Fig. 5.1
Diagram of a basal lamina. The various components are shown with reference to histological markers (bold) and their major biochemical components (small print). The lamina lucida and lamina densa are the light blue and dark green regions beneath a basal cell (epithelial or epidermal cell). The lamina lucida is usually composed of a laminin-1 network (light blue) within filaments of laminin-5 (white) (From Fig. 4 in Giehl KA, Ferguson, DJP, Dean D, Chuang, YH, Allen J, De Berker, DAR, Tosti, A., Dawber RPR and F.Wojnarowska, F. Alterations in the basement membrane zone in pili annulati hair follicles as demonstrated by electron microscopy and immunohistochemistry. British Journal of Dermatology, 150:722–727, 2004. With copyright permission from Wiley-Blackwell, PO Box 805, 9600 Garsington Road, Oxford OX4 2DQ, UK, as modified by Dr Wirsig-Wiechmann.)
Laminins are composed of three polypeptides, α, β, and γ that are homologous and encoded by separate genes in the mammalian genome. What genes are expressed is determined by tissue type. There are five different types of α chain, three types of β chain, and three types of γ chain, but fewer laminins than integrins (Sect. 4.4.1). The major laminin in the lamina lucida is laminin-1 (1α,1β,1γ; Fig. 5.2) which is secreted by fibroblasts, but its minor laminin, laminin-5 (5α1β1γ) is secreted by epithelial cells. The nomenclature for laminins was recently changed to indicate which of the alpha, beta and gamma chains are used in a particular laminin. Thus, the two laminins referred to above are now written laminin-111 and laminin-511. The older laminin nomenclature is used in this book. Laminin-1 assembles into a clear, web-like polymer (Fig. 5.2) above the lamina densa, whereas laminin-5 forms long, thin strands that pass through the lamina lucida and bind to type IV collagen in the lamina densa. A few laminin-5 strands pass right through lamina lucida and lamina densa and become attached to the head region of type VII collagen anchoring fibrils (see end of Sect. 5.1.2).
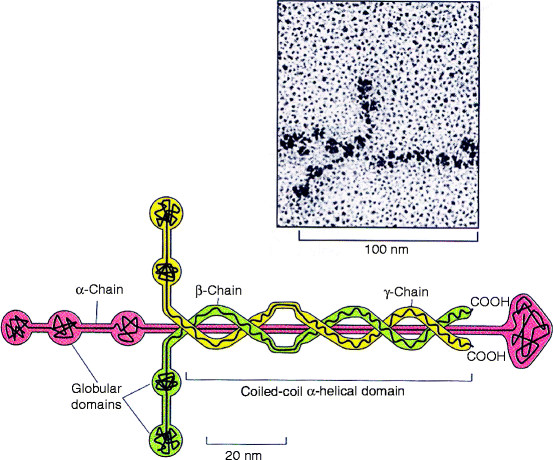

Fig. 5.2
Structure of laminin. Laminin is composed of three homologous polypeptides (α, β, and γ), each more than 1,500 amino acids long. They contain many domains that come together in a central, disulfide-bonded, triple helical coiled-coil with globular ends. Each trimer spontaneously interacts with others to form a web-like polymer illustrated in the electron microscope photomicrograph (upper right). Laminin-1 (α1, β1, γ1) forms an asymmetric cross-linked structure shown on the electron micrographs of laminin shadowed with platinum, whereas laminin-5 (α5, β1, γ1) forms a filament resembling a thin, barbell rod shown in Fig. 5.6 (Upper figure; Reprinted from J. Mol. Biol. Vol. 150, Shapes, domain organizations and flexibility of laminin and fibronectin, two multifunctional proteins of the extracellular matrix. Engel J, Odermatt E, Engel A, Madri JA, Furthmayr H, Rohde H, and Timpl R. Pages No. 97–120, 1981: with permission from Elsevier. Lower figure is adapted from Fig.19-57 in The Molecular Biology of the Cell. B. Alberts et al., 4th Ed. 2002. Garland Science, Taylor & Francis Group, New York)
Type VII collagen is a non-fibrous, anchoring fibril that binds large type I collagen fibers in the stroma to the lamina densa section of the basal lamina. Type VII procollagen has a 145-kDa N-terminal non-collagenous (NC1) domain, an extended central, triple-helical domain, and a short 34-kDa non-collagenous C-terminal (NC2) domain (Fig. 5.3a). The contribution of type VII procollagen to the mechanical stability of the dermal–epidermal junction depends on the ability of single molecules to self-assemble into highly ordered anchoring fibrils. On secretion, some of the C-terminal NC2 domain is proteolytically removed and cysteine cross-links form triple helical reverse dimers (Fig. 5.3b). At either end of a dimer, the large NC1 domain can interact with type IV collagen, type I collagen, or laminin-5. The interaction with type I collagen fibers may promote both type VII dimerization and fibril formation. As type VII fibrils form, the stromal ends bind to type IV collagen anchoring plaques deep within the stroma (Fig. 5.1).
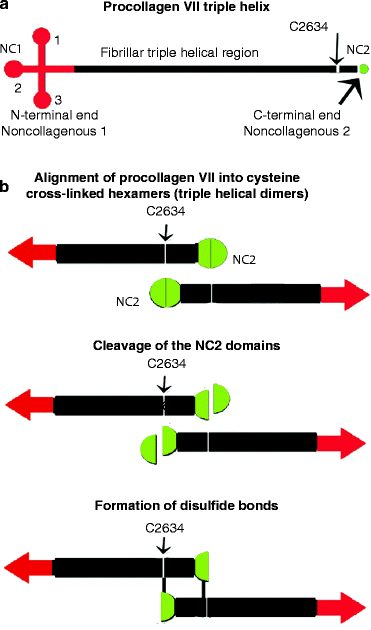

Fig. 5.3
Dimeric structure of collagen type VII anchoring fibrils. (a) Type VII collagen triple helix. The non-collagenous (NC) N- and C-terminal procollagen domains (NC1 and NC2 regions) are respectively colored red and green. (b) Fibril formation. Antiparallel binding of two triple helices may be orientated by each helix binding to a type I collagen fiber followed by proteolytic cleavage within the C-terminal non-collagenous region (NC2 region). The cysteine at residue 2634 aligns with a cysteine residue near the cleaved C-terminus so that two triple helical regions are correctly cross-linked to form a hexamer from which a triple helix extends in either direction (Modified from Figs. 1 and 2 in R. Brittingham et al. (2005 Jan 7) “Single amino acid substitutions in procollagen VII affect early stages of assembly of anchoring fibrils.” J. Biol. Chem. 280(1):191–198)
Type IV collagen is responsible for the dense network that characterizes the lamina densa and anchoring plaques in all mammals. It is made up from two or three of six homologous α-procollagen type IV polypeptide chains (Fig. 5.4a) that assemble at their C-terminal procollagen domain into one of three heterotrimers called protomers (Fig. 5.4b): [α1(IV)]2[α2(IV)] beneath an epidermis such as the gingival mucosa or skin; [α3(IV)] [α4(IV)][α5(IV)] in the glomerulus of the kidney and lungs; and [α5(IV)]2[α6(IV)] around smooth muscle cells. Each individual polypeptide has a long, collagenous domain (gly-X-Y repeats) but frequent interruptions reduce rigidity. The C-terminal non-collagenous region (NC1 region) of the three polypeptides making up each protomer (Fig. 5.4C) links the C-terminal end of each pair of protomers (Fig. 5.4D).
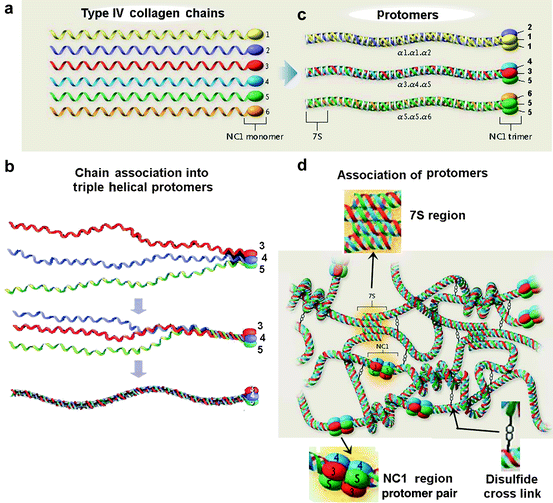

Fig. 5.4
Schematic illustration of type IV collagen supramolecular network assembly. (a) Type IV collagen chains. There are 6 α-chains (α1-6) encoded in the mammalian genome, each characterized by a long central collagen triple helix, a 7S domain at the N terminus, and a globular, non-collagenous (NC) trimer at the C terminus (NC1). (b) Association into protomers. The NCl domain at the C-terminus (Type IV collagen has only one non-collagenous domain) induces triple helix formation from C- to N-terminus in a protomer. The formation of protomer [α3,α4,α5] is illustrated. (c) Only a few protomers form. The ability of only certain 7S or NCl domains to associate [(α1)2 α2], [α3,α4,α5] and [(α5)2 α5] explains the limited triple-helical associations of the polypeptides. (d) Network formation and cross-linking. The supramolecular network is assembled by four protomers associating at the non-collagenous 7S domain, followed by dimerization and disulfide cross-linking at the C-terminus. (b – Slightly modified from Fig. 1B from Borza DB, Hudson BG (2003 May) “Molecular characterization of the target antigens of anti-glomerular basement membrane antibody disease.” Springer Semin Immunopathol. 24(4):345–361); a, c, d – Slightly modified from Hudson BG, Tryggvason K et. al. (2003 Jun 19) “Alport’s Syndrome, Goodpasture Syndrome, and Type IV Collagen.” N. Engl. J. Med. 348(25):2543–2556)
5.2 Hemidesmosomal Proteins
Hemidesmosomes (HDs) are membrane-associated adhesive junctions linked to the filamentous networks of the epithelial cell cytoskeleton and the lamina lucida ( light green/dark blue region in Fig. 5.1). The cytoskeleton of all mammalian cells is composed of three kinds of filaments: microfilaments, intermediate filaments and microtubules. Microfilaments are thin and made of actin, whereas microtubules are thick and made of tubulin. Intermediate filaments are of intermediate size (∼10 nm thick) and they include keratins in epithelial cells (which also secrete them to form hair and nails); nuclear lamins (which form a network that stabilizes the inner membrane of the nuclear envelope); neurofilaments (which strengthen the long axons of neurons); and vimentins (which provide mechanical strength to muscle and other cells). All intermediate filaments possess a pronounced α-helix. Keratins are described in detail in Sect. 5.2.2).
Hemidesmosomes are made up of four proteins: two bullous pemphigoid (BP) antigens, (BP180, an anchoring fibril identified as type XVII collagen, and BP230, a plakin protein), and two other proteins; plectin (a second plakin protein), and integrin α6β4. BP antigens are proteins that function abnormally in the disease, epidermolysis bullosa (Sect. 5.1.3.). Plakin proteins link intracellular keratin intermediate filaments to hemidesmosomes of basal epidermal cells, or to desmosomes on suprabasal epidermal cells (Sect. 5.2.1), whereas type XVII collagen (the only collagen secreted by epithelial cells) and integrins attach hemidesmosomes to a basal lamina. Three β1 integrins (with α2, α3 or α5 partners) together with integrin α6β4 can attach any of the laminins in the lamina lucida, not just laminin-5. The β1 integrins attach laminin-5 by various laminin motifs, not exclusively the arg-gly-asp (RGD) motif described in Sect. 3.2.1.
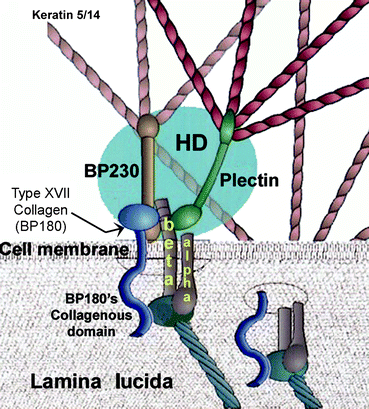

Fig. 5.5
Model of the hemidesmosome (HD)–basal lamina junction. This is an enlarged region of one of the hemidesmosomes pictured in Fig. 5.1. There are 4 components: integrin alpha and beta polypeptides (dark brown/yellow typescript), BP180 (type XVII collagen, dark blue tadpole-shaped), BP230 (light brown) and plectin (dark green). They all cluster together around the intracellular side of the integrin beta polypeptide within a hemidesmosome (HD, blue oval). The head portion of type XVII collagen (BP180) is attached intracellularly along with BP230 to the β-integrin subunit. The tail portion extends through the cell membrane alongside the β-integrin subunit and together they enfold a laminin-5 head (dark bluish gray). The β1 integrins which are present in addition to integrin α6β4 bind to many other lamina lucida components besides laminin-5, and initiate keratinocyte differentiation intracellularly (see text). BP230 and plectin are each connected to keratin 5/14 dimers intracellularly and therefore attach the hemidesmosome to keratin intermediate filaments that hold an epithelium together with desmosomes (Sect. 5.2). (Modified from Fig. 8 in Colognato H, Yurchenko PD (2000 Jun) “Form and Function: the Laminin Family of Heterotrimers.” Dev. Dyn. 218(2):213–234)
Type XVII collagen is composed of three identical procollagen polypeptides, each about 1,500 amino acids in length. It has a tadpole-like shape under physiological conditions (deduced from rotary shadowing electron microscopy of bovine cell lines or the pure protein). The protein is one of very few proteins whose N- and C-termini are inverted with respect to the membrane
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


