7 Lasers in Implant Dentistry
Progress in the design and engineering of dental implants has continued over several decades. These improvements have led to a success rate of 95% or greater over 10 years and beyond.1–3 Thus, implants have become an extremely successful treatment for the replacement of missing teeth.4,5
In the field of dental education, a survey of the curricula of graduate programs shows that dental implantology is taught in oral and maxillofacial surgery (OMS), periodontics, endodontics, and prosthodontics. Most general dentistry residency programs and advanced education in general dentistry (AEGD) programs also include dental implants as part of their curricula. Even orthodontic programs are using dental implants as anchors to aid in moving teeth.6
Generally, lasers aid the clinician in obtaining better visualization of the surgical site by reducing bleeding,7–9 thus often reducing the duration of a given procedure.10 By creating more sterile conditions both during and after surgery, complications and infections are reduced significantly.11
Figure 7-1 shows an incision for a sinus lift using a carbon dioxide (CO2) laser, with the expected excellent postoperative result. Had this same incision been performed with more conventional methods, such as a scalpel, postoperative redness and swelling would have been the rule rather than the exception. For the patient, the benefits of reduced pain and swelling and more rapid healing10,12,13 are invaluable.
Laser Wavelengths
Diode Lasers
Diodes come in different wavelengths, with 810, 940, and 980 nm the most common. The energy from these lasers targets pigments such as hemoglobin and melanin in the soft tissue. The energy is delivered by a fiber in contact mode. By conditioning, or carbonizing, the fiber, the tip heats up to between 500° and 800° C.14 This heat is transferred to the tissue and effectively cuts by vaporizing the tissue. The tissue is vaporized because of the physical contact of the heated tip of the laser with the tissue, rather than from the optical properties of the laser light itself.14,15 The 980-nm wavelength is absorbed into water at a slightly higher rate than the 810-nm wavelength. This makes a 980-nm diode laser potentially safer and therefore more useful around implants.
Absorption of the wavelength is the primary desired laser-tissue interaction; the better the absorption, the less the collateral thermal heat directed toward the implant.7 According to Romanos,16 the 980-nm diodes are safe for titanium surfaces even at higher power settings. Studies show that the 810-nm diode laser creates a high temperature rise at the implant surface.17 Romanos18 also reports that the use of 810-nm diode lasers may damage the surface of the implant. A literature search did not produce documents discussing the 940-nm diode wavelength with regard to implant therapy. For the purposes of this chapter, the 980-nm diode is the only diode considered useful in implant therapy.
Diodes are considered similar to neodymium-doped yttrium-aluminum-garnet (Nd:YAG) lasers in dental applications. The advantage of a diode is less depth of penetration than the Nd:YAG.7 This allows the operator greater control of the laser and reduces the risk of lateral thermal damage. Disadvantages include slowness in speed of cutting, and a gated-pulse delivery mode that translates into potential heat buildup in tissue, leading to lateral thermal damage. Thus, the clinician should be aware of the power density of the diode, especially when close to the surface of implants.17
The fiber delivery system of diode and Nd:YAG lasers allow debris to build up on the fiber tip. This requires frequent cleaning and cleaving of the tip.19 Uncovering implants, if the tissue is relatively thin, is an appropriate use for a diode. A full-thickness flap or incision down to the periosteum to place implants is much more difficult with a diode than with a CO2 laser.
Neodymium:YAG Lasers
The Nd:YAG lasers operate at a wavelength of 1064 nm. These lasers are fiberoptic-delivered contact lasers that generate a free-running pulsed beam of energy. This pulsing mechanism is more sophisticated and the potential for heat penetration even greater than with a diode laser. The 1064-nm wavelength is poorly absorbed in water but readily absorbed into tissue pigments such as hemoglobin and melanin. The Nd:YAG laser is effective at producing coagulation and hemostasis but, because of its penetrating depth of up to 4 mm, has the greatest potential for damaging soft and hard tissues as well as implant surfaces.16 The energy is delivered through the carbonized tip of a fiber, as with a diode laser. However, the maximum peak power emitted by the Nd:YAG is much greater than for a diode and therefore could penetrate the carbonized debris at the laser tip.20
The Nd:YAG laser is useful in periodontal therapy and has had positive effects in pocket therapy.21 However, Block et al.22 report that the Nd:YAG laser energy can melt the surface of implants or remove the surface layer from plasma-coated titanium implants. This laser also produces craters and cracks on different surfaces of titanium. Further, Walsh23 and Chu et al.24 found contraindications for the use of Nd:YAG lasers near implants. Therefore, use of this wavelength is considered inherently unsafe for implant-related procedures or periimplant surgery. Nd:YAG lasers will continue to be used successfully in periodontal therapy.16
Carbon Dioxide Lasers
The CO2 lasers have a wavelength of 10,600 nm and can be delivered in continuous-wave mode, gated-pulse mode, and more recently, in extremely short pulses of high peak power, labeled superpulsed and ultrapulsed (ultraspeed) modes. This wavelength is highly absorbed in water, collagen, and hydroxyapatite19 and thus is extremely efficient for soft tissue vaporization. The delivery system is usually a mirrored handpiece (thus noncontact) at the end of an articulated arm or a waveguide.
The CO2 lasers have been used for decades in surgical procedures because of their speed and efficiency in cutting soft tissue.25 They also offer strong hemostatic and bactericidal effects and create minimal wound contraction, thus minimizing scarring. CO2 lasers also have minimal depth of penetration, reducing lateral thermal damage.12,16 The early devices produced a high degree of carbonization because of the high energy densities created. However, the newer pulsed models reduce the energy density to between 180 and 300 millijoules per square centimeter (mJ/cm2), delivered at an average speed of 400 to 800 microseconds (μsec). This creates less carbonization and charring of tissue and improves the working speed and efficiency of the CO2 laser. This technology has been further refined with the advent of even shorter pulsing with higher peak powers. By increasing the speed of transmission and decreasing the pulsewidth, the laser can cut deeper and carbonize less tissue. Thus, energy density is now reduced to between 50 and 300 mJ/cm2, delivered in speeds of 30 to 80 μsec. This creates an extremely versatile CO2 laser that can safely treat tissue in periodontal pockets and also make surgical incisions up to 4 to 5 mm deep rapidly and efficiently.
The CO2 laser is safe around implants because the energy is absorbed into water and not pigments.26,27 By affecting the intracellular water of bacteria, the CO2 wavelength can safely and effectively treat periimplantitis and mucositis,28 because the energy is not absorbed into the implant’s surface. At the same time, this laser’s hemostatic properties are excellent, allowing the clinician to better visualize the surgical field, often decreasing procedure time and postoperative complications (pain, swelling).29
With the newer devices, the energy is safe when it comes into contact with bone. When exposed to CO2 laser energy, the water molecules on the surface of the bone are dehydrated, forming a thin, carbon layer of approximately 0.1 mm. The resulting surface will no longer absorb energy, and the damage to bone is clinically insignificant.30 However, if the CO2 energy causes hemostasis of bony structures during surgery, curetting the bone to reestablish bleeding for healing is indicated. In the author’s opinion, the newer CO2 laser technology creates the most versatile of all the soft tissue lasers available for implant therapy.
Erbium Lasers
Because it is absorbed into water, the erbium laser is safe around implants and can treat periimplantitis and mucositis safely.31,32 It will leave the bony surface bleeding (for healing), so curettage is not necessary, but it will not harm the implant’s surface.33 It has excellent bactericidal properties because the energy ruptures the cell membranes of bacteria when absorbed into intracellular water.
In summary, erbium lasers are versatile with excellent hard tissue applications, although their soft tissue applications are limited compared with true soft tissue lasers because of poor hemostasis.13,34
Laser Applications in Clinical Practice
Preoperative Frenectomy and Tissue Ablation
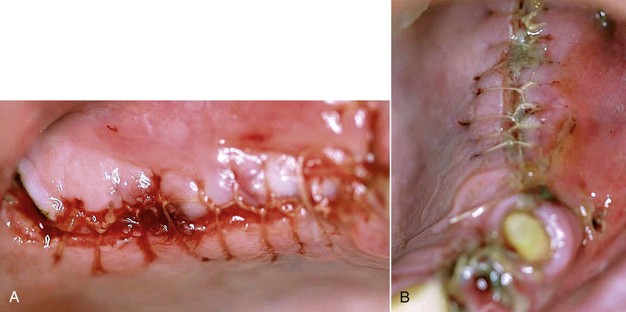
In certain clinical situations, the clinician may need to alter the soft tissue architecture adjacent to the surgical site before the implant is placed. For example, a patient with a high muscle attachment too close to the surgical site would benefit from a frenectomy, to alleviate any tension on the tissue around the implant site. The more complex the surgery, such as bone grafting with a flap, the more important it is to release the muscle tension. The release of muscle tension provides a greater opportunity for success, without sutures pulling, with less postoperative pain and swelling. A frenectomy could be accomplished by any of the soft tissue lasers discussed earlier (Figure 7-2).
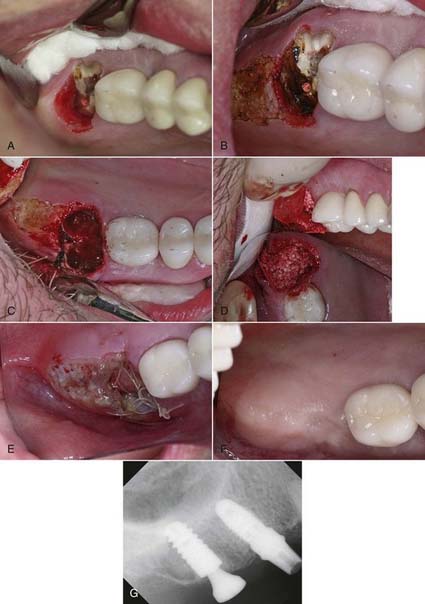
Before tooth extraction, the clinician also may need to alter the soft tissue if it is too thick or uneven in thickness. In Figure 7-3 (p. 18), by ablating 2 to 3 mm of tissue in a broad area distal and palatal to the upper right second molar, the resulting tissue thickness can then conveniently accommodate the abutment and crown with hygienically manageable architecture. The ability to remove tissue easily without bleeding, swelling, or postoperative pain is a tremendous advantage to both the clinician and the patient.
Preparation of Surgical Site
Site preparation is the first step of implant surgery. To prevent contamination of the surgical site, clinicians have used a variety of antimicrobial rinses, including chlorhexidine, before surgery.35,36 However, these decontamination efforts have been only partially effective because of the myriad bacteria in the oral cavity. Furthermore, if the site were to become contaminated with saliva during surgery, it would not be practical or effective to stop and rinse again.
Lasers present an excellent solution to the problem of surgical site contamination. All lasers are bactericidal. The clinician simply needs to expose the surgical site to the laser energy for a few seconds. The bactericidal effects are profound and almost instantaneous, and an implant site can be sterilized.37 Before osteotomy development, the soft tissue can be sterilized much more effectively with a laser than with rinsing or swabbing. Further, if saliva were to contaminate the surgical site during the surgical procedure, it would be simple to release the area and reestablish sterility so that the procedure can continue with the greatest chance for success. The erbium and diode wavelengths can accomplish decontamination if the contact laser physically “touches” every square millimeter of the surface to be sterilized.7,38 This requires a slow, deliberate application; the larger the osteotomy site, the longer the sterilization procedure.
Decontamination and Implant Placement
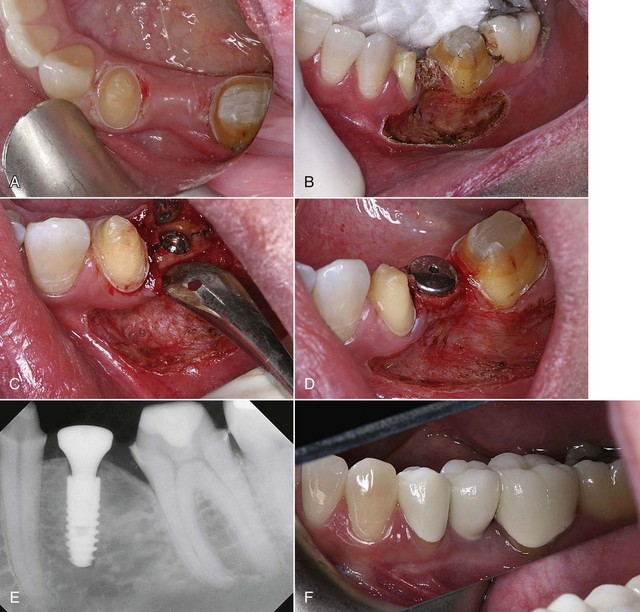
A CO2 laser has a distinct advantage over contact lasers. Because CO2 lasers are noncontact, it is a simple procedure to place a wide-angle handpiece on the CO2 laser and use the laser out of focus, which would increase the spot size on the tissue even more. Sterilization of a large osteotomy site with a CO2 laser takes mere seconds. As the surgery progresses, the clinician and assistant should be diligent in keeping the surgical site free of saliva. In most single-implant scenarios, this can easily be accomplished. However, on large-scale surgeries with multiple implants or large incisions, it may be difficult to maintain a sterile environment. When necessary, laser energy can be redirected to the tissues to decontaminate the surgical site as often as the surgeon deems necessary, using an appropriate power setting for sterilization (Figure 7-4).
As with decontaminating the soft tissue of the surgical site immediately before raising the flap, it is difficult to “touch” all the surfaces of the socket with a diode or Nd:YAG fiber. Of course, neither diode nor Nd:YAG lasers are indicated for use on bone. The erbium wavelengths are effective in removing the remaining soft tissue and decontaminating the bony surfaces at lower power settings with a water-coolant spray.13 Because they are not as effective as other wavelengths in creating hemostasis, erbium wavelengths would leave the bone surfaces bleeding, which enhances healing of the socket, whether an implant or a graft is placed, or the socket is simply left to fill in. The CO2 lasers are also a good choice because they will also remove soft tissue tags and decontaminate bony surfaces, again at low power densities.39 However, CO2 is an excellent hemostatis wavelength, so the effect of hemostasis39 on the tissues must be overcome for healing.35
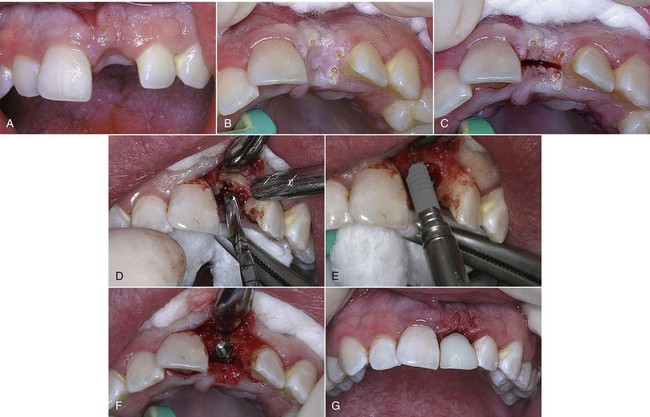
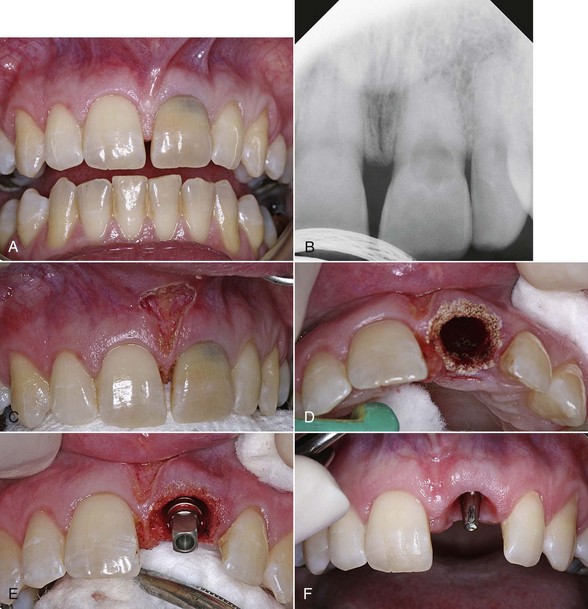
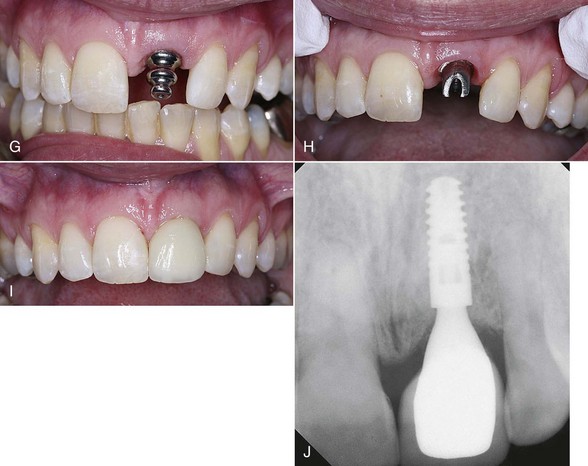
Figure 7-5 (pp. 121-122) illustrates the procedure for decontaminating a surgical site and socket, involving an upper left central incisor planned for extraction. Note the frenum’s proximity to the surgical site. A laser frenectomy is performed to ensure no tension on the tissues immediately surrounding the site. After sterilizing the bony crypt and surrounding marginal tissue, the implant is placed with confidence that the soft and hard tissue of the surgical area is free of disease and bacteria.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses