodysplasia ossificans progressiva (FOP) patient. FOP patients carry rare genetic mutations in activin-like kinase-2 associated with BMP type 1 receptor, which causes heterotopic ossification in soft tissues such as thoracic muscles. Once the chest muscles are calcified, patients experience difficulty in inflating their lungs and may die from the resulting complications. The application of etidronate appeared to control the patient’s ectopic bone formation (Bassett et al. 1969).
Later, BP compounds became commercially available and were used for treating Paget’s disease (Altman et al. 1973) and osteoporosis (Harris et al. 1999). Daily or weekly oral administration of BP was well accepted. However, due to side effects affecting the esophagus, intravenous infusions of BP have become available. While different BP compounds were generated to treat these metabolic bone diseases, nitrogen-containing BPs were found to be far more effective in decreasing bone resorption. They also controlled hypercalcemia caused by bone-residing or metastasizing tumors such as multiple myeloma and breast cancer. For these patient groups, high doses of BP are administered by intravenous infusion.
Bisphosphonates are released when bone is resorbed, but the released BP subsequently decreases the osteoclastic activity leading to a decrease in bone resorption (Fig. 5.3). The molecular structure of nitrogen-containing BPs was found to be very similar with an endogenous compound, isopentenyl pyrophosphate (IPP). IPP is a metabolite in the mevalonate pathway synthesizing cholesterols in the cell. In order to maintain cellular functions and viability, IPP needs to be converted to farnesyl pyrophosphate (FPP) by FPP synthase. After BP is internalized in the cell, BP can inhibit FPP synthase due to its molecular similarity to IPP and effectively prevent the FPP synthesis. It has been demonstrated that FPP plays an essential role in functionalizing small GTPases by adding the lipid to these proteins. This process is called prenylation, by which small GTPases now become able to integrate in the cell membrane. These activated GTPases in the cell membrane are critical in organizing the actin ring of osteoclasts, allowing them to adhere to the bone surface and further develop the specific osteoclast morphology called the “ruffled border.” The lack of FPP results in the inadequate development of the osteoclastic cell morphology, leading to premature dehiscence as well as apoptosis (programmed cell death) (Itzstein et al. 2011; Li et al. 2011).
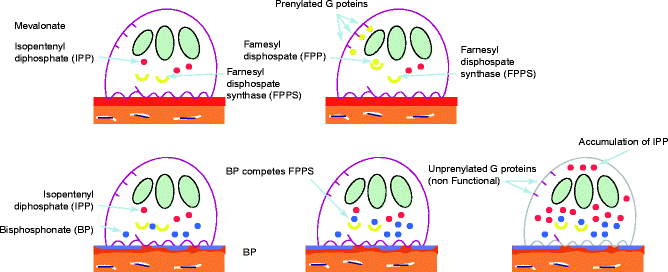

Fig. 5.3
Pharmacological effect of nitrogen-containing BP on osteoclasts. Osteoclasts maintain their viability through cholesterol prenylation of membrane-bound G proteins (above). Osteoclasts internalize BP through bone resorption and are thought to lose their viability due to the blocked mevalonate pathway by BP. BP presents a structural similarity to isopentenyl pyrophosphate (IPP) and can occupy farnesyl pyrophosphate synthase. As the result, the conversion from IPP to farnesyl pyrophosphate (FPP) is prevented (bottom). The lack of FPP significantly reduces GTPase prenylation and leads to premature termination of osteoclastic activities and cell death
5.2 Osteonecrosis of the Jaw (ONJ)
Remarkably, clinical trials of BPs in patients with breast cancer metastatic to bone and in patients with multiple myeloma indicated the beneficial effect on hypercalcemia, without causing immediate, serious side effects (Body et al. 1999). In 2003, however, an unusual oral condition became evident where necrotic bone was exposed for longer than 8 weeks among patients who were treated with BPs (Marx 2003; Ruggiero et al. 2004). This condition is called osteonecrosis of the jaw (ONJ).
The majority of ONJ cases were reported as a complication of tooth extraction procedures, which increased the odds of developing ONJ by 16 times (Kyrgidis et al. 2008). If possible, medical practitioners should consider referring patients for dental treatment so that tooth extractions can be undertaken prior to prescribing intravenous BP therapy (Weitzman et al. 2007). In those situations where tooth extraction is required, antibiotic prophylaxis has prevented ONJ (Montefusco et al. 2008).
Tooth extraction is known to induce transient but robust osteoclastogenesis at the periphery of the extraction socket (Fig. 5.4a). In normal wound healing following tooth extraction, the active osteoclastic bone resorption reduces the sharp edge of the socket and eventually forms a saddle shape residual ridge. It is highly conceivable that BP adsorbed onto the alveolar bone affects the viability of osteoclasts. Hokugo et al. (2010) generated the rat model of ONJ and reported that the prevalence of ONJ was positively associated with the abnormally increased number of osteoclasts in the tooth extraction area (Hokugo et al. 2010). Furthermore, during the early stages of ONJ pathogenesis, it was found that a cluster of inflammatory cells such as neutrophils and lymphocytes gathered around the osteoclasts on the surface of the alveolar bone (Fig. 5.4b). This unusual observation may suggest that the BP-affected osteoclasts could recruit the inflammatory/immune cells in the oral mucosa through yet unknown signaling mechanisms, and the activated inflammatory/immune cells then induce the cytotoxic effect to alveolar bone resulting in osteonecrosis.
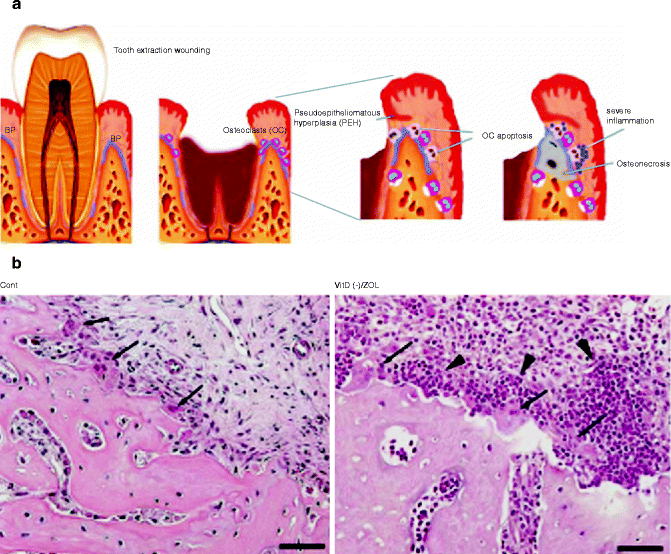

Fig. 5.4
(a) Hypothetical diagram of the pathological mechanism of osteonecrosis of the jaw (ONJ) following tooth extraction. (b) Osteoclasts (arrows) recruited as part of tooth extraction wound healing of rat alveolar bone (cont). In ONJ created by vitamin D deficiency and intravenous injection of zoledronate (VitD(−)/ZOL), osteoclasts (arrows) were surrounded by a cluster of neutrophils and lymphocytes (arrowheads) (From Hokugo et al. (2010))
Because ONJ biopsy specimens that had been exposed to the oral cavity consistently show bacterial infection, it has been proposed that the pathological development of ONJ may be initiated by infectious agents (Mawardi et al. 2011). However, spontaneous development of ONJ has been reported particularly at the palatal torus and mandibular posterior plate areas, where the bone exposure appears to be secondary to the osteonecrosis. Furthermore, removable dentures were found to increase the odds of developing ONJ without the primary bacterial infection (Kyrgidis et al. 2008). It is well established that extensive denture wearing results in the loss of residual ridge alveolar bone, which is caused by osteoclastic bone resorption. Therefore, while the necrotic bone exposure invites opportunistic infection, which should require treatment, the common phenomenon among these susceptible conditions may lie in the increased osteoclastogenesis, which may play a key role in the development of ONJ. Although still speculative, the BP-affected osteoclasts may abnormally activate the oral mucosa immunity, which then induces cytotoxic overreaction, leading to osteonecrosis and/or oral epithelial ulceration. The more powerful intravenous BPs such as zoledronate are mainly responsible for the vast majority of ONJ cases reported, 88 % in one review (Filleul et al. 2010).
While the adverse effects of zoledronate can be devastating, other beneficial antitumor effects in breast and prostate cancer cells in culture have been demonstrated. Zoledronate can inhibit bony metastasis in highly tumorigenic cell lines by reducing cell proliferation and increasing cell death (Almubarak et al. 2011).
An alternative hypothesis for the pathogenesis of ONJ proposed that inflammation and microcalcification of small blood vessels were responsible for the mucosal soft tissue ischemia (Meiller et al. 2012). In this theory, inflammation causes an acidic environment which would cause further release of calcium from its bound state with BP. This released calcium would induce more ischemia and mucosal ulceration, and a vicious circle ensues. This hypothesis would explain the difficulty in treating these lesions, but no new therapies are anticipated. Other theories have proposed that the primary effect of a potent BP is to cause premature senescence of oral mucosal cells (Kim et al. 2011b), thereby preventing wound closure following trauma.
The role of inflammation in the etiology of ONJ is unclear; however, severe periodontitis has been described as a risk factor (Yamazaki et al. 2012). Other studies have contrasted ONJ with other conditions that cause necrotic bone, such as suppurative osteomyelitis, and have described ONJ as having no inflammatory etiology but with drug toxicity as the main causative agent affecting normal bone turnover (Marx et al. 2012). Severe ONJ has been induced in rats given a potent BP (zoledronic acid), in the region where periodontitis had been induced with a ligature (Aghaloo et al. 2011). This would indicate that inflammation was a potent cofactor in the pathogenesis of ONJ. The features of this animal model included sequestration of necrotic alveolar bone in the inflamed region where plaque had accumulated around the ligature and extensive periosteal alveolar bone formation. These histological features are typically seen in ONJ-affected patients (Bedogni et al. 2008). Also, bone formation has been shown not to be affected in alendronate-treated rats (Kim et al. 2011a). It is unclear if ONJ could be completely prevented from occurring if oral hygiene was optimal.
The challenge is to identify those patients who have an early or subclinical bisphosphonate-associated ONJ rather than when necrotic bone becomes evident in the mouth. Before ONJ becomes evident, a 2–3-year symptom-free period is usually seen after treatment begins with high doses of BP. In one study of 24 patients, most of whom were treated with intravenous BP for malignancy, the mean length of time between starting treatment with BP and the development of clinical symptoms of ONJ was 31.8 months (Abu-Id et al. 2008). Other studies have shown similar lag times, e.g., (Kos et al. 2010), but the delay cannot be related to the toxicity of the BP used as trauma is a major factor in the clinical onset of ONJ. A subclinical ONJ may not be evident on an X-ray because a bone loss of about 30–50 % is necessary before it becomes radiographically detectable. The ONJ lesion can be lytic, sclerotic, or a mixture of both (Morag et al. 2009). Cone beam CT can detect increases in cortical thickness, but because the changes are nonspecific, it is not yet known whether the excellent 3-D image quality is useful in early diagnosis. Magnetic resonance imaging, combined with contrast agents, may provide a useful imaging modality for early diagnosis (Khosla et al. 2007), but its main use may be in assessing the extent of BP-induced disease (Garcia-Ferrer et al. 2008).
Zoledronic acid has been shown to have in vivo antiangiogenic properties by reducing vascular endothelial growth factor (VEGF) (Vincenzi et al. 2005). Mucosa from patients with ONJ have been shown to produce less VEGF than mucosa from patients with no necrosis (Mozzati et al. 2012), which would reduce the ability of the mucosa to heal following minor trauma. In this hypothesis, BP released from the underlying bone following minor trauma causes reduced VEGF production by the mucosa and new blood vessels are unable to form.
It is hoped that future research may produce a simple laboratory test, perhaps involving the noninvasive sampling of gingival crevicular fluid, which would allow the identification of ONJ before symptoms develop. The serum concentration of cross-linked C-terminal telopeptide of collagen type I has been recommended to assess the surgical risk in those taking BP, with values of less than 100 pg/mL, representing a high risk (Marx et al. 2007). However, patients who have developed BP-induced ONJ are found with normal values (Carini et al. 2012), (Conte-Neto et al. 2011). Bone turnover is suppressed in the majority of patients treated with BP (Eekman et al. 2011), but normal values of C-terminal telopeptide are seen in those with ONJ who continue receiving BP. For the individual patient, the C-terminal telopeptide test does not accurately predict the development of ONJ (Kunchur et al. 2009).
5.3 The Nonexposed Variant of Bisphosphonate-Induced ONJ
The American Association of Oral and Maxillofacial Surgeons has described the staging of bisphosphonate-induced ONJ and included a nonexposed variant. This stage is characterized by a lack of bone exposure but which may include a variety of nonspecific signs and symptoms such as alveolar bone expansion, dull jaw pain, and either bony sclerosis or osteolysis (Ruggiero et al. 2009). Osteosclerosis may be an important radiological sign as it was present consistently in clinically symptomatic areas of one third of these patients (Hutchinson et al. 2010). Other clinical symptoms associated with this condition include gingival swelling and a sinus tract, but their nonspecific nature makes it difficult to distinguish them from other dental infections. In a European study involving five centers, of those patients diagnosed with the nonexposed variant of ONJ, most (92 %) presented with jaw pain, about half (51 %) with a sinus tract, and about a third (36.4 %) with bone enlargement (Fedele et al. 2010).
5.4 Implant Osseointegration in BP-Treated Patients
Surgical manipulation of alveolar bone will induce inflammation and bone remodeling. In this process, if excessive numbers of inflammatory cells are recruited in the jawbone of BP-treated patients, there may be an increased chance of developing ONJ. There has been an intensive debate over whether surgical procedures involving osteotomy and implant fixture placement could also result in ONJ. Patients taking oral BP for longer than 3 years are at an increased risk of ONJ, especially if corticosteroids are also taken concurrently. The American Association of Oral and Maxillofacial Surgeons has advised that oral BP be discontinued 3 months before and after implant placement, if planned in consultation with the patient’s physician (Ruggiero et al. 2009). However, these practical guidelines are empirical and lacking high-quality evidence.
There is a clear trend that 1–10 % of cancer patients with intravenous infusion of high-dose BPs might experience ONJ (Reid and Cornish 2011). The intravenous infusion of high doses of BP presents a significant risk for ONJ, and thus patients receiving this treatment may not be suitable for surgical implant placement. On the contrary, osteoporosis patients taking oral BPs develop a much lower ONJ prevalence (Solomon et al. 2012). However, the number of osteoporotic patients with alendronate alone exceeds 20 million in the USA, and thus they are more relevant to routine dental treatment, including implant therapy.
Jeffcoat (2006) reported a single-blind controlled study involving 50 postmenopausal osteoporotic female patients with a total of 210 implants (Jeffcoat 2006). One half of the patients received oral BP treatment. Following implant placement, all patients were followed for 3 years or longer. None of the 102 implants in the BP patients were lost, whereas the success rate of the control patient group was 99.2 %. ONJ was not reported during this study.
A short-term follow-up study of 61 female patients receiving oral BPs was reported (Fugazzotto et al. 2007). One hundred and sixty-nine implants were placed in these patients either immediately after tooth extraction or in the edentulous area. At the 12- to 24-month follow-up examination, all implants were present and restored with appropriate prostheses. This study also reported that one patient exhibited 2 × 3-mm bone exposure in a mandibular torus adjacent to the first molar extraction site. The exposed bone was noticed at the 1-week post extraction examination and was debrided, and the implant placement took place 4 weeks later without any further complications. The ONJ prevalence in this report was therefore 1.6 %; however, no implant loss was experienced.
A direct mail survey was undertaken of 1,319 female patients over the age of 40, who had surgical implant placement (Grant et al. 2008). Out of 458 responses, 343 patients did not take oral BPs, whereas 115 patients reported the use of oral BP. Of these 115 patients, 89 patients reported the use of oral BP before implant surgery, and 26 patients started oral BP treatment after implant surgery. None of the patients responding to the survey reported symptoms related to ONJ. Two of the 86 patients experienced an episode of implant loss before osseointegration. Both of the failed implants were placed in the posterior region. However, the incidence of unsuccessful osseointegration in oral BP-treated patients appears to be within the normal range and similar to that of untreated patients.
A retrospective chart review was reported (Koka et al. 2010). Three hundred and seventy postmenopausal female patients received implant placement, and 69 patients listed oral BP as one of their medications. The subsequent telephone interview obtained the answers to the questionnaire from 55 patients with oral BP treatment and found that 1 out of 121 implants was lost.
These reports suggest that oral BP treatment does not influence implant osseointegration per se. Furthermore, the failed implants in patients treated with oral BP were not due to the development of ONJ. As such, the American Dental Association (ADA) issued a position paper in 2008 stating that oral BP treatment would not contraindicate dental implant treatment (Edwards et al. 2008).
5.5 Implant Failure in Patients Treated with BPs
Despite the assurance from ADA supported by the published reports, implant failures in those treated with BP continue to appear in the literature (Tables 5.1 and 5.2). The following are some of the highlights.
Table 5.1
Case reports in patients treated with BPs exhibiting implant failure prior to loading
|
Patient
|
Implant
|
Symptom
|
BP
|
Primary diagnosis
|
Biopsy
|
Treatment
|
Reference
|
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Age
|
Sex
|
Location
|
Surgery
|
BP drug holiday
|
Service
|
BP
|
Route
|
Duration
|
|||||
|
63
|
M
|
#4, #12
|
One stage
|
None reported
|
None specified: No loading
|
Pain (#12) after implantation; probing depth 12 mm (#4)
|
Alendronate
|
Oral
|
>10 years?
|
Osteoporosis, erosive osteoarthritis, history of wrist and thoracic spine fractures
|
None reported
|
#12 removed in 4 days; #4 removed in 3 weeks followed by teriparatide 20 μg SQ
|
Narongroeknawin et al. (2010)
|
|
65
|
F
|
#18, #19, #20, #29, #30
|
Prophylactic antibiotics, acetaminophen, ibuprofen as needed
|
None reported
|
6 weeks post implantation
|
Fluctuant swelling, rediolucency and large bone resorption (#9, #20): after the corrective surgery, necrotic bone exfoliated but otherwise no symptoms
|
Alendronate
|
Oral
|
>10 years
|
Osteoporosis, arthritis, history of hip fracture
|
None reported
|
Drainage, antibiotics, CHX followed by open degranulation, human mineralized cancellous bone graft with tetracyline
|
Wang et al. (2007)
|
|
62
|
F
|
#2, #3, #4, #6, #8, #9, #11, #13, #15
|
#2, #8, #9, #10, #11 extraction and socket preservation with freeze-dried bone; and sinus grafting prior to implant placement
|
None reported
|
2 month post implantation
|
Necrotic bone exfoliation at #11, #13, #15; bone defect at sinus floor; sinusitis
|
Risedronate (35 mg/week)
|
Oral
|
4 years
|
Osteoporosis
|
Biopsy, osteonecrosis, and chronically inflamed granulation tissue
|
Antibiotics, necrotic bone removal; open sinus exploration; #15 explantation
|
Brooks et al. (2007)
|
|
68
|
F
|
#21, #19, #18
|
None specified
|
None reported
|
1 year; 4 years
|
Pain, gingival bleeding 1 year after implant placement/ #9: gingival swelling, bleeding, radiolucency 4 year after implant placement
|
Alendronate
|
Oral
|
2 years at implant placement
|
Osteoporosis, rheumatoid arthritis
|
Biopsy, acute and chronic non-specific inflammation with necrotic bone fragment
|
Discontinuation of alendronate, curettage, antibiotics, CHX
|
Park et al. (2010)
|
|
65
|
F
|
#30, #31?
|
None specified
|
3 months
|
|||||||||
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


