The biological effects associated with X-rays, risk and practical radiation protection
Radiation-induced tissue damage
The action of radiation on cells and the resultant damage are classified as:
• Direct action or damage as a result of ionization of macromolecules
• Indirect action or damage as a result of the free radicals produced by the ionization of water.
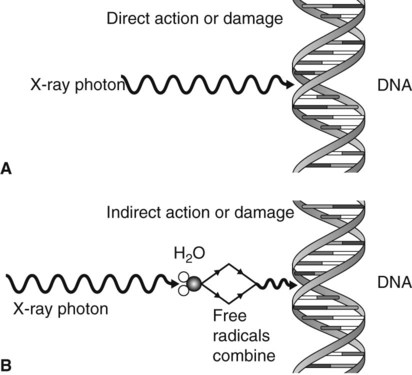
Direct action or damage
The X-ray photons, or high-energy ejected electrons, interact directly with, and ionize, vital biologic macromolecules such as DNA, RNA, proteins and enzymes, as shown in Fig. 7.1A. This ionization results in the breakage of the macromolecule’s chemical bonds, causing them to become abnormal structures, which may in turn lead to inappropriate chemical reactions. Rupture of one of the chemical bonds in a DNA macromolecule may sever one of the side chains of the ladder-like structure. This type of injury to DNA is called a point mutation. The subsequent chromosomal effects from direct damage could include:
• Inability to pass on information
• Only temporary damage – the DNA being repaired successfully before further cell division.
If the radiation directly affects somatic cells, the effects on the DNA (and hence the chromosomes) could result in a radiation-induced malignancy. If the damage is to reproductive stem cells, the result could be a radiation-induced congenital abnormality.
What actually happens in the cell depends on several factors, including:
Indirect action or damage
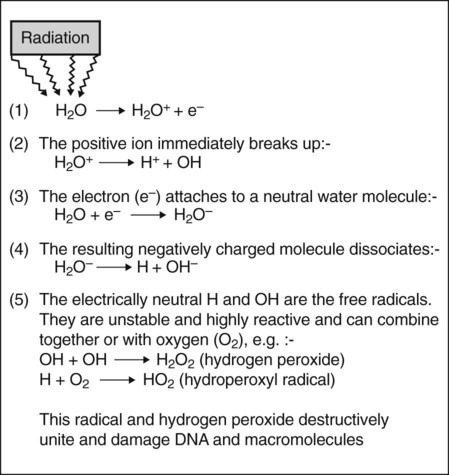
This process, which is shown in Fig. 7.1B, involves the ionization of the water molecule producing both ions and free radicals which can combine to damage the vital biologic macromolecules such as DNA. The sequence of events involved is summarized in Fig. 7.2. The free radicals can recombine to form hydrogen peroxide, a cellular poison, and a hydroperoxyl radical, another toxic substance. Both of these substances are highly reactive and produce biological damage. By themselves, free radicals may transfer excess energy to other molecules, thereby breaking their chemical bonds and having an even greater effect. As about 80% of the body consists of water, the vast majority of the interactions with ionizing radiation are indirect.
Classification of the biological effects
Stochastic effects
Cancer induction
• The survivors of the atomic explosions at Hiroshima and Nagasaki
• Patients receiving radiotherapy
• Radiation workers – people exposed to radiation in the course of their work
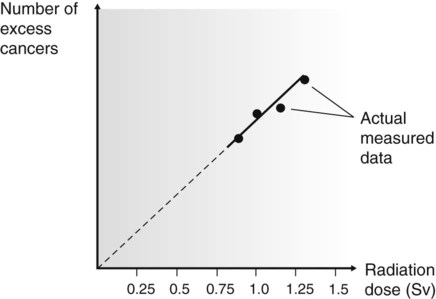
The problem of quantifying the risk is compounded because cancer is a common disease, so in any group of individuals studied there is likely to be some incidence of cancer. In the groups listed above, that have been exposed to high doses of radiation, the incidence of cancer is likely to be increased and is referred to as the excess cancer incidence. From the data collected, it has been possible to construct dose–response curves (Fig. 7.3), showing the relationship between excess cancers and radiation dose. The graphs can be extrapolated to zero (the controversy on risk assessment revolves around exactly how this extrapolation should be done), and a risk factor for induction of cancer by low doses of radiation can be calculated.
After reviewing all the available evidence, the International Commission on Radiological Protection suggest there is a 1 in 20,000 chance of developing a fatal cancer for every 1 mSv of effective dose. Using this estimate, a broad estimate of risk from various X-ray examinations may be calculated and these are shown in Table 7.1.
Table 7.1
| X-ray examination | Estimated risk of fatal cancer |
| Bitewing/periapical radiograph (70 kV, round collimation, D-speed film) | 1 in 1,000,000 |
| Bitewing/periapical radiograph (70 kV, rectangular collimation, F-speed film) | 1 in 10,000,000 |
| Panoramic radiograph (average) | 1 in 1,000,000 |
| Upper standard occlusal | 1 in 2,500,000 |
| Lateral cephalometric radiograph | 1 in 5,000,000 |
| Skull radiograph (PA) | 1 in 1,000,000 |
| Skull radiograph (lateral) | 1 in 1,250,000 |
| Chest (PA) | 1 in 1,430,000 |
| Chest (lateral) | 1 in 540,000 |
| CT head | 1 in 14,300 |
| CT chest | 1 in 3000 |
| CT abdomen | 1 in 3500 |
| CT mandible and maxilla | 1 in 80,000 to 1 in 14,300 |
| Barium swallow | 1 in 13,300 |
| Barium enema | 1 in 9100 |
| Dento-alveolar cone beam CT | 1 in 2,000,000 to 1 in 30,000 |
| Craniofacial cone beam CT | 1 in 670,000 to 1 in 18,200 |
Risk is age-dependent, being highest for the young and lowest for the elderly. The risks shown in Table 7.1 are for a 30-year-old adult. The 2004 European Guidelines on Radiation Protection in Dental Radiology recommend that these should be modified by the multiplication factors shown in Table 7.2, which represent averages for the two sexes. In fact, at all ages risks for females are slightly higher and risks for males slightly lower.
Table 7.2
| Age group (yr) | Multiplication factor for risk |
| <10 | ×3 |
| 10–20 | ×2 |
| 20–30 | ×1.5 |
| 30–50 | ×0.5 |
| 50–80 | ×0.3 |
| 80+ | Negligible risk |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses