6
Treatment of Nonvital Teeth
By definition, a nonvital tooth is a tooth with a necrotic pulp. Pulp necrosis is suspected when a tooth does not react to thermal, electric, or mechanical stimulation, but the definite diagnosis is established only after inspection and probing of the pulp chamber and the root canal. As was discussed above, the necrotic pulp tissue and the root canal space are infected almost without exception, even in teeth which clinically appear intact (Fig. 6.1). Over time, bacteria in the root canal will induce inflammation outside the tooth, resulting in the formation of an apical granuloma or cyst (see p. 33). Treatment of a nonvital tooth, therefore, invariably involves the treatment of an infectious disease process.
The objective of treating a nonvital tooth is to remove the necrotic tissue and tissue breakdown products from the root canal, eliminate infection, seal the root canal bacteria-tight, and thereby establish a functioning tooth in a healthy periodontium. An apical periodontitis which is refractory to conservative endodontic treatment may be treated successfully by a surgical–endodontic approach in which the apical granulation tissue, which may be infected, is curetted out.
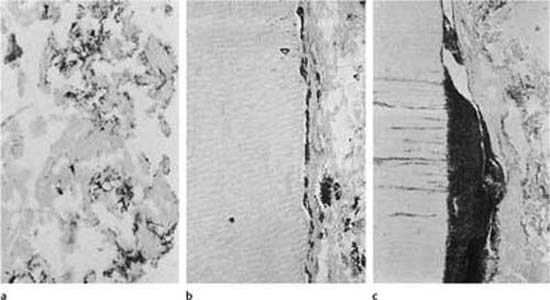
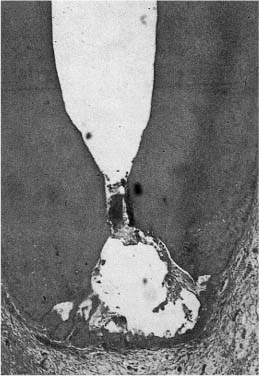
Fig. 6.1
a Colonies of microorganisms in necrotic tissue in a root canal.
b Bacterial colonies in tissue and on the root canal wall.
c Large bacterial colony in the cervical area of a root canal subjacent to dentin exposed to the oral cavity. The root canal of this tooth was sealed to the mouth, and the bacteria have gained access to the canal through the tubules of the exposed dentin (Brown–Brenn stain).
Conservative Endodontic Treatment
Indications
In principle, a conservative approach is indicated in the treatment of all nonvital teeth. Traditionally, there have been some doubts as to whether large periapical lesions would heal following treatment of the root canal, but with modern methods the size of the lesion is of no real concern when the treatment plan is made (Fig. 6.2). However, there is still no definite evidence that periapical cysts respond to nonsurgical treatment, although the circumstantial evidence to that effect is considerable (Fig. 6.2). The main rule, therefore, is that all nonvital teeth with or without apical periodontitis should be treated conservatively.
Treatment Principles
The treatment of the root canal of a nonvital tooth can be divided into three main phases:
1) Chemomechanical instrumentation of the canal with removal of necrotic pulp tissue and bacteria;
2) Final disinfection of the canal and creation of favorable conditions for periapical healing; and
3) Obturation of the canal with materials rendering a bacteria-tight seal.
Chemomechanical instrumentation. The mechanical instrumentation of the root canal is of the utmost importance in the treatment of nonvital teeth. During this phase the necrotic tissue with its colonies of bacteria is physically removed from the main root canal. By enlarging and preparing the canal and giving it its shape for obturation, most bacteria in the dentinal tubules are physically removed as well. However, the mechanical instrumentation obviously has no effect in crevices and lateral or accessory root canals where the instruments do not reach.
The effect of the mechanical instrumentation can be enhanced by the use of chemically active preparations during the instrumentation phase. These preparations are referred to as root canal irrigants, or irrigation solutions, and they are injected continuously or at intervals into the canal during the instrumentation. Different irrigants may be used in an alternate fashion to obtain a combined effect of the medicaments. Thus, medicaments are available which have a cleansing effect, an antibacterial effect, a detoxifying and denaturing effect, and a hard- and soft-tissue dissolving effect. Depending on the clinical situation, different medicaments may be selected. However, the main purpose of a root canal irrigant should be to aid in the removal of necrotic tissue and tissue components, bacteria, blood clots, exudate, and pus. Irrigants with a tissue-dissolving ability have a good cleansing effect, even in crevices and lateral canals where the mechanical instrumentation has no effect. The cleansing action and the tissue dissolution will further lead to an exposure of bacteria that are normally embedded in and protected by necrotic tissue and tissue components or by bacterial extracellular polymeric materials so that the antimicrobial action of the irrigant and other root canal antiseptics is maximized.
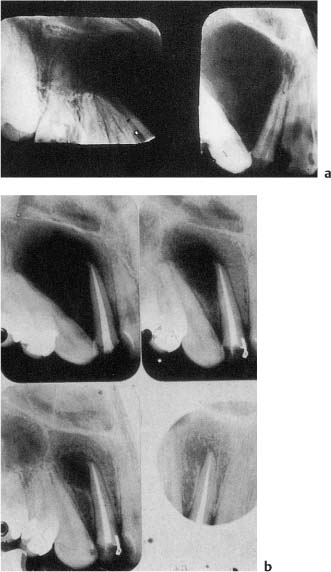
Fig. 6.2
a Radiographs showing a large radiolucent lesion extending from the maxillary right central incisor to the third molar. Note the deviation of the roots of the teeth. The right lateral incisor is non-vital.
b Radiographs showing resolution of the lesion over a 4-year period following endodontic treatment of the lateral incisor.
The classic root canal irrigant is 0.5% sodium hypochlorite at pH 9. This solution was first used in medicine in the form of a drip in the treatment of soldiers with grenade wounds in World War I. By constantly dripping onto a wound surface, the sodium hypochlorite would cleanse the wound, dissolve necrotic tissue at the wound surface, and keep the wound clean and bacteria-free. The effect of the medicament is virtually the same in the root canal. It has an excellent cleansing ability, dissolves and detoxifies necrotic tissue, has a certain antibacterial effect, and is well tolerated by vital tissue. However, the effect of sodium hypochlorite is quickly neutralized by organic components like tissue remnants, blood, and exudate in the root canal, and consequently the irrigant has to be renewed continuously or at frequent intervals. An amount of at least 10 mL should be used during the instrumentation of a single-rooted tooth and comparably more in premolars and molars.
Sodium hypochlorite is used in concentrations between 0.5 and 10%. Since the medicament when used for irrigation is applied in excess, a low concentration will be about as effective as a solution with higher concentration, and it will be less toxic. Thus, a 5% solution is 10 times as toxic as a 0.5% solution, but has only about twice the antibacterial effect. However, in a 5% concentration, sodium hypochlorite has an improved tissue-dissolving effect. This may be beneficial when mechanical removal of tissue is difficult, as in side canals and in teeth with resorption defects.
Chlorhexidine gluconate is an antiseptic that is effective against oral bacteria and has been used in the treatment of periodontal disease for two generations. Lately, chlorhexidine has been used as a root canal irrigant to replace sodium hypochlorite, apparently since sodium hypochloride leaves an oxygen-rich surface in the root canal that might influence negatively the bonding action of resin-based root filling materials. Unlike sodium hypochlorite, chlorhexidine lacks bleaching and tissue dissolving properties. It is therefore not well suited for irrigation of the root canal during the instrumentation phase of the treatment.
Quaternary ammonium compounds are surface-active agents with a soap-like character, and because of these characteristics are widely used as root canal irrigants. They have a bacteriostatic effect and are well tolerated by the tissues. However, like sodium hypochlorite and chlorhexidine, their effect is quickly reduced by organic components, and renewed injection into the canal at frequent intervals is necessary for maximal effect. Like chlorhexidine, the quaternary ammonium compounds lack bleaching and tissue dissolving properties.
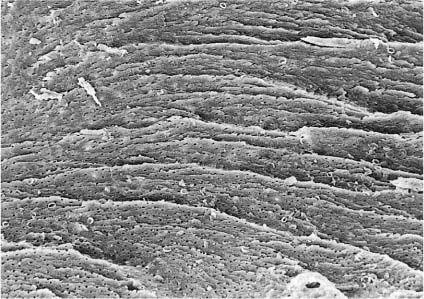
Fig. 6.3 Scanning electron micro-graph of a root canal wall following instrumentation and irrigation with sodium hypochlorite. A smear layer is covering the wall so that the dentin surface cannot be seen (×470).
Fig. 6.4 Scanning electron micro-graph of a root canal wall following instrumentation and irrigation with sodium hypochlorite as well as a final rinse with EDTA. The smear layer is removed and the dentin surface with its tubule orifices can be seen (×300).
Oxidizing agents like a 3% solution of hydrogen peroxide are used as root canal irrigants as well, usually in combination with sodium hypochlorite. However, sodium hypochlorite is as effective when used alone as when combined with hydrogen peroxide, and hydrogen peroxide, therefore, is presently not widely used.
None of the irrigants mentioned above has an effect on hard tissue or on the smear layer of the root canal walls (Fig. 6.3). However, irrigation of the root canal with a chelating agent like ethylenediamine tetraacetic acid (EDTA) which binds Ca++ ions, will effectively remove the smear layer and dentin chips remaining on the canal wall after the instrumentation (Fig. 6.4).
In the root canal ethylenediamine tetraacetic acid (EDTA) is used in a 15% solution at pH 7.4. A certain volume of the solution can only bind a limited number of Ca++ ions, and additional EDTA must be injected into the canal at frequent intervals in order for continued Ca++-binding to take place. EDTA is well tolerated by soft tissues and, from a biological point of view, can be used safely. The most effective use of EDTA as an irrigant is in combination with sodium hypochlorite. These two irrigants supplement each other in that one has its effect on necrotic tissue, bacteria, and other organic components in the root canal system, whereas the other gives a clean root canal wall. Clinically, sodium hypochlorite and EDTA may be used alternately during the instrumentation phase. Excellent results are also obtained when sodium hypochlorite is used during the instrumentation, and EDTA is used for a final irrigation of the canal after the instrumentation is completed.
A new irrigating solution, MTAD, containing a mixture of a tetracycline isomer, an acid and a detergent has come on the market. It was initially meant to replace both sodium hypochlorite and EDTA, but is now recommended to be used together with sodium hypochlorite. The acid in MTAD may remove the smear layer of the canal walls, but the antibacterial effect of the combined use of sodium hypochlorite and EDTA is reported to be superior to the effect of the MTAD and sodium hypochlorite combination.
If at all possible, the chemomechanical instrumentation of the root canal should be completed in the first visit. If a partial instrumentation is done, the environment in the canal is changed and the balance that existed in the bacterial flora is altered as well. Chances are that certain pathogenic bacteria now may have an opportunity to dominate the infection and cause a flare-up or exacerbation of the periapical inflammation.
Iatrogenic flare-ups occur first and foremost during treatment of teeth with apical periodontitis, and with optimal treatment will be seen in about 5% of such teeth. The frequency with which flare-ups occur is an excellent indicator of a dentist’s effectiveness and carefulness in endodontic treatment of nonvital teeth. If the overall frequency is much higher than 2–3% (5% in teeth with apical periodontitis), it is a clear message that something is wrong with the dentist’s therapeutic approach, either conceptually, clinically, or both.
Disinfection of the root canal. A thorough and complete chemomechanical instrumentation in the first visit may render the root canal system bacteria-free. However, in most instances it results in only an effective reduction in the number of bacteria, and an antiseptic medicament must be applied to the root canal for a period of time in order to assure that the last remnants of the root canal flora are eliminated.
Presently, there is disagreement about how important this is. It is felt by many that it is of no clinical consequence that bacteria are left in the root canal system if only the root canal is obturated bacteria-tight. Remaining bacteria will then be sealed in the root and have no effect on the periapical tissues. However, from a medical point of view this concept may seem hard to defend. Firstly, pathogenic bacteria should not be left in a patient if they can be eradicated, and secondly, not all bacteria may be “blocked off,” but be present in root canal deltas and lateral canals that are in continuity with the periodontium. With our present methods and techniques, therefore, endodontic treatment of a nonvital tooth should as a rule not be completed in the first visit, but in a second visit after a period of final disinfection of the root and root canal system.
In traditional endodontic treatment of nonvital teeth, little emphasis was put on instrumentation and tissue management, and much emphasis was put on disinfection. Sophisticated disinfection methods such as diathermy and iontophoresis were advocated, and a large number of imaginative concoctions of powerful antiseptics were used as well. Today, we know that medicaments which are effective against microorganisms also, as a rule, kill human cells, and consequently may cause tissue damage. The damage which is caused by uncritical use of medicaments in the root canal may, therefore, at times exceed the advantages of their use. Recognition of this fact led to attempts to find antiseptics which have the desired effect on the microorganisms of the root canal system without causing unacceptable injury to the periapical tissues (selective toxicity). Iodine and chlorine preparations as well as quaternary ammonium compounds and chlorhexidine have shown the best results in these studies, and from the point of view of selective toxicity, are suitable intracanal medicaments in the treatment of nonvital teeth. Formocresol and monochlorphenol are other medicaments used widely for this purpose (Fig. 6.5).
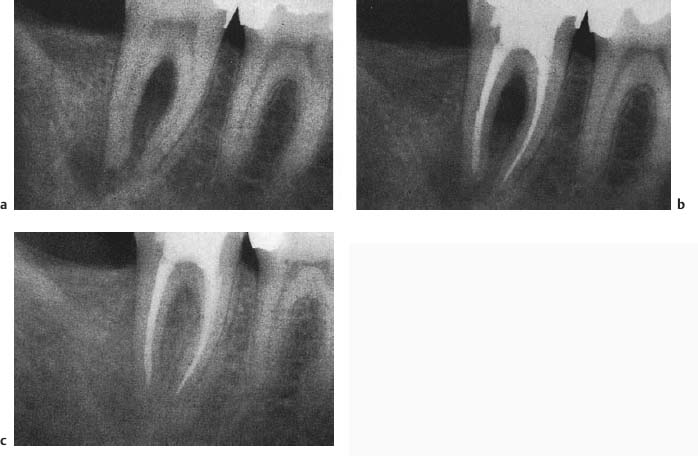
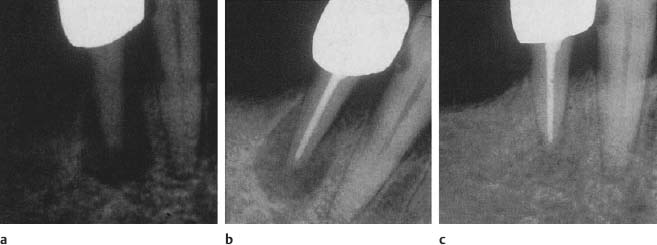
Fig. 6.5
a Radiograph of a mandibular molar with apical and interradicular periodontitis. The tooth was completely instrumented under irrigation with sodium hypochlorite and EDTA in the first visit and formocresol was placed in the pulp chamber by means of a cotton pellet.
b In the second visit after 1 week, the patient was asymptomatic and the root canals were dry. The canals were then obdurated.
c After 6 months, resolution of the lesion is observed.
Fig. 6.6
a A cotton pellet with fresh formocresol is placed on a pour plate with Streptococcus species. After incubation for 24 h, a zone of inhibition of the growth of the bacteria is seen surrounding the cotton pellet with the medicament.
b Cotton pellet with formocresol which was sealed in the pulp chamber of an instrumented, nonvital dog tooth for 24h is placed on a pour plate with Streptococcus species. After incubation for 24h, the bacteria are seen growing in contact with the cotton pellet. The formocresol had lost its antibacterial effect.
Although the selective toxicity of the medicaments mentioned above varies, and thereby their suitability for use, they have one feature in common, namely the fact that they are quickly rendered inactive by contact with the dental tissues and tissue fluids, usually within 24 hours (Fig. 6.6). Thus, if any of these medicaments is used in the root canal, the canal will be unprotected 1 day after the placement of the medicament or even sooner. This means that if the bacteria in the root canal system are not killed during the first day, surviving organisms will begin to multiply again, since the canal will contain tissue fluids, exudate, and blood, which constitute an excellent substrate for bacterial growth. Also, when the root canal is no longer protected by the antiseptic, it may become reinfected by bacteria from outside the root canal system.
Ideally, a root canal antiseptic should have a long-lasting antimicrobial effect. In the future, intracanal medicaments in controlled-release delivery systems will undoubtedly be available (Fig. 6.7). At present, prolonged antibacterial action can be readily and effectively obtained by filling the root canal with a paste of calcium hydroxide and isotonic saline. The use of calcium hydroxide in the treatment of nonvital teeth developed empirically. It was noted that when placed in the root canal, calcium hydroxide has a striking effect on periapical exudation. This was taken as evidence that the material has an anti-inflammatory effect, i.e., it has the ability to bring a periapical inflammation from the exudative stage to a stage of repair. It was also noted that periapical lesions quickly show radio-graphic signs of healing in teeth with calcium hydroxide in the root canal, and that complete repair regularly occurs in 2–3 months (Fig. 6.8).
The modes of action of calcium hydroxide are only partially understood. When mixed with saline, the resulting paste has a pH of 12.5. Moreover, calcium hydroxide has a disassociation coefficient of 0.17, which means that if placed in the root canal, it will become ionized and dissolve slowly and steadily in the fluids of the dentinal tubules, the lateral and accessory canals, and the periapical tissue. In other words, there is a controlled, long-lasting release of Ca++ and OH– ions from the paste in the root canal. Consequently, calcium hydroxide will have a controlled, long-lasting therapeutic effect.
The therapeutic effect of calcium hydroxide is clearly related to the hydroxyl ions, which will lead to a lowered oxygen tension and an increase in the pH in the inflamed periapical tissue. A low oxygen tension in the tissue favors bone formation and repair, and a favorable effect of an alkaline environment on mineralization is also evidenced by the high pH found in the growth plate at the mineralization front of bones. A possible therapeutic effect of the calcium ions is less well understood, but apparently they may have a stimulating effect on certain alkaline phosphatases, which are enzymes connected with hard-tissue formation. It is also possible that the calcium ions have a beneficial effect on the local immune response.
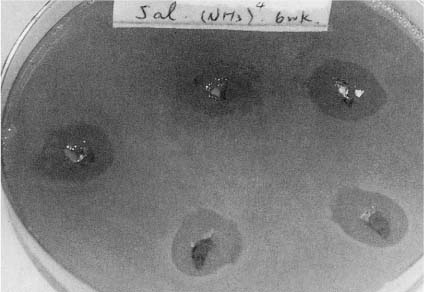
Fig. 6.7 Antiseptic in a controlled-release delivery system. The dispensers were sealed in the pulp chamber of instrumented, nonvital dog teeth for 6 weeks and were, after removal, placed on a pour plate with Streptococcus species. After incubation for 24 h, zones of inhibition of bacterial growth are seen surrounding the dispensers, indicating that the antiseptic is still being released after 6 weeks of contact with dentin and tissue fluids in the root canal.
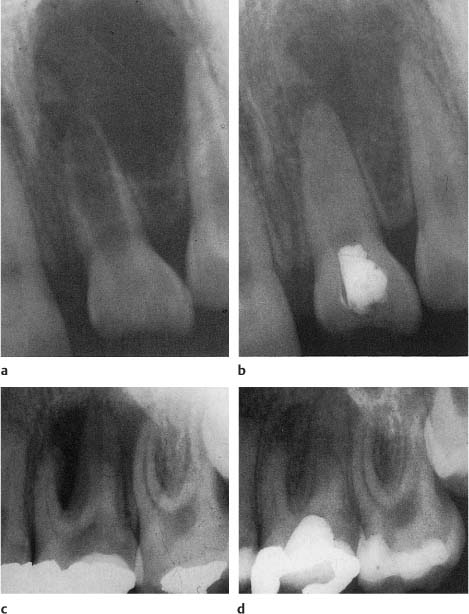
Fig. 6.8
a Radiograph of a maxillary incisor with root fracture and a large radiolucency in conjunction with the fracture line. Calcium hydroxide treatment of the coronal fragment (apexification) is initiated.
b After 2 months, near complete resolution of the radiolucent lesion is observed.
c Radiograph of a maxillary molar with interradicular periodontitis and suspected oral communication. Long-term calcium hydroxide treatment is initiated.
d After 10 weeks, resolution of the radiolucent lesion is evident.
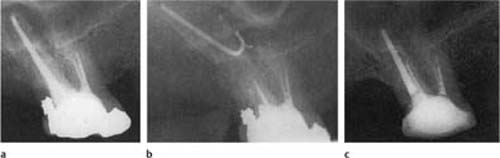
Fig. 6.9
a Radiograph of a maxillary molar with apical periodontitis and a palatal fistula from the palatal root. The tooth was completely instrumented in the first visit (buccal root canals calcified) and treated with calcium hydroxide for 3 weeks. At the second visit, the patient was asymptomatic, the fistula had closed, and the root canals were obturated.
b At 3-month control, the palatal fistula has recurred, the root filling is removed from the palatal canal and long-term calcium hydroxide treatment is initiated.
c The fistula closed immediately, and after 3 months the periapical lesion had resolved. Only then was the palatal root canal again permanently obturated.
With a pH of 12.5, a calcium hydroxide paste has an excellent and exceptionally broad antibacterial effect. No known endodontopathogens can survive at this pH, and will be killed instantly when directly exposed to the paste. However, in crevices of the main canal and especially in lateral and accessory canals where the chemomechanical instrumentation has only a limited effect, the bacteria may be embedded in and protected by necrotic tissue components and bacterial extracellular material, and may survive the influence of an antiseptic for considerable time. Even in such inaccessible areas of the root canal system, the bacteria can be expected to eventually die out under the constant and long-lasting influence of calcium hydroxide, but a long-term exposure to the drug, i. e., weeks and even months may be necessary in some instances (Fig. 6.9).
The long-lasting antibacterial effect of the calcium hydroxide paste is highly dependent on a constant, high alkalinity of the paste. Thus, rather common root canal organisms like Enterococcus faecalis may survive in an environment with a pH up to 11. Blood, exudate, dental tissues, and tissue fluids will readily lower the pH of the calcium hydroxide paste, and in teeth with severe exudation from the periapical tissues, the paste in the apical part of the canal may have a pH of 8 within 2–3 weeks after placement. The paste will then have lost its antibacterial effect. However, due to its anti-inflammatory effect, even a severe periapical exudation may have stopped at this time. The “used” calcium hydroxide paste is, therefore, removed, the root canal is irrigated and dried, and new, clean calcium hydroxide paste with the right alkalinity is packed into the canal. Bacteria anywhere in the root canal system that might have survived the first application of calcium hydroxide should now effectively be killed. However, in most instances the root canal can be dried to such an extent at the first visit that a one-time application of calcium hydroxide for 2–3 weeks is sufficient to obtain a bacteria-free root canal system.
An additional aspect of the therapeutic effect of calcium hydroxide is its ability to denature proteins. Necrotic tissue influenced by calcium hydroxide will swell to twice its size and be dissolved by sodium hypochlorite twice as fast as tissue untreated by calcium hydroxide. Tissue remnants left behind in the root canal after the chemomechanical instrumentation can, therefore, readily be dissolved and rinsed out with sodium hypochlorite following a period of a week or longer with calcium hydroxide in the canal. Similarly, the content of lateral and accessory canals and other areas in the root canal system which cannot be reached by instruments will be influenced by the combined action of calcium hydroxide and sodium hypochlorite, resulting in an optimally cleaned root canal system.
It also appears that the actual filling of the root canal with a material with the properties of a calcium hydroxide–saline paste is beneficial for the periapical repair processes. When a traditional antiseptic is applied to an empty root canal by means of a cotton pellet, an exudate consisting of tissue fluids, cells, bacteria, and bacterial products and components will fill the canal. The exudate as such may maintain a periapical inflammation, but most importantly it is an excellent substrate for bacterial growth. The possibilities of reinfection of the root canal between visits under these circumstances are quite real. The use of calcium hydroxide, on the other hand, will effectively prevent exudate from filling the root canal, and by virtue of its presence and its long-lasting antibacterial effect will prevent reinfection of the canal.
Obturation of the root canal. The effectiveness of the chemomechanical instrumentation and disinfection of the root canal is normally evaluated clinically. If the patient is asymptomatic and the root canal remains dry after the intracanal medicament has been removed and the canal has been irrigated and dried, it is assumed that the periapical inflammation is at a stage of repair. This can be expected at the first visit following the visit with complete instrumentation of the canal, i. e., usually in the second visit, and the root canal can then be permanently obturated.
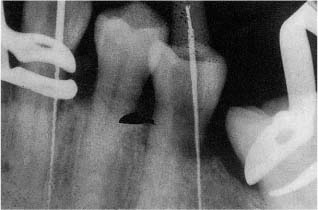
Fig. 6.10 Radiograph showing perforations of teeth in the cervical area during preparation of access cavities under a rubber dam. The operator has clearly misjudged the longitudinal direction of the teeth.
Sometimes the criteria for final obturation are not achieved in two visits. The reasons for the unfavorable tissue reactions should then be determined, if possible, before the treatment is continued. The primary concern is whether the chemomechanical instrumentation has been performed satisfactorily. The canal may not have been enlarged enough or a canal may have been missed. In any case, insufficient instrumentation with remaining infection is by far the dominating cause of persistent tissue reactions. Microbiological culturing of the root canal content may provide useful information in such instances (see p.117).
If infection is suspected at the second visit, and the instrumentation of the root canal appears to have been carried out well, the problem may be caused by reinfection because of a leaky temporary filling, inadequate old restorations, caries, cracks, fractures, or similar causes. Therefore, after renewed chemomechanical instrumentation and application of the intracanal antiseptic, the coronal seal must be checked most carefully. Remember that a temporary filling should preferably be 4–5 mm thick in order to provide a safe bacteria-tight seal. If symptoms, exudation, or fistulae persist after repeated treatment of a tooth, it is useless to keep changing the intracanal dressing. An analysis of the probable causes of the problem must be carried out and effective therapeutic measures taken (see p.116).
Clinical Considerations
The access cavity is prepared in the tooth and the root canal orifices are located before a rubber dam is applied. This is even more important in nonvital than in vital teeth in that, in these teeth, the root canals are often calcified because of a long-standing chronic inflammation before the pulp finally necrotizes. It may, therefore, be difficult to localize the root canal orifices, and calcified tissue may have to be removed with rotating instruments. In this situation it is helpful to see the entire tooth and the neighboring teeth with adjacent structures for orientation purposes so as to avoid root perforations and other mishaps (Fig. 6.10).
When the orifices are located, a rubber dam is applied and the field of operation is disinfected as described for vital teeth (see p.101). The treatment then continues with sterile instruments and under surgical–aseptic conditions. First, the length of the root canal is established by means of a tooth-length radiograph taken with an instrument in the root canal or with an apex locator. Theoretically, the root canal of a nonvital tooth should be instrumented to the apical foramen to ensure the removal of all necrotic tissue. However, as with vital teeth, it is usually not possible to determine the exact position of the foramen. Therefore, the practical rule to follow in nonvital teeth is to end the mechanical instrumentation of the root canal 1 mm short of the radiographic apex. In this way practically all necrotic tissue will be removed, and in most instances the instrumentation will be confined within the canal with the possibility of creating an apical stop to facilitate the obturation of the canal (Fig. 6.11).
Remember that over-instrumentation of the root canal of nonvital teeth is an effective way to carry bacteria to the periapical tissues, often resulting in an exacerbation of an asymptomatic periapical inflammation.
As discussed above, the root canal will not routinely be bacteria-free after the chemomechanical instrumentation. Consequently, the root canal in nonvital teeth should not be obturated in the first visit. For further disinfection of the canal it is important to understand that the antiseptic medicament used has to come in contact with the bacteria in order to kill them. Thus, vaporizing medicaments like iodine- and formalin-containing drugs may be applied to the pulp chamber, whereas nonvaporizing medicaments will have to fill the root canal system to have an effect.
If calcium hydroxide is used, it is mixed with saline to a creamy mix. It is applied to the pulp chamber with a plastic instrument and introduced into the root canal by means of a lentulo. When the canal appears filled, excess water is removed with paper points and the paste is condensed with a plugger or similar instrument. Additional calcium hydroxide paste is then applied with the lentulo and condensed at the canal orifice with cotton pellets. When the canal is adequately filled, excess paste on the walls of the access cavity is carefully removed and a temporary filling which gives a bacteria-tight seal is finally placed in the access cavity.
The time between the first and second visit depends on the type of intracanal antiseptic used. With a traditional antiseptic the time should be short (days) since the medicament loses its effect within hours of placement in the canal. A third appointment may, therefore, become necessary before the criteria for obturating the root canal are fulfilled (asymptomatic patient, dry root canal). An antiseptic in a controlled-release delivery system offers greater convenience because the time interval between appointments may be considerably increased. Possible symptoms of an apical periodontitis may then have time to subside and periapical exudation may stop. At present a calcium hydroxide–saline paste appears to fulfill these requirements of an intracanal antiseptic the best. The paste in the canal will act as a depot and continuously release Ca++ and OH– ions for weeks and months. A period of 2–3 weeks between appointments is as a rule enough in that the pH in the dentinal tubules reaches its peak during this time. The bacteria in the root canal system and the root dentin should then be killed and the peri-apical inflammation will have subsided so that the root canal can be dried and filled under optimal conditions. This method, which is referred to as short-term calcium hydroxide treatment, makes for effective and problem-free endodontics (Fig. 6.12).
Fig. 6.11 Section of a nonvital dog tooth. The instrumentation of the root canal has been carried out according to the apical box method and ends 1 mm short of the apex. In this way, a stop to facilitate the obturation of the canal has been created in spite of apical resorption. The root canal is clean and if the infection has been eliminated, periapical healing will be predictable and fast (hematoxylineosin).
Fig. 6.12
a Radiograph of a mandibular premolar with asymptomatic apical periodontitis. The root canal is completely instrumented in the first visit and calcium hydroxide is packed into the canal to complete the disinfection of the root. (When an ill-fitting crown is not removed until after the endodontic treatment, it is imperative to establish an intracoronal bacteria-tight seal.)
b In the second visit after 3 weeks, the patient remains asymptomatic and no periapical exudation is seen. The root canal is then obturated.
c At the 3-month control, resolution of the periradicular lesion is evident.
Long-term calcium hydroxide treatment may be the method of choice in special cases. It is used in the treatment of teeth with large periapical lesions, severe periapical exudation, incomplete root formation, progressive external inflammatory resorption, and in the retreatment of teeth where conventional treatment has failed. In other words, the long-term calcium hydroxide method is used in situations in which we want to maximize the effect of our treatment.
With this method the tooth is treated with calcium hydroxide until periapical healing is complete or is at least well under way, i. e., for 3–12 months or even longer (Figs. 6.8, 6.9, 6.13, 6.14). The medication is changed after 2–3 weeks and then if necessary after 3 months. The purpose of the treatment is to maximize the antibacterial treatment of the root canal system and to favorably influence hard- and soft-tissue repair processes in the tooth and periradicular tissues. An additional benefit of the method is that apical repair or, just as importantly, lack of repair can be determined relatively quickly. This may be important for the overall treatment plan of the patient.
Antibiotics should be given prophylactically in conjunction with endodontic treatment of nonvital teeth to certain patients. They are patients with heart valve injury, acute glomerulonephritis, or diabetes that is difficult to control, patients on steroid therapy, and a steadily increasing number of patients who have undergone open heart surgery. Patients with implants and pacemakers should also be included in this group. Prophylactic antibiotic treatment should be performed with bactericidal medicaments, normally amoxicillin. The dose is given a half to 1 hour before the treatment begins, which means that the patient may receive the medicament in the waiting room. The regime for prophylactic use of antibiotics may differ somewhat from country to country. For legal reasons, therefore, one should use the regime exactly as recommended by local authorities, heart associations, or similar institutions.
Follow-up Examinations and Prognosis
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses