Chapter 3 Endodontic problems in advanced operative dentistry
General anatomical and physiological considerations
The dentine pulp complex
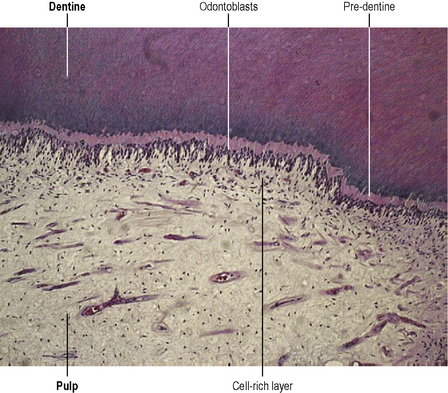
The dentine is derived from complex cells, the odontoblasts, that form a layer at the periphery of the dental pulp (Figure 3.1). The dentine forms a tubular structure, each tubule filled with an odontoblastic process. The tubules are more highly concentrated at the pulp surface than at the enamel–dentine junction. There is a diffusion gradient across the dentine that is related to the number of tubules and the thickness of the dentine. The response of the pulp to progressive injury is by the deposition of dentine (reactionary or tertiary dentine) from the intact odontoblasts and also mineralization within the dentinal tubules (peritubular dentine), thereby reducing their diameter and their ability to allow diffusion through them (Figure 3.2). Thus, the tissue has modulating effects on restorative procedures and that, combined with inherent neutralizing capabilities of tissue fluids from the pulp for both acids and alkalis, means that the amount of residual dentine thickness, after tooth preparation, is inversely proportional to the damage that may occur to the pulp after chemical application to the dentine surface. This is important when considering the use of filling materials, including temporary dressings, and bonding techniques, where an acid is applied to the dentine surface.
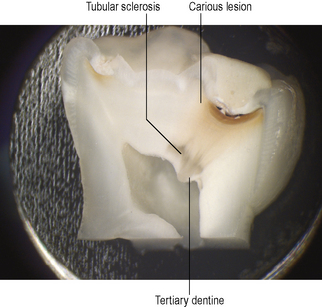
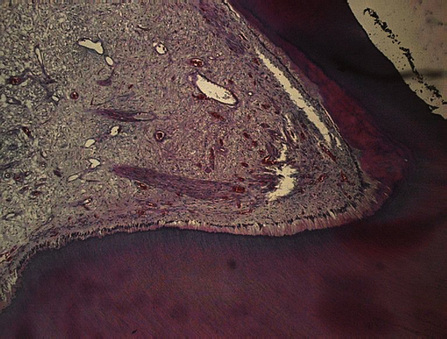
Figure 3.2 A hemisected tooth showing a carious lesion and dentine–pulp complex reactions.
(Courtesy of Dr Chris Longbottom)
Dentine is laid down throughout the life of the tooth: primary dentine as the tooth develops, secondary dentine after eruption of the tooth and tertiary dentine as a result of some insult to the tooth which may result in invasion of the dentine by bacteria or their toxic products. The aetiology of the last is usually dental caries or tooth wear, but may be induced by a restorative procedure (Figures 3.2 and 3.3). Odontoblasts are post-mitotic cells and may easily be damaged during restorative procedures. If this occurs, they are replaced by stem cells from the pulp that differentiate into odontoblast-like cells and lay down mineralized tissue at the injury site. The quality of tertiary dentine is dependent upon the intensity of the insult to the pulp: in general terms, the greater the intensity of the insult (e.g. rapidly progressing caries), the more rapid the formation and the poorer the quality (Figure 3.3). Tertiary dentine is often porous, which makes it susceptible to invasion by micro-organisms and toxins. Therefore, once tertiary dentine is exposed by tooth wear or caries there is a greater risk of subsequent pulp pathology.
Nerves in the pulp
The principal elements of the dental pulp consist of stromal cells, fibroblasts, and an extensive network of nerves, blood vessels and lymphatics (see Figure 3.1). The nerves of the pulp conduct pain sensation. They consist of two main types: myelinated Aδ-fibres that conduct rapid, sharp pain sensation, and unmyelinated C-fibres that transmit the dull aching sensation that is typical of ‘toothache’. In addition, nerves of the autonomic nervous system are also present. Various vasoactive neuropeptides are contained within these nerves. They are produced in the cell bodies of the trigeminal ganglion and are stored in the terminals of the nerves in the pulp. Release of these peptides causes a number of tissue reactions including changes in pulpal blood flow, blood vessel permeability and recruitment of immunocompetent cells as part of the inflammatory reaction. This is known as neurogenic inflammation, which may be damaging to the dental pulp. There are also effects on the growth of cells in the pulp, including fibroblasts and odontoblasts, where release of neuropeptides such as calcitonin gene-related peptide may cause the expression of bone morphogenetic protein-2 transcripts which results in more dentine formation. Recently it has been shown that fibroblasts in the pulp may also release these peptides, particularly substance P.
Pulpal defence mechanisms
The dental pulp is a complex structure that demonstrates various strategies to protect itself from injury. It has been shown to have good powers of healing through the deposition of tertiary dentine and mineralization of the peritubular dentine, even in the presence of bacterial contamination. The peritubular sclerosis gives rise to the histological appearance of translucent dentine (seen in ground sections; Figure 3.2) and the dentine in this region becomes more resistant to diffusion of bacteria and their noxious products. If, however, the damage is so severe that odontoblasts adjacent to the region of irritation die, then the tubules are left empty and are much less resistant to ingress of toxins and bacteria. These tubules are known as dead tracts.
The effect of restorative procedures on the dental pulp
Health of the pulp
Clinically it is difficult to tell the health of the dental pulp, especially if there are no outward clinical signs and symptoms. Previously restored teeth that require further clinical intervention may be at risk. Abou-Rass, in 1982, coined the term ‘the stressed pulp’. This was a pulp that was neither healthy nor diseased but one that might be compromised to such an extent that further operative intervention would lead to irreversible damage. Taking a careful history from the patient regarding previous symptoms from, and treatment to, the tooth should be taken.
Nevertheless, the dental pulp may be injured during operative procedures on the tooth.
Frictional heat
Frictional heat occurs more readily with the following conditions:
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses