Dental Ceramics
Chapter Objectives
On completion of this chapter, the student will be able to:
1. Describe the mechanism for bonding porcelain to metal for porcelain-fused-to-metal (PFM) crowns.
2. List the types of alloys used in PFM crowns.
3. Describe where most failures occur in PFM crowns.
4. List the methods of fabrication for all-ceramic restorations.
5. Explain how CAD/CAM technology is used to “make impressions” and fabricate a ceramic crown.
6. List the indications for porcelain veneers.
7. Define chroma, value, and hue.
8. Describe steps to be taken to ensure proper conditions for shade taking.
Key Terms defined within the chapter
Porcelain-Metal Restoration restoration that has a metal core over which porcelain is fused at high temperature
All-Ceramic Restoration ceramic restoration with no metal core
Heat-Pressing pressing ceramic material into a mold at high temperature and pressure
Hue the basic color of an object (e.g., red, yellow, green, blue)
Value the brightness of a color or its lightness or darkness
Ceramics were introduced into dentistry over a hundred years ago with the advent of porcelain jacket crowns and inlays. These were very esthetic materials but were prone to fracture because they are brittle and weak when used in high-function areas of the mouth. For the first half of the 20th century, amalgam and gold were the primary restorative materials for posterior teeth. Some anterior teeth also had metal in the form of gold margins of three-quarter crowns, class III gold foils, or class V inlays or amalgams that were visible when the patient smiled. In the latter half of the 20th century, a variety of direct-placement tooth-colored restorative materials were introduced. In the 1960s, composite resins were introduced and have been continually improved upon ever since by making them more durable and color stable. All-ceramic materials have been introduced within the past three decades that are much stronger than original porcelains with some improvements in esthetics. As a result, the use of all-ceramic restorations has dramatically increased. Currently, ceramic materials are used in a variety of restorations, such as crowns, inlays, onlays, and veneers. Selection of the type of material to be used depends, in part, on the extent of damage to the tooth, the stresses that will be placed on the restoration, and the esthetic requirements of the patient.
By enhancing the ability to bond restorative materials to metal and tooth structure, advances in esthetic materials and techniques have assisted the dental team in delivering the esthetic results that patients demand. The dental team must keep current with the rapid changes that occur in materials and techniques. Good listening skills are needed to determine the types of esthetic services the patient is requesting, so that the dental team and the patient are working in concert toward the same goal. Esthetic materials must be carefully selected, so that their properties are compatible with the patient’s oral condition and occlusion. Dental hygienists and dental assistants must understand the properties of these materials, so that as important members of the dental team, they can help the dentist to assess the performance of the restorations and alert the dentist when they perceive that a restoration may be failing. They need to be familiar with the physical properties of materials, so that they do not damage the restorations during routine oral hygiene, coronal polishing, and preventive procedures. Dental assistants need to know the handling characteristics of esthetic materials, so that they can either assist the dentist in their placement or perform steps in their placement as permitted by state dental practice acts. Additionally, they will be called upon to assist in shade taking for the restorations.
This chapter describes the physical properties, clinical applications, and shortcomings of esthetic ceramic materials. Guidelines for selection of the shade of these materials to obtain satisfactory cosmetic results also are discussed.
Dental Ceramics (Porcelain)
The general term ceramics is used to describe porcelain and a variety of new materials that are similar in appearance to porcelain but vary in their composition, mode of fabrication, and physical properties. Porcelain is a term that has been used in dentistry for many years to describe the class of tooth-colored dental materials that are composed of feldspar, quartz, and kaolin; are glass-like; and are fired at high temperatures.
Feldspathic Porcelain
Until the recent advances in ceramic materials were made, the dental ceramic most commonly used was feldspathic porcelain manufactured from fine crystalline powders of alumina, feldspar, and silica mixed with a flux of sodium or lithium carbonate. As the powder is heated to certain critical temperatures, the porcelain particles fuse together to form a type of glass. Within this glass, leucite crystals are formed from feldspar. Leucite increases the hardness of the porcelain and its fusing temperatures. The porcelain is manufactured in a variety of colors that are produced from the addition of metal oxides to create the different colors (shades) that will match the teeth. The laboratory technician selects powders based on the shade prescription provided by the dentist. These porcelains were initially used for all-porcelain jacket crowns. These jacket crowns were very esthetically pleasing but had a high fracture rate. Later, the feldspathic porcelains were used in conjunction with a cast metal substructure to produce the porcelain-fused-to-metal (PFM) crown. Porcelains can be classified according to their fusing temperature as high fusing (2360°F to 2500°F [1288°C to 1371°C]), medium fusing (2000°F to 2300°F [1093°C to 1260°C]), and low fusing (1600°F to 1950°F [871°C to 1066°C]) (see Figure 6-12). High-fusing porcelain is used most often for the manufacture of denture teeth.
Porcelain-Metal Restorations
The most commonly used restorations in fixed (crown and bridge) prosthodontics are combinations of porcelain and metal (porcelain-metal restorations). Before the development of the porcelain-fused-to-metal technique in the 1950s, all-porcelain jacket crowns or metal crowns with acrylic or porcelain facings were used most often in the esthetic zone of the mouth. Acrylic facings stain and wear over time. Cemented porcelain facings often fracture or come uncemented. The main advantages of the combination of porcelain and metal are the strength and durability given to the restoration by the bond between a metal internal core and the esthetic external porcelain covering. The restorations are strong enough to be used in the posterior part of the mouth, where biting forces are greater than in the anterior, and can be provided as single crowns or multi-unit bridges.
Low-fusing porcelain is used for the bonding of porcelain to metal, which occurs through an oxide on the metal surface. These restorations are referred to as porcelain-fused-to-metal (PFM) or porcelain-bonded-to-metal (PBM) restorations. The metals that are used as the core for the PBM/PFM crowns are alloys of specific metals that will form an oxide layer as the metal is heated. When porcelain is applied to the metal and the two materials are heated together, the porcelain chemically fuses to the oxides on the metal, forming a durable bond. The metal alloys are classified as high noble (precious), noble (semiprecious), or base (nonprecious) metal alloys, based on the presence and amount of gold, palladium, and other precious metals (see Chapter 10). The metal in the area where the porcelain is to be bonded is usually relatively thin, approximately 0.3 to 0.5 mm thick.
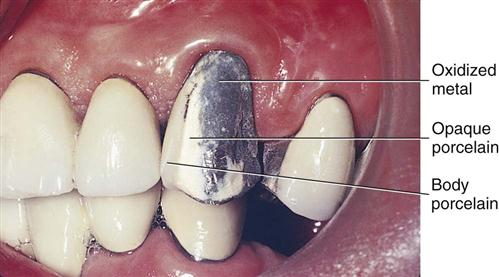
A color of porcelain is selected that corresponds to the color or shade that matches the patient’s dentition. The first layer of porcelain applied to the metal is an opaque porcelain that keeps the oxide color from showing through the porcelain and is the main color used for the crown (Figure 8-1). Base metals often form darker oxides that are more difficult to hide with the opaque porcelain. The porcelain comes as a powder that is mixed into a paste with water or a water-based liquid or may be in a paste form already. The porcelain paste is applied (or stacked) on the metal and is shaped and blotted to remove excess moisture. The oxidized metal and porcelain are heated under a vacuum at temperatures ranging from 870°C to 1370°C, depending on whether the porcelain to be used fuses at low, medium, or high temperature. The porcelain particles melt at their borders and fuse together (called sintering) and also wet the metal oxides. The oxides and porcelain chemically fuse together and mechanically interlock. Sintering results in shrinkage of the porcelain mass by about 25% to 40%. Then additional layers of porcelain called body and incisal porcelains are built up or stacked to simulate dentin and enamel colors and translucency (Figure 8-2). The incisal porcelain is more translucent, so the body color shows through readily and has a greater influence on the final appearance. The layers of porcelain are fired in the oven until they fuse to each other and to the underlying opaque porcelain. After final contouring of the crown, another firing maintaining the temperature at the fusing temperature for a while will produce a surface glaze. Glazing provides a dense, shiny, smooth surface. Some technicians use a layer of special translucent porcelain that fuses at relatively low temperatures to form the glazed surface. This is called overglazing.
Occasionally, special porcelain stains containing metal oxides are used on the surface to help in matching the natural tooth color and characteristics such as discolorations, white spots, or fine crack lines in enamel. These stains are fused to the porcelain during the firing process. Very translucent porcelains can be used on incisal edges to replicate the translucency often found on the incisal edges of maxillary incisors.
Porcelain Failures
Most porcelain failures result from small cracks in the porcelain that develop when the porcelain is put under occlusal loading, and they propagate (spread) over time until the porcelain gives way. Other modes of failure are caused by problems related to the chemical bond between the porcelain and the metal oxides. The oxide layer may be too thick or inadequate in quantity and quality, leading to failure at the interface of the porcelain and metal. It is important for the coefficient of thermal expansion of the porcelain and the metal to be compatible. The best arrangement is for the porcelain to have slightly less thermal expansion than the metal. This will keep it from cracking at the metal-porcelain interface and will reduce the chance of failure (see Figure 8-1). When porcelain failures occur in non–stress-bearing areas, repairs may be possible using composite resin and bonding techniques, but the repairs are not as strong as the original bonded porcelain (see Chapter 5) and may fail if put under too much biting pressure. The alternative is to do expensive replacements of the entire crown or bridge.
The porcelain surface, once it has been fused under temperature, is very hard and smooth. When porcelain or PBM restorations are delivered, the proximal contacts or occlusal surfaces often must be adjusted. These restorations could be returned to the laboratory to be reglazed (refired at porcelain-fusing temperatures to form a glassy surface) before cementing. Because this is seldom practical, a variety of abrasives have been developed for polishing the porcelain surface after adjustment (see Chapter 12).
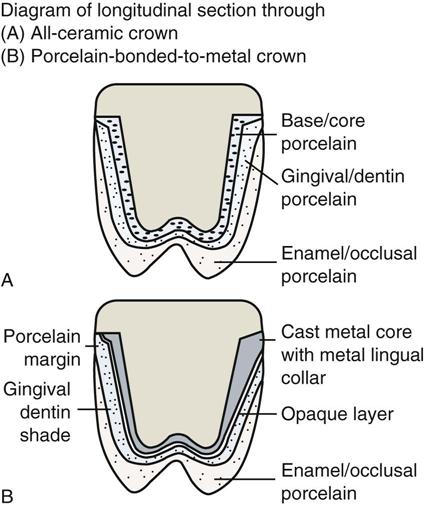
The low-fusing porcelains are less abrasive to opposing tooth structure and can be repolished easier after adjustments. All-ceramic crowns have a more life-like appearance (sometimes called vitality) than PBM crowns, because they do not have an opaque layer to hide metal. Ceramics appear vital (similar to natural teeth), because they are fluorescent, that is, they emit light in the visible wave spectrum when ultraviolet light hits them. They are also opalescent because they take on a bluish tinge when light reflects off them, and an orange-yellow tinge when light passes through them. Strong core porcelain acts as the foundation, and conventional “stacked” porcelains are added to enhance the esthetics.
All-Ceramic Materials
New ceramic materials have been developed that are stronger, and when used in conjunction with resin bonding to tooth structure, they have found wider application, including use in the posterior region of the mouth. The all-ceramic restorations have wide acceptance because of their excellent esthetic appearance, biocompatibility, low wear rate, and stain resistance. They can be used for inlays, onlays, crowns, and veneers. The major drawback of ceramic restorations is their potential to fracture. The fracture rate for all-ceramic crowns in the posterior part of the mouth is higher than for PFM crowns. Although ceramic materials are very strong in compression, they are brittle and weak when placed in tension (pulling). What is more important is their transverse strength (a combination of compressive and tensile strength), which is a better indicator of their resistance to fracture. The transverse strength of dental ceramics is in the range of 50 to 600 MPa (megaPascals; 1 MPa = 150 pounds per square inch). The traverse strength varies with the type of ceramic material used. When ceramic restorations are placed in parts of the mouth where they are under heavy loading, they are at greater risk for fracture. Many patients are willing to accept the risk of ceramic fracture to obtain the esthetic appearance they desire.
Ceramic Dental Materials
Dental ceramics have undergone numerous changes over the past three decades to improve their strength and esthetics. They can be clas/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses