Chapter 9 Oral fungal infections
Candida species are fungi that are frequently encountered in the mouth of healthy individuals and as such can be considered to be normal residents of the oral microflora. The actual incidence of oral candidal carriage is estimated to be between 35 and 55% of healthy individuals, depending on the population group studied. Other fungi, such as Saccharomyces spp., Geotrichum spp. and Cryptococcus spp. are also encountered (Table 9.1), but their occurrences are rare and these are not generally implicated with oral infection. While Candida species are normally harmless commensals, when conditions in the mouth alter to one that favours proliferation of Candida, a shift to a pathogenic state can occur. As such, Candida infection is invariably an opportunistic one dependent upon some form of underlying host predisposition. Infection with Candida is described in the literature as a candidosis (candidoses, pl) or candidiasis (candidiases, pl). Both terms are widely used, although candidosis is often preferred due to its consistent use of the ‘osis’ stem with the terminology for other fungal infections. The term Candida originates from the Latin word candid, meaning white.
| Candida species | Other fungal species (rare) |
|---|---|
| Candida albicans | Paracoccidioides brasiliensis |
| Candida glabrata | Aspergillus spp. |
| Candida tropicalis | Cryptococcus neoformans |
| Candida krusei | Histoplasma capsulatum |
| Candida lusitaniae | Mucor spp. |
| Candida dubliniensis | Saccharomyces spp. |
| Candida kefyr | Geotrichum spp. |
| Candida guilliermondii | Rhizopus spp. |
| Candida parapsilosis |
Pathogenic Candida species
The genus Candida contains over 200 different species that are ubiquitously distributed. However, only a few of these have been implicated in human infection. The most prevalent Candida species recovered from the human mouth, in both commensal state and cases of oral candidosis, is Candida albicans. It is estimated that this species accounts for over 80% of all oral yeast isolates. In terms of oral prevalence, C. albicans is followed by C. glabrata, C. krusei, C. tropicalis, C. guilliermondii,C. kefyr and C. parapsilosis. In more recent years there has been a greater recognition of the importance of the non-albicans Candida species in human disease. Candida glabrata and C. krusei are species that have received attention due to their enhanced resistance to certain antifungal agents. Candida dubliniensis is a recently identified pathogenic species, first described in 1995 when it was co-isolated with C. albicans from cases of oral candidosis in HIV-infected individuals.
Candida virulence factors
The transition of Candida from a harmless commensal to a pathogenic organism is complex and may relate to subtle environmental changes that lead to the expression of a range of virulence factors (Table 9.2). It is likely that it is the combined effect of both host and candidal factors that ultimately contribute to the development of oral candidosis.
Table 9.2 Putative virulence factors associated with Candida albicans.
| Virulence factor | Effect |
|---|---|
| Adherence | Promotes retention in the mouth |
| Evasion of host defences | Promotes retention in the mouth |
|---|---|
| Invasion and destruction of host tissue | Enhances pathogenicity |
|---|---|
Adherence
The cell surface molecules on Candida that are involved in its specific adherence are described as adhesins (Ch. 4). The host cell component that these adhesins interact with, are referred to as receptors. A wide range of adhesins have been identified for C. albicans and include mannoproteins, fibrillar adhesins that bind to fucosyl receptors and proteins that bind to complement receptors on host cells. Candidal adherence is a complex and multifactorial process, dependent on both host and candidal characteristics.
Morphology
Candida have the ability to grow in several morphological states including budding yeast cells, pseudohyphae (elongated chains of yeast cells), and also true filamentous hyphae (Fig. 9.1). Pseudohyphae and hyphae are distinguishable as the former contains constrictions at the septa of the filaments. Once attached to host surfaces the ability of Candida and in particular C. albicans to switch from its yeast morphology to a filamentous form may promote penetration of the epithelium and increase resistance of cells to phagocytosis by host immune cells.
Oral candidosis
Candidosis is a ‘disease of the diseased’ which highlights the importance of host-related factors on the development of oral candidosis. This is most clearly exemplified by the high incidence of oral candidosis in Human Immunodeficiency Virus (HIV)-positive individuals and Acquired Immunodeficiency Syndrome (AIDS) sufferers. However, transition from healthy oral carriage to a diseased state can be triggered by less extreme changes within the oral environment and numerous other factors have been implicated (Table 9.3).
Table 9.3 Host related factors associated with oral candidosis.
| Suggested host factor |
|---|
| Local host factors |
| Systemic host factors |
|---|
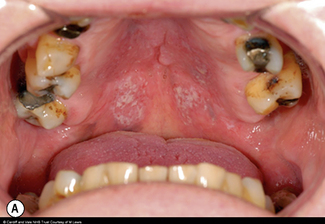
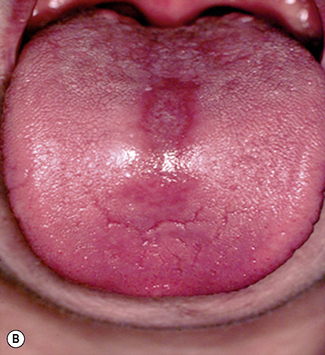
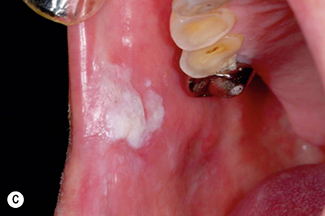
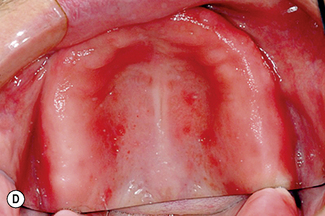
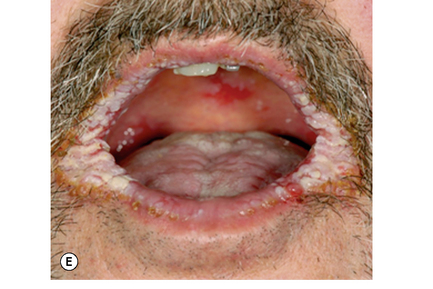
Oral candidosis is not a single entity, and four distinct primary oral candidoses are frequently described, based on their clinical presentation (Fig.9.2).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses