Chapter 7 Cores
Introduction
A core material replaces missing coronal tooth structure prior to restoration with an indirect extracoronal restoration and helps to stabilize weakened parts of the tooth. It is generally recommended that a core should be considered when more than 50% of the coronal part of the tooth is missing. Existing restorations should be investigated radiographically to diagnose residual caries or caries adjacent to the restoration and evaluated clinically to determine if the existing restoration needs replacing. An assessment of the depth of the cervical margins should highlight potential challenges with preparation and impression taking. Some clinicians recommend the routine removal of existing directly placed restorations to confirm these criteria and replacement with a new core to ensure that the newly placed core is adequately retained (see Chapters 3 and 8 for further discussion). But this is not always necessary, particularly if the restoration was placed recently or there are no clinical or radiographic reasons to do so.
Materials
Composite resin (Figure 7.1)
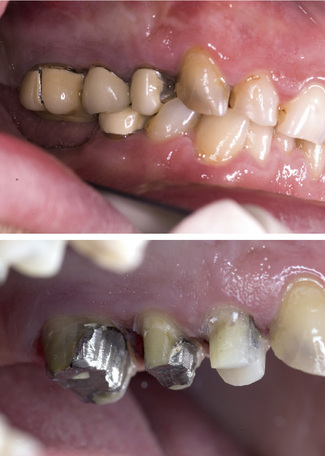
Figure 7.1 Composite resin cores placed in root-filled teeth in the upper right sextant in preparation for all ceramic dentine-bonded crowns. See Chapter 11 for preparations and cemented restorations.
When using composites it is important to prevent moisture contamination with saliva or blood and, therefore, where possible, rubber dam isolation is recommended. Visible light cured composites should be placed in increments to reduce problems with shrinkage on setting. Polymerization contraction may increase the risk of marginal leakage and post-treatment sensitivity. The lower shrink composites that are being introduced may overcome this problem in the future but will need assessing as the materials develop. Water sorption occurs with composites and can lead to expansion of the material. This, however, takes some time to occur and is unlikely to have an impact on the provision of indirect restorations. Any dimensionalchange that does occur will be easily compensated for by the placement of die relief on the master die in the laboratory. Composite is the material of choice for a core when an all-ceramic crown is planned (Figure 7.1).
Glass ionomer cement (GIC)
The original cements contain a fluoroaluminosilicate glass, which reacts with a polyalkenoic acid to form a cement. Many studies have shown that glass ionomer is not sufficiently strong to be used as a core build-up material unless there are two intact walls remaining. There should also be at least 1–2 mm of remaining sound tooth structure coronally that can be prepared as a ferrule. This is an important distinction when compared to composite materials that have greater strength and are more suitable for extensive restorations. Glass ionomer can also be useful as a filler to block out undercuts (e.g. when preparing a tooth for an inlay) and to make good any defects or irregularities in a tooth preparation for an indirect restoration (e.g. when a filling or piece of tooth is lost during tooth preparation; see Chapter 12). Variations of glass ionomer cements have been developed in the past in an attempt to improve the physical properties of the material and these have included the addition of metal particles by a fusion process resulting in a cermet, or addition of amalgam alloy particles resulting in an admix.
Resin-modified glass ionomers (RMGIs)
Advantages of GIC and RMGIs as core materials
Vital teeth
Methods of core retention for vital teeth
Adhesive retention
Amalgam bonding – technique
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses